The Faint Young Sun Problem Jim Kasting Astrobiology

The Faint Young Sun Problem Jim Kasting Astrobiology 474
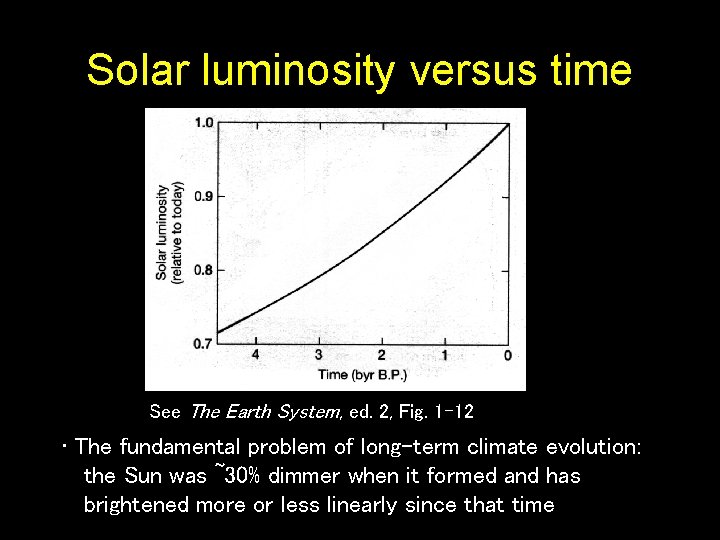
Solar luminosity versus time See The Earth System, ed. 2, Fig. 1 -12 • The fundamental problem of long-term climate evolution: the Sun was ~30% dimmer when it formed and has brightened more or less linearly since that time
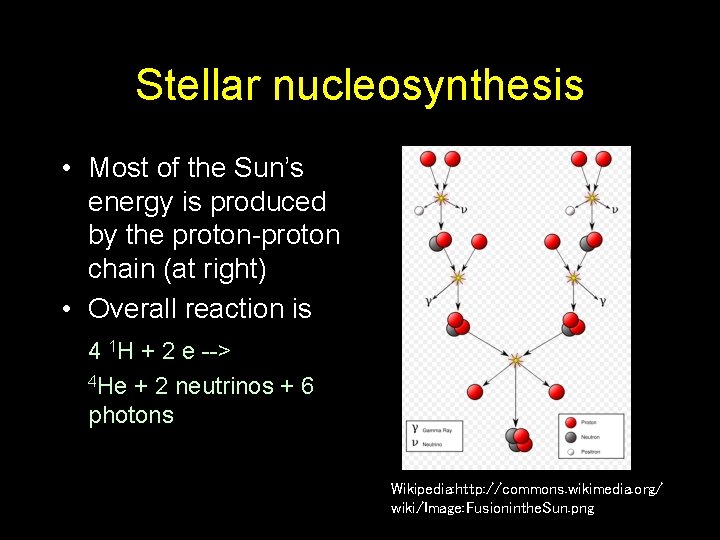
Stellar nucleosynthesis • Most of the Sun’s energy is produced by the proton-proton chain (at right) • Overall reaction is 4 1 H + 2 e --> 4 He + 2 neutrinos + 6 photons Wikipedia: http: //commons. wikimedia. org/ wiki/Image: Fusioninthe. Sun. png
Why the Sun gets brighter with time • H fuses to form He in the core • Core becomes denser • Core contracts and heats up • Fusion reactions proceed faster • More energy is produced more energy needs to be emitted

• Now, consider the implications for Earth’s climate • This problem was first pointed out in 1972 by Carl Sagan and George Mullen
![The faint young Sun problem Te = effective radiating temperature = [S(1 -A)/4 ]1/4 The faint young Sun problem Te = effective radiating temperature = [S(1 -A)/4 ]1/4](http://slidetodoc.com/presentation_image_h2/64b51b38569f02af0c4d534cf38b5239/image-6.jpg)
The faint young Sun problem Te = effective radiating temperature = [S(1 -A)/4 ]1/4 TS = average surface temperature Kasting et al. , Scientific American (1988)

Question: • How did Earth remain habitable during its early history when the Sun was less bright? Was it Gaia, or was it something else?

The Gaia hypothesis • According to this hypothesis, organisms (plants and algae pulled CO 2 out of the atmosphere at precisely the right rate to offset increasing solar luminosity CO 2 + H 2 O CH 2 O + O 2 • Is this idea teleological? http: //www. paleothea. com/Majors. html

• Before trying to solve the faint young Sun problem, let’s look briefly at Earth’s longterm climate history

Ice age (Pleistocene) Dinosaurs go extinct Phanerozoic Time First dinosaurs Ice age Age of fishes First vascular plants on land Ice age First shelly fossils

Geologic time First shelly fossils (Cambrian explosion) Snowball Earth ice ages Warm Rise of atmospheric O 2 (Ice age) Ice ages Ice age (? Questionable)

• Now, let’s return to the FYS problem…

Possible solutions to the FYS problem • Think back to the planetary energy balance equation Te 4 = S (1 – A) 4 • Albedo changes? – A possible feedback involving biogenic sulfur gases and cloud condensation nuclei has been suggested (Rosing et al. , Nature, 2010), but it is probably not strong enough to solve the problem • Geothermal heat? – Too small (0. 09 W/m 2 vs. ~240 W/m 2 from absorbed sunlight

Possible solutions to the FYS problem • Increasing the greenhouse effect also works Ts = T e + Tg • Possible greenhouse gases – NH 3: Doesn’t work very well (photolyzes rapidly) – CO 2: Works! (supplied by volcanoes) – CH 4: Also works (probably requires life) – H 2: This is a new (and surprising!) idea • Sagan and Mullen (1972) liked methane (CH 4) and ammonia (NH 3) because they were aware that O 2 levels were low on the early Earth. We’ll return to that thought later.

• To get a handle on climate evolution, it’s important to understand climate feedbacks • For this, it is useful to define some systems notation…
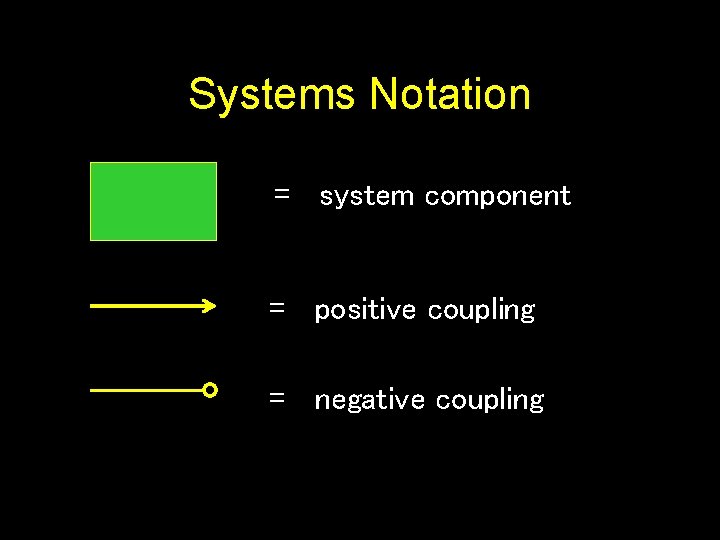
Systems Notation = system component = positive coupling = negative coupling
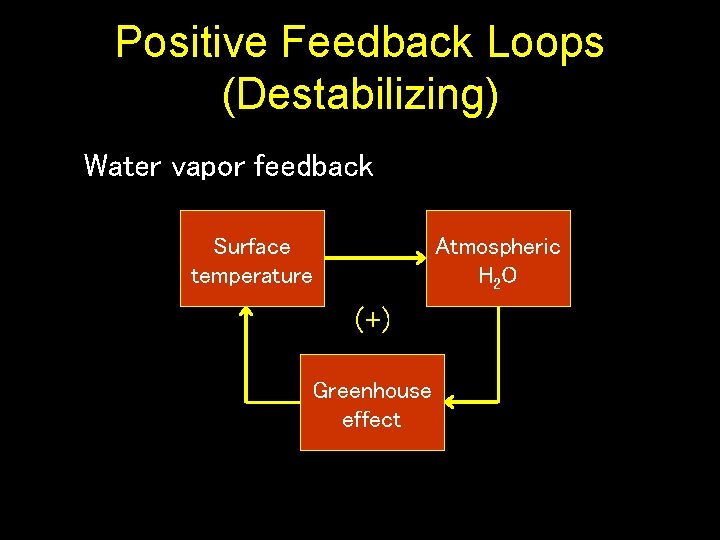
Positive Feedback Loops (Destabilizing) Water vapor feedback Surface temperature Atmospheric H 2 O (+) Greenhouse effect
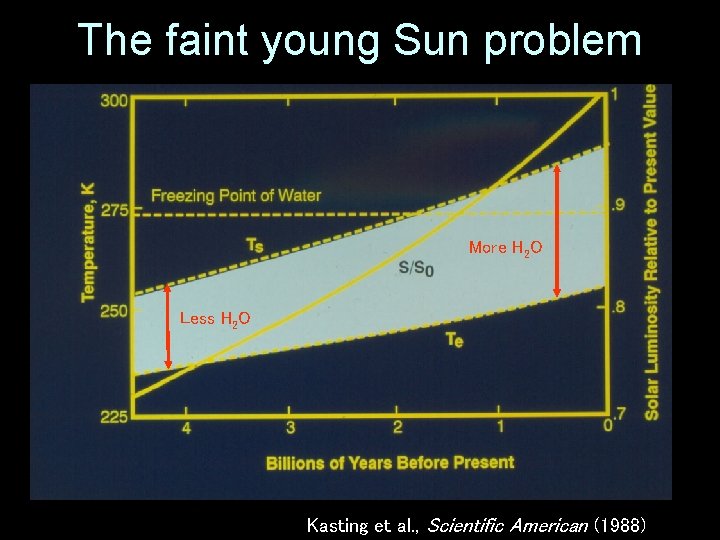
The faint young Sun problem More H 2 O Less H 2 O Kasting et al. , Scientific American (1988)

Positive feedback loops (destabilizing) Snow/ice albedo feedback Surface temperature Snow and ice cover (+) Planetary albedo • If this feedback were included in models of early climate, the FYS problem would be even worse

• So, we need one or more negative feedback loops to stabilize Earth’s climate • What could these be?
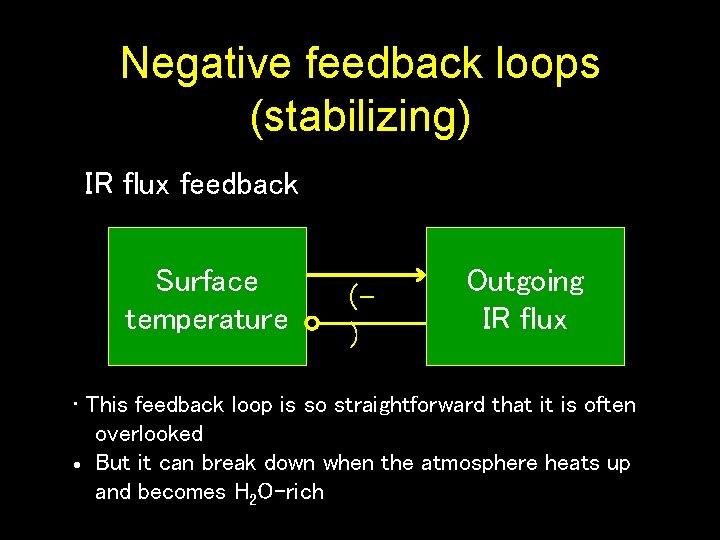
Negative feedback loops (stabilizing) IR flux feedback Surface temperature () Outgoing IR flux • This feedback loop is so straightforward that it is often overlooked But it can break down when the atmosphere heats up and becomes H 2 O-rich

• There must also be negative feedbacks that operate on long time scales – Need this to counter the change in solar luminosity • What could these be?

The carbonate-silicate cycle • Atmospheric CO 2 builds up as the climate cools, so higher atmospheric CO 2 is a natural solution to the FYS problem

Negative feedback loops (stabilizing) The carbonate-silicate cycle feedback Rainfall Silicate weathering rate Surface temperature (−) Greenhouse effect Atmospheric CO 2
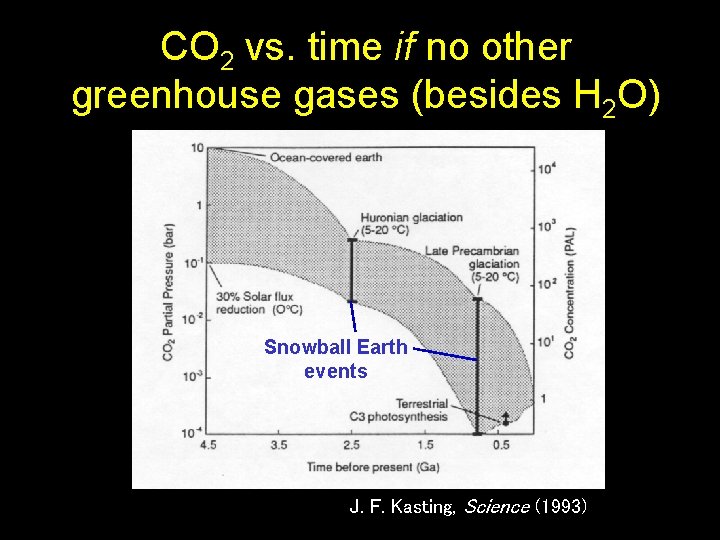
CO 2 vs. time if no other greenhouse gases (besides H 2 O) Snowball Earth events J. F. Kasting, Science (1993)
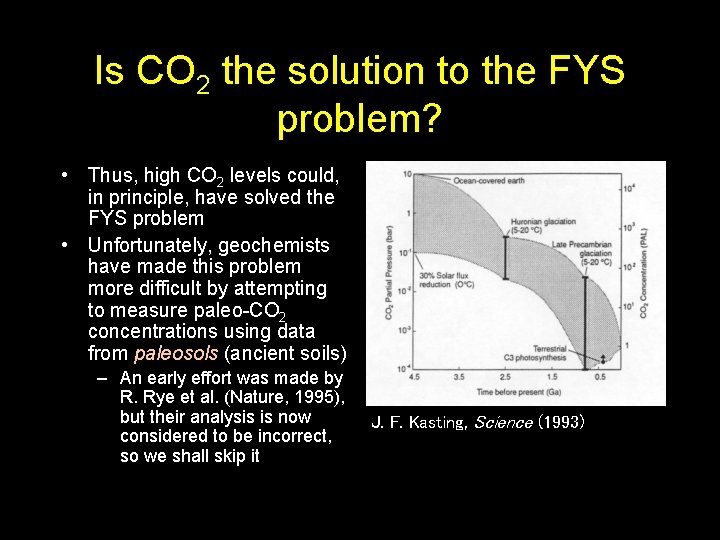
Is CO 2 the solution to the FYS problem? • Thus, high CO 2 levels could, in principle, have solved the FYS problem • Unfortunately, geochemists have made this problem more difficult by attempting to measure paleo-CO 2 concentrations using data from paleosols (ancient soils) – An early effort was made by R. Rye et al. (Nature, 1995), but their analysis is now considered to be incorrect, so we shall skip it J. F. Kasting, Science (1993)
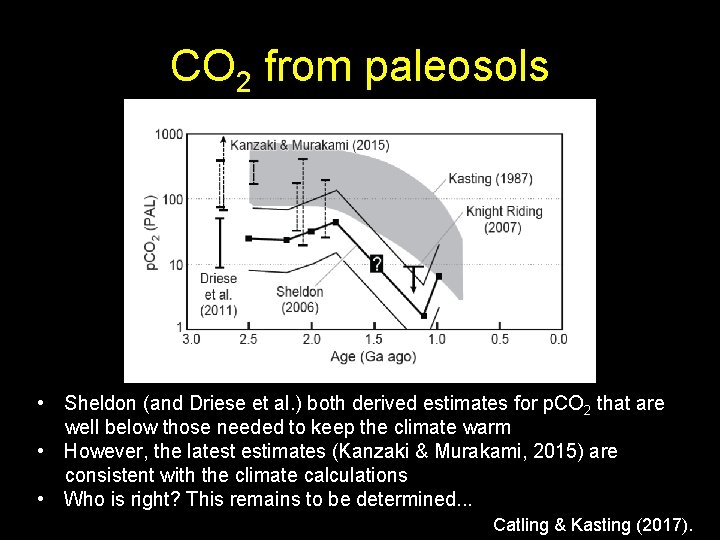
CO 2 from paleosols • Sheldon (and Driese et al. ) both derived estimates for p. CO 2 that are well below those needed to keep the climate warm • However, the latest estimates (Kanzaki & Murakami, 2015) are consistent with the climate calculations • Who is right? This remains to be determined. . . Catling & Kasting (2017).

• What about other greenhouse gases? Sagan and Mullen like ammonia (NH 3) and methane (CH 4) • Both of these should have been more abundant in a low-O 2 early atmosphere
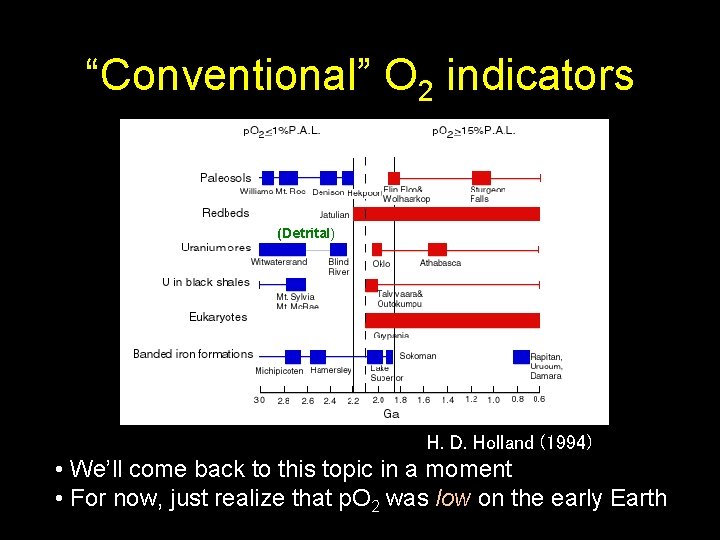
“Conventional” O 2 indicators (Detrital) H. D. Holland (1994) • We’ll come back to this topic in a moment • For now, just realize that p. O 2 was low on the early Earth

Problems with Sagan and Mullen’s hypothesis • Ammonia is photochemically unstable with respect to conversion to N 2 and H 2 (Kuhn and Atreya, 1979)

• Thus, ammonia was probably not abundant in the early atmosphere – That said, there are some complications with shielding by organic haze that we can come back to

• But, once life had originated, hydrogen would likely have been consumed on the early Earth by organisms that turned it into methane • Today, CH 4 is produced in restricted, anaerobic environments, such as the intestines of cows and the water-logged soils underlying rice paddies • Methanogenic bacteria are responsible for virtually all methane production • One common metabolic pathway forming methane is: CO 2 + 4 H 2 CH 4 + 2 H 2 O

• Furthermore, methanogens are thought to be evolutionarily ancient, based on sequencing of ribosomal RNA

Ribosomal RNA • Ribosomes are organelles (inclusions) within cells in which proteins are made • Surprisingly, ribosomes contain their own RNA (ribonucleic acid) – The RNA is also the catalyst for protein synthesis, indicating that life may have passed through an “RNA World” stage • The RNA in ribosomes evolves very slowly, so looking at differences in the RNA of different organisms allows biologists to look far back into evolution http: //www. scilogs. eu/en/blog/lindaunobel/ 2010 -06 -29/mountains-beyond-mountains
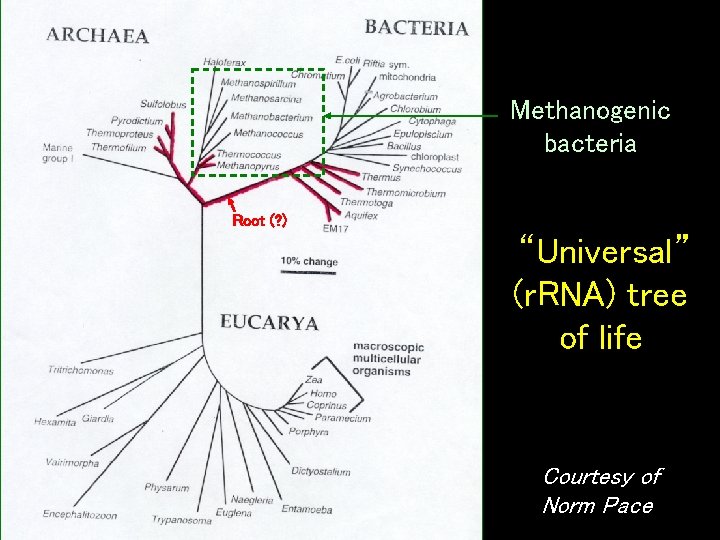
Methanogenic bacteria Root (? ) “Universal” (r. RNA) tree of life Courtesy of Norm Pace

Furthermore… • Photochemical lifetime of CH 4 is relatively short (~10 years) today, because it reacts with the hydroxyl radical, OH: O 3 + h O 2 + O(1 D) + H 2 O 2 OH OH + CH 4 CH 3 + H 2 O ( < 310 nm) • But, its lifetime in a low-O 2 atmosphere would be ~1000 times longer
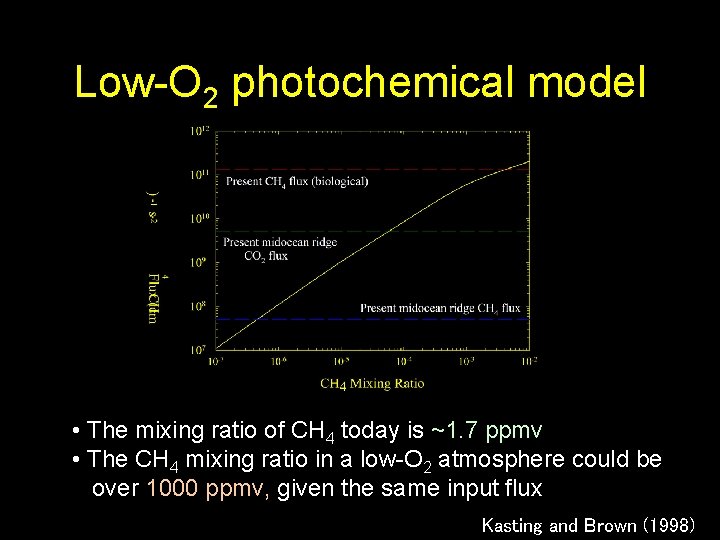
Low-O 2 photochemical model • The mixing ratio of CH 4 today is ~1. 7 ppmv • The CH 4 mixing ratio in a low-O 2 atmosphere could be over 1000 ppmv, given the same input flux Kasting and Brown (1998)

Anoxic ecosystem model P. Kharecha et al. , Astrobiol. (2005) • Methane flux from a hydrogenbased ecosystem limited by transfer of H 2 between the atmosphere and ocean • Prediction: Even a totally anoxic ecosystem should have a CH 4 flux comparable to today

• But, there is a limit to how much warming one can get out of CH 4 because if the CH 4/CO 2 ratio gets too high (<0. 1), then methane begins to polymerize to form haze, and this haze cools the planet’s surface

Titan’s organic haze layer • Haze is thought to form from photolysis (and charged particle irradiation) of CH 4 • It can produce an anti-greenhouse effect (Picture from Voyager 2)

• Greenhouse effect – Incoming visible/near-IR radiation mostly makes its way through the atmosphere – Outgoing thermal-IR radiation is absorbed and reradiated – Net effect is to warm the surface • Anti-greenhouse effect – Incoming visible/near-IR radiation is absorbed and re-radiated high in the atmosphere – Net effect is to cool the surface
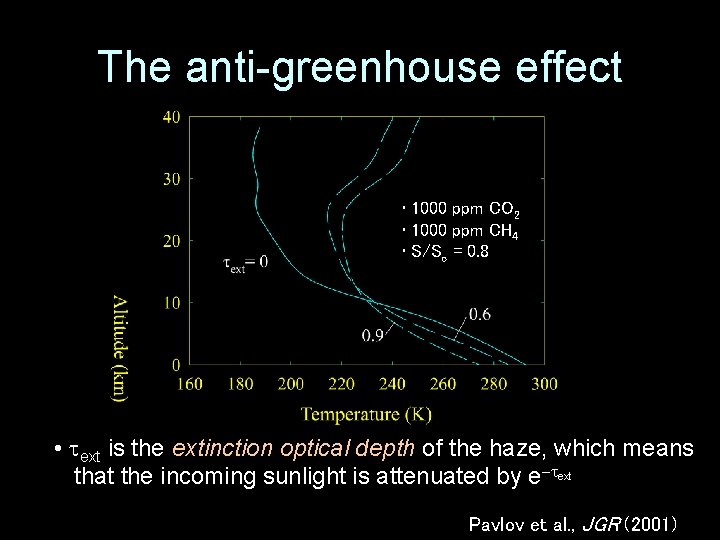
The anti-greenhouse effect • 1000 ppm CO 2 • 1000 ppm CH 4 • S/So = 0. 8 • ext is the extinction optical depth of the haze, which means that the incoming sunlight is attenuated by e ext Pavlov et al. , JGR (2001)
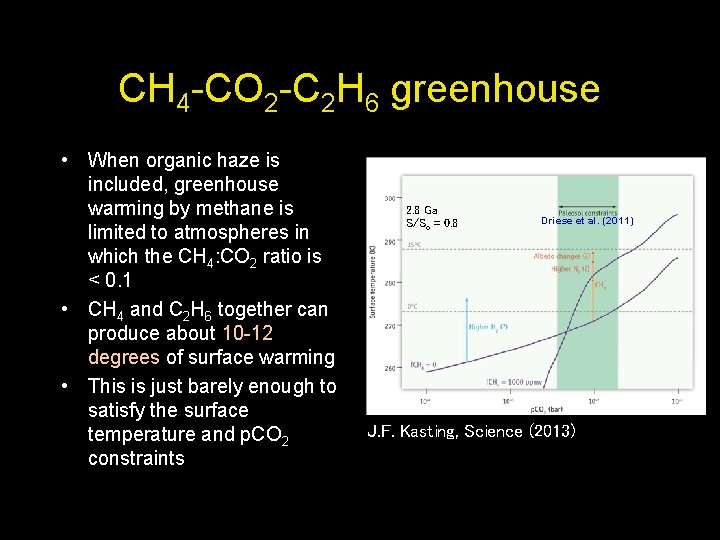
CH 4 -CO 2 -C 2 H 6 greenhouse • When organic haze is included, greenhouse warming by methane is limited to atmospheres in which the CH 4: CO 2 ratio is < 0. 1 • CH 4 and C 2 H 6 together can produce about 10 -12 degrees of surface warming • This is just barely enough to satisfy the surface temperature and p. CO 2 constraints 2. 8 Ga S/So = 0. 8 Driese et al. (2011) J. F. Kasting, Science (2013)

Back to the main story… • When atmospheric O 2 levels rise, most of the CH 4 and C 2 H 6 was lost, and the greenhouse effect decreased, triggering glaciation
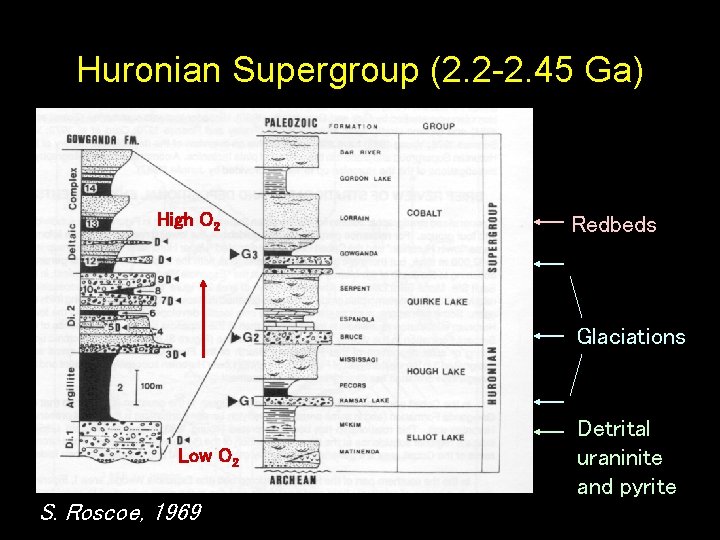
Huronian Supergroup (2. 2 -2. 45 Ga) High O 2 Redbeds Glaciations Low O 2 S. Roscoe, 1969 Detrital uraninite and pyrite

Conclusions • Higher atmospheric CO 2 was probably a major factor in solving the faint young Sun problem – This was a natural response caused by negative feedback in the carbonate-silicate cycle • Methane and ethane may have contributed to the greenhouse effect back when atmospheric O 2 levels were low • High atmospheric CH 4/CO 2 ratios can trigger the formation of organic haze. This has a cooling effect. • The Paleoproterozoic glaciation at ~2. 4 Ga may have been triggered by the rise of O 2 and loss of the methane component of the atmospheric greenhouse
- Slides: 46