THE ENERGY OF LIFE Chapter 6 Bioenergetics study

THE ENERGY OF LIFE Chapter 6

Bioenergetics- study of how living organisms use energy to perform the activities of life.

A. Energy The ability to do work (change or move matter against an opposing force). Measured in calories (the amount of energy needed to raise the temperature of 1 gram of water by 1°C). • A kilocalorie is 1000 calories

Energy made available for cellular functions

1. Sources of energy Ultimately, all life on earth is dependent on energy from: • the sun (solar energy) • inorganic chemicals (geothermal energy)

Photoautotrophs - organisms that use the energy in sunlight to manufacture nutrient molecules. Ex. plants, algae, cyanobacteria Chemoautotrophs - organisms that use the energy in inorganic chemicals to manufacture nutrient molecules. Ex. certain bacteria

2. Basic Forms of Energy Potential energy - stored energy available to do work. Kinetic energy - energy being used to do work.

Energy- capacity to do work- vital to life Comes from the sun Potential energy is stored in matter and in chemical bonds Kinetic energy- energy of motion or under use Organisms – extract energy from the sun or from molecules.

B. Laws of Thermodynamics Regulate energy conversions. 1. First Law of Thermodynamics (aka. Law of energy conservation) Energy can be converted from one form into another, but it CANNOT be created or destroyed. 2. Second Law of Thermodynamics Energy conversions are inefficient. Results in an increase in ENTROPY (disorder).

• Energy- can not be created or destroyed but can change form. • The physical laws explain energy transformation. With each transfer some energy becomes unusable as heat. • Cells extract energy from chemical bonds in controlled steps to capture as much as possible in a usuable form.

Coupling reactions • A reaction involving energy always happens at the expense of another reaction. • Organisms can increase in complexity with the contingency that something else must decrease in complexity.

C. Biochemical Reactions 1. Metabolism Chemical reactions that change or transform energy in cells.
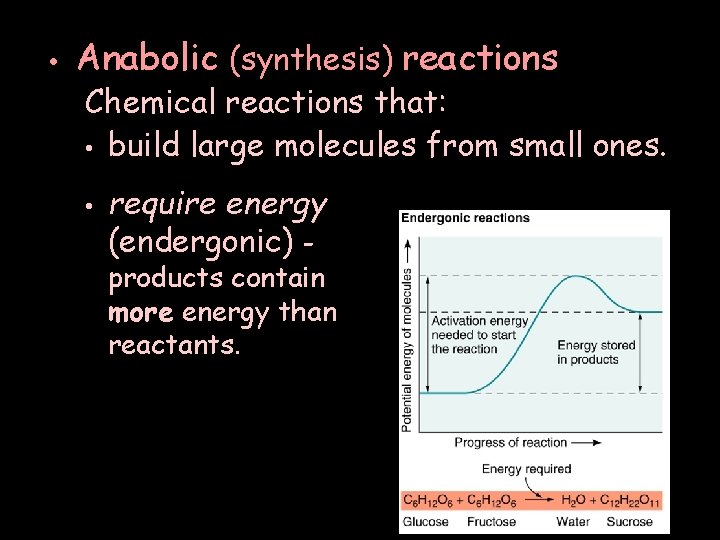
• Anabolic (synthesis) reactions Chemical reactions that: • build large molecules from small ones. • require energy (endergonic) - products contain more energy than reactants.
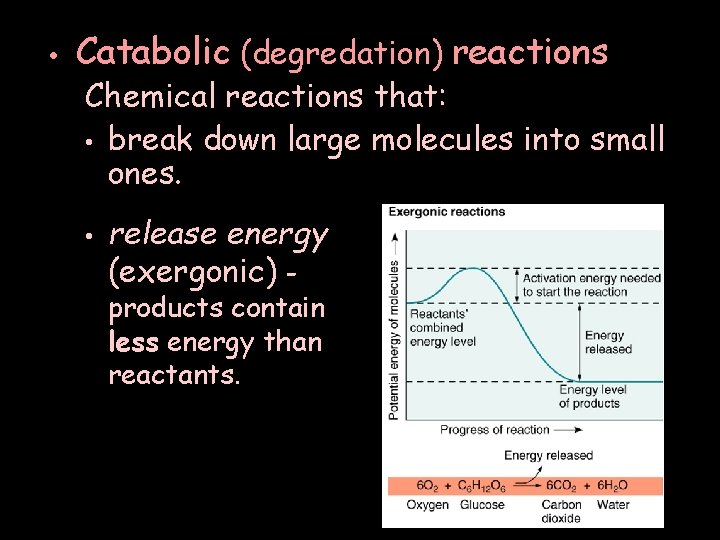
• Catabolic (degredation) reactions Chemical reactions that: • break down large molecules into small ones. • release energy (exergonic) products contain less energy than reactants.

Chemical equilibrium • Reactions that can proceed in two directions eventually reach equilibrium where the reaction is going in both directions at the same rate. • Energy is no longer being gained or lost once the reaction has reached equilibrium.

2. Oxidation - Reduction Reactions Most energy transformations in organisms involve oxidation & reduction. Oxidation - a molecule loses one or more electrons. Reduction - a molecule gains one or more electrons. Oxidations tend to be linked to reductions. Ex. electron transport chain
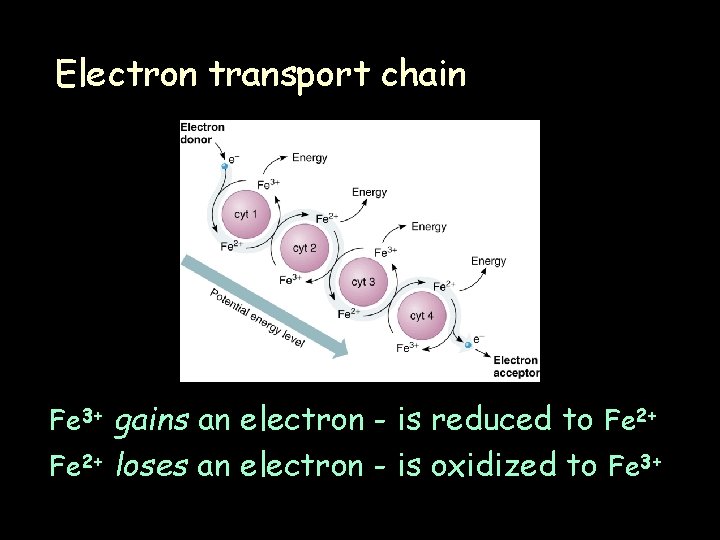
Electron transport chain Fe 3+ gains an electron - is reduced to Fe 2+ loses an electron - is oxidized to Fe 3+

v. Energy reactions in cells are either anabolic, building molecules or catabolicdegrading them. v. Endergonic reactions require energy and exergonic reactions release it. v. All energy in cells is passed with electrons from molecule to molecule.

Molecules involved in energy transformation
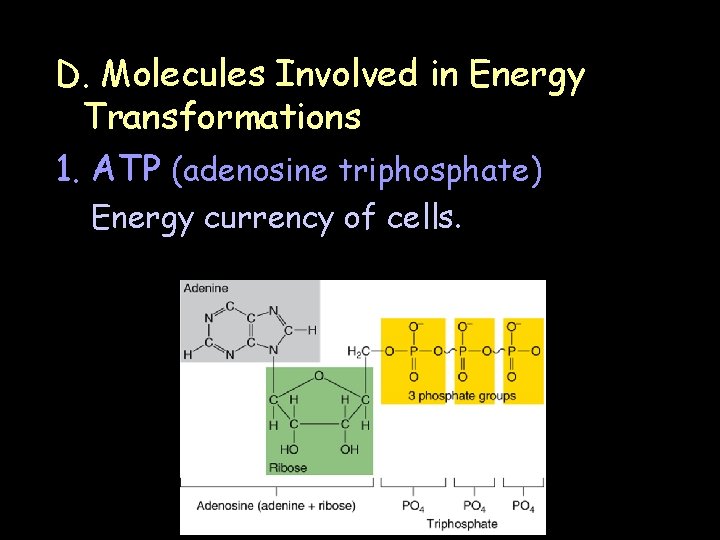
D. Molecules Involved in Energy Transformations 1. ATP (adenosine triphosphate) Energy currency of cells.
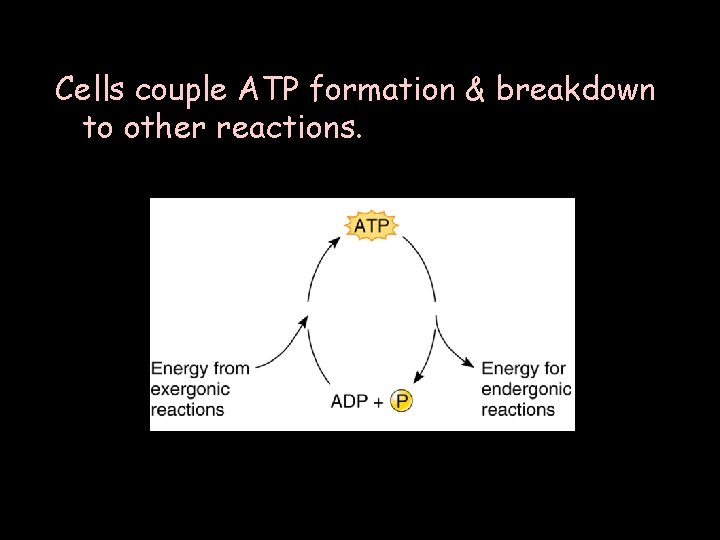
Cells couple ATP formation & breakdown to other reactions.

2. Cofactors Inorganic helpers (usually ions) needed by some enzymes to function. Ex. Mg 2+ 3. Coenzymes Organic cofactors (vitamin derived) needed by some enzymes to function. • Nicotinamide adenine dinucleotide (NAD+) - coenzyme that transfers electrons; derived from niacin.

• • Nicotinamide adenine dinucleotide phosphate (NADP+) - coenzyme that transfers electrons. Flavin adenine dinucleotide (FAD) coenzyme that transfers electrons; derived from riboflavin. 4. Cytochromes Iron containing molecules that transport electrons. Ex. electron transport chains

ØATP, through high-energy phosphate bonds, temporarily stores energy that a cell uses for a wide variety of activities. ØBreaking the bonds through transient phosphate transfers releases energy for cell’s reactions. ØSome of energy is lost as heat. ØATP is constantly recycled. ØOther molecules assist the cell as carriers of energetic electrons.
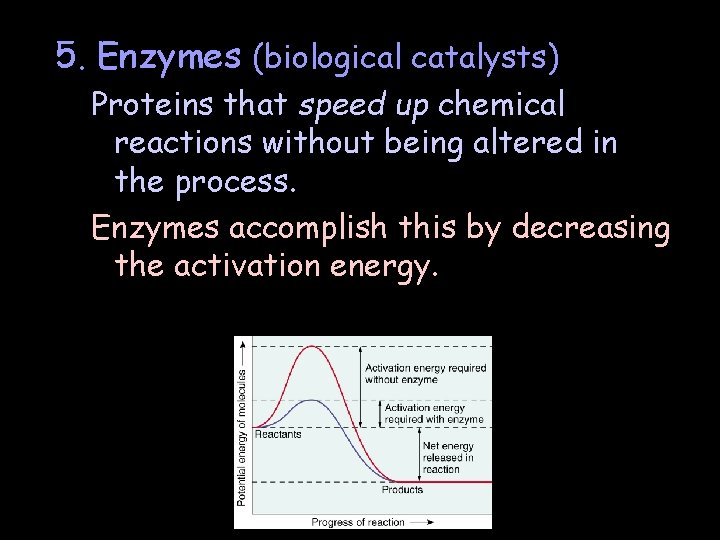
5. Enzymes (biological catalysts) Proteins that speed up chemical reactions without being altered in the process. Enzymes accomplish this by decreasing the activation energy.
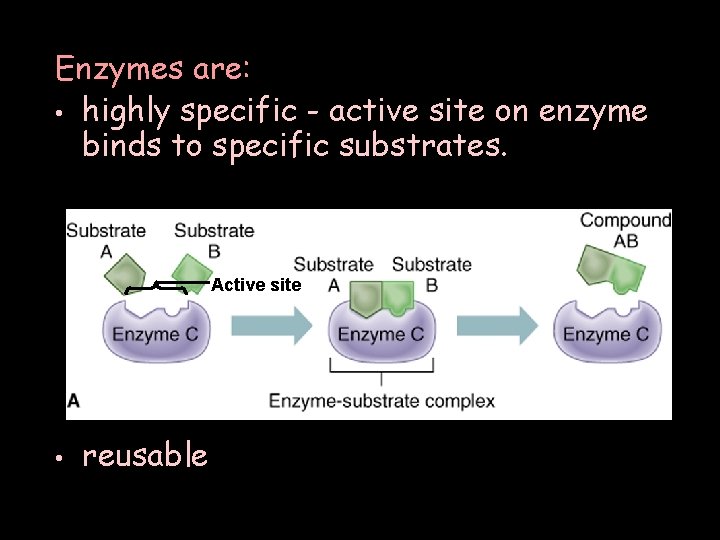
Enzymes are: • highly specific - active site on enzyme binds to specific substrates. Active site • reusable
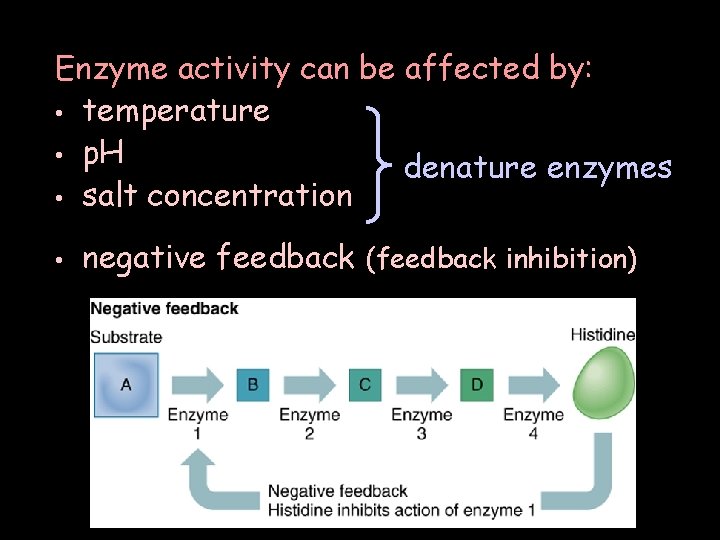
Enzyme activity can be affected by: • temperature • p. H denature enzymes • salt concentration • negative feedback (feedback inhibition)
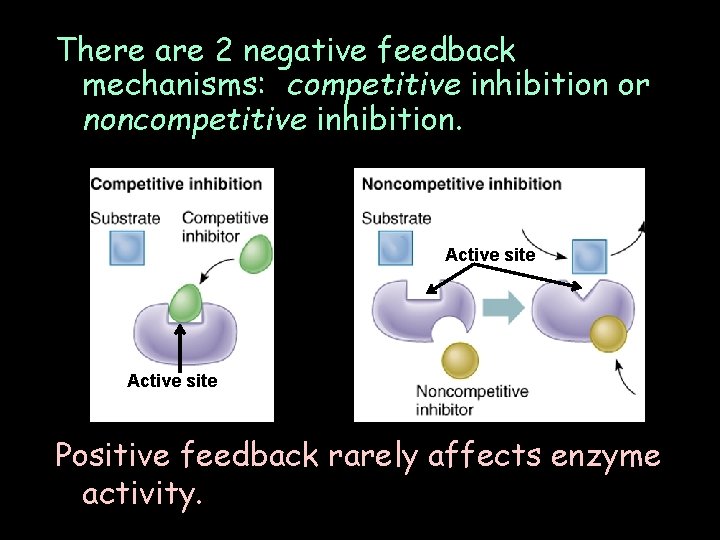
There are 2 negative feedback mechanisms: competitive inhibition or noncompetitive inhibition. Active site Positive feedback rarely affects enzyme activity.

Enzymes allow the biochemical reactions to proceed faster. Enzymes lower the energy required to start biochemical reactions. Cells use several mechanisms to switch enzymes off to control chemical processes. Enzymes are very sensitive to regulation. Feedback inhibition prevents the cells from overproduction.
- Slides: 29