The Functional Properties of Protein Denaturation Coagulation The










- Slides: 12

The Functional Properties of Protein • Denaturation • Coagulation • The factors contributing to denaturation and coagulation

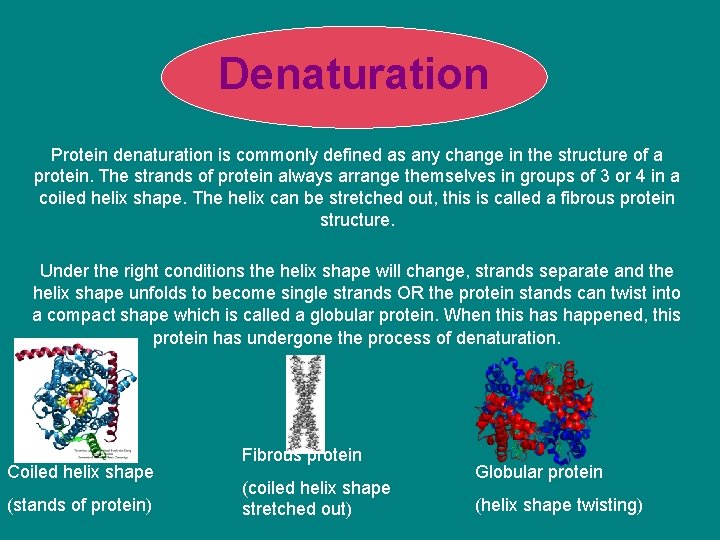
Denaturation Protein denaturation is commonly defined as any change in the structure of a protein. The strands of protein always arrange themselves in groups of 3 or 4 in a coiled helix shape. The helix can be stretched out, this is called a fibrous protein structure. Under the right conditions the helix shape will change, strands separate and the helix shape unfolds to become single strands OR the protein stands can twist into a compact shape which is called a globular protein. When this happened, this protein has undergone the process of denaturation. Coiled helix shape (stands of protein) Fibrous protein (coiled helix shape stretched out) Globular protein (helix shape twisting)

Coagulation • Once a protein has undergone the process of denaturation it will separate from other nutrients and clump into a semi-soft, solid like substance. The process by which a protein becomes solid is called coagulation. • Coagulations turns proteins into solids not through evaporation by some kind of chemical reaction. • Separated strands of amino acids reform into groups of three with new cross bonds between them. • Liquid can then trap in between these coagulated proteins to make a gel or elastic solid. E. g. When gelatine turns solid on cooling. • If liquid is not trapped in between coagulated proteins it then forms clots that float in a liquid. E. g. This could happen in a stirred custard if temperatures are not controlled.

• Proteins will denature and coagulate when one or more of the necessary conditions exist. • An example of coagulation is the solidifying of the egg by the application of heat • Coagulation and denaturation can make foods more pleasant to eat and easier to digest.

Factors contributing to denaturation and coagulation

Temperature • Heat causes proteins to denature • Higher temperatures will set proteins more quickly and cause them to coagulate. For example; An egg is cracked into a frying pan, the heat causes the protein strands in the egg to uncoil (Denaturation) More heat causes the proteins strands to reform in a new bond & the egg sets and form a solid (Coagulation)

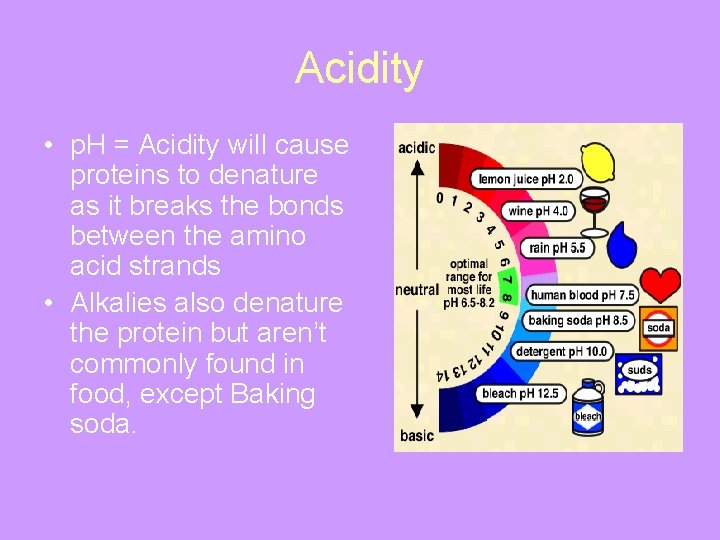
Acidity • p. H = Acidity will cause proteins to denature as it breaks the bonds between the amino acid strands • Alkalies also denature the protein but aren’t commonly found in food, except Baking soda.

Agitation • Mechanical actions; (kneading dough, whipping cream, or beating eggs) cause protein strands to stretch • Too much mechanical action = protein strands to break and will denature the protein Example: Beating egg whites – if over beaten the mixture will clot

Enzymes • Enzymes will speed up the chemical reaction time, i. e. the denaturation process. Example: Soaking meat in a marinade containing enzymes will speed up the time taken for the meat to cook when heated.

Sodium Concentration • Salt attracts water and will help to keep protein moist • If too much salt it used then the bonds between amino acids will break and denature Example: Meat with too much salt on the outside, when cooked will cause meat to toughen

Freezing • When heat is removed during freezing, proteins with a high water content can denature • This is due to the water being drawn out of the cells, and salt will concentrate. Amino acid strands will separate and denature.

Influence of sugar on protein structure • If sugar is added when a protein is heated; it will protect the protein structure and allow it to tolerate higher temperatures • For example; the sugar content in custard means that more heat is necessary to coagulate the protein in the eggs and milk, and thicken the custard