The Equilibrium Constant Kc Lesson Outline The Equilibrium

The Equilibrium Constant (Kc)

Lesson Outline The Equilibrium Constant (Kc) Kc Expression (The Equilibrium Law) Kc Expression Examples 1 -4
![Equilibrium Constant (Kc) Kc is a ratio between the [products] and the [reactants] at Equilibrium Constant (Kc) Kc is a ratio between the [products] and the [reactants] at](http://slidetodoc.com/presentation_image_h2/01ba1a844822171bd2af55cab814999e/image-3.jpg)
Equilibrium Constant (Kc) Kc is a ratio between the [products] and the [reactants] at equilibrium at a specific temperature. Kc does not have Si Units It is a numerical constant. It is the same number, regardless of the [initial]s. If you change the temperature, the Kc also changes.

Equilibrium Constant (Kc) The higher the value of Kc represents a greater tendency to favor the forward reaction. Kc>1, the Eq system favors products Kc<1, the Eq system favors reactants Kc=1, neither products or reactants are favored Equilibrium contains 50% products and 50% reactants

Kc Expression (Equilibrium Law) For the general forward reaction: a. A + b. B �c. C + d. D Kc – equilibrium constant [C] and [D] – [equilibrium] of products [A] and [B] – [equilibrium] of reactants (a, b, c, d) – coefficients of the balanced equation The reverse reaction, the Kc is the reciprocal. (1/Kc)

General Rules for Kc 1. Solids are not included in the Kc expression 2. If liquids are to be included, all species must be liquid 3. Gases and Aqueous states are included. 4. Kc is not affected by catalysts or reaction rates, only temperature

Example #1 White board Activity 2 SO 2(g) + 1 O 2(g) � 2 SO 3(g) At equilibrium [SO 2(g)] = 4. 50 mol/L [O 2(g)] = 6. 25 mol/L [SO 3(g)] = 6. 00 mol/L Determine the Kc for the reaction Write the balanced equation. Write the Kc expression. Substitute in the [ ]’s for all reactants and products

Forward and Reserve Reaction Kc White board Activity Write the Kc Expression for the following reaction Ca. CO 3(s) �Ca. O(s) + CO 2(g) Forward Reserve
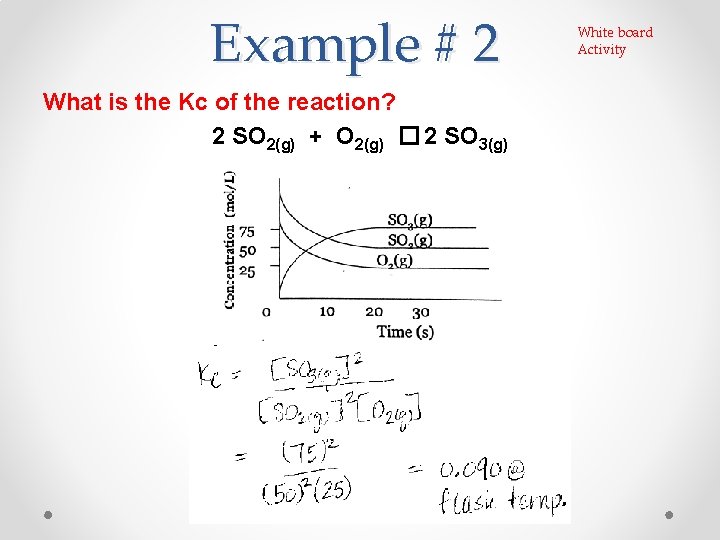
Example # 2 What is the Kc of the reaction? 2 SO 2(g) + O 2(g) � 2 SO 3(g) White board Activity

Example # 3 White board Activity Initially, 3. 5 mol of ammonia gas are placed into a 5. 0 L flask and sealed. At equilibrium, there are 0. 20 mol of hydrogen present. Find the Kc. Are reactants or products favored? 2 NH 3(g) � N 2(g) + 3 H 2(g)

Example # 4 White board Activity Initially, 2. 0 mol of each CO 2(g) and H 2(g) are introduced into a 1. 0 L container. What would be the equilibrium concentrations of the products when the Kc = 35 @ 1823 C? 1 CO 2(g) + 1 H 2(g) � 1 H 2 O(g) + 1 CO(g) + energy

Example # 4

Looking Forward Today E 5 The Equilibrium Constant (Kc) Tomorrow More Kc Expression Problems
- Slides: 13