The ENABLERP A Novel CTO Crossing System Maurice

The ENABLER-P A Novel CTO Crossing System Maurice Buchbinder, MD Foundation for Cardiovascular Medicine La Jolla, CA

DISCLOSURES Maurice Buchbinder, MD Honoraria – Boston Scientific Corporation, Cordis, a Johnson & Johnson company, Abbott Vascular

Current Limitations of CTO Treatments Despite the advent of new devices / guide wires as well as improved operator technique, success rates remain limited (60 -80%) Unsuccessful attempts are primarily due to inability to cross lesions with a guidewire Typically CTO procedures are associated with extended procedural time, radiation exposure and increased contrast load. Treatment of CTO lesions remains highly dependent on operator skill and experience.

ENABLER-P System Includes 2 components: Support balloon catheter: Catheter lumen 0. 35 compatible Catheter length 135 cm Balloon length 20 mm Balloon Diameter 6 mm

ENABLER-P System Pressure Control Unit (PCU) : Disposable, battery operated unit Allows additional cyclical balloon inflations and deflations
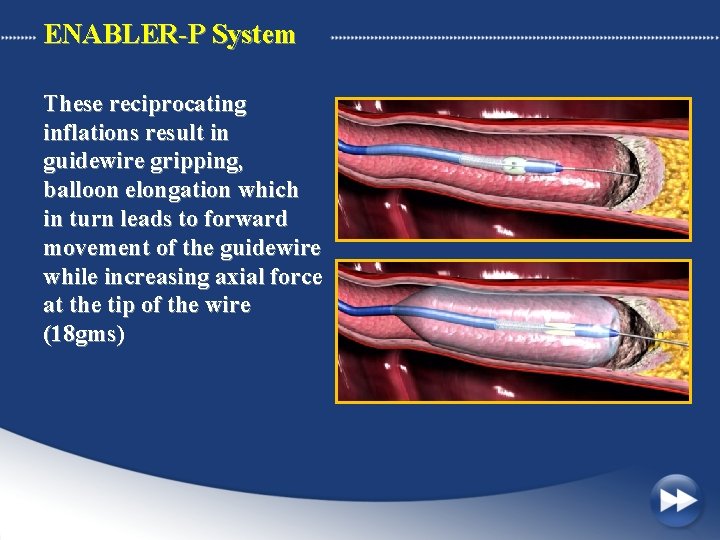
ENABLER-P System These reciprocating inflations result in guidewire gripping, balloon elongation which in turn leads to forward movement of the guidewire while increasing axial force at the tip of the wire (18 gms)
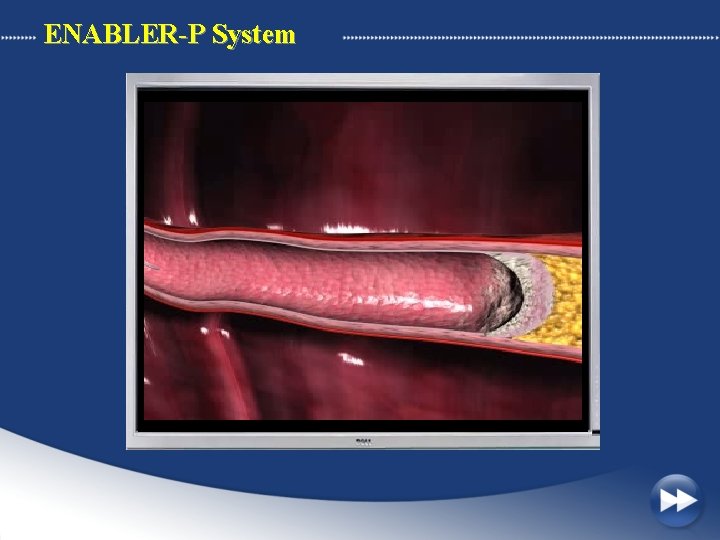
ENABLER-P System

First Human Use Experience : Apollo I Designed to evaluate the Safety and Effectiveness of the ENABLER-P System in crossing Chronic Total Occlusions located in the Superficial Femoral and popliteal Artery
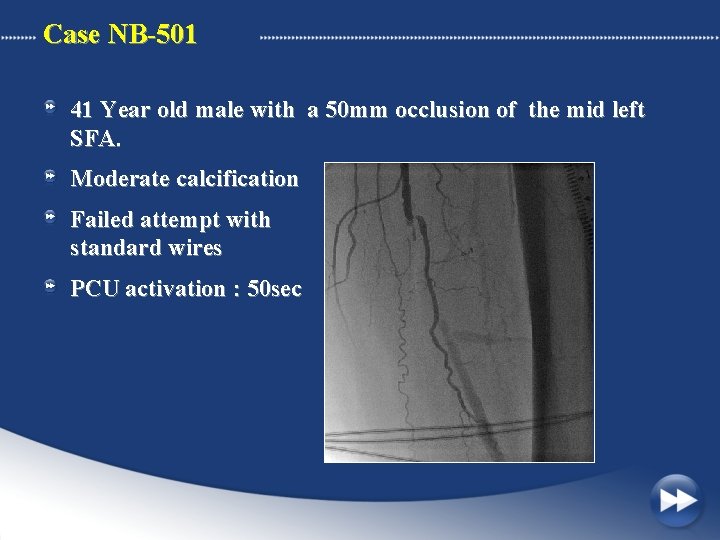
Case NB-501 41 Year old male with a 50 mm occlusion of the mid left SFA. Moderate calcification Failed attempt with standard wires PCU activation : 50 sec
Case NB-501
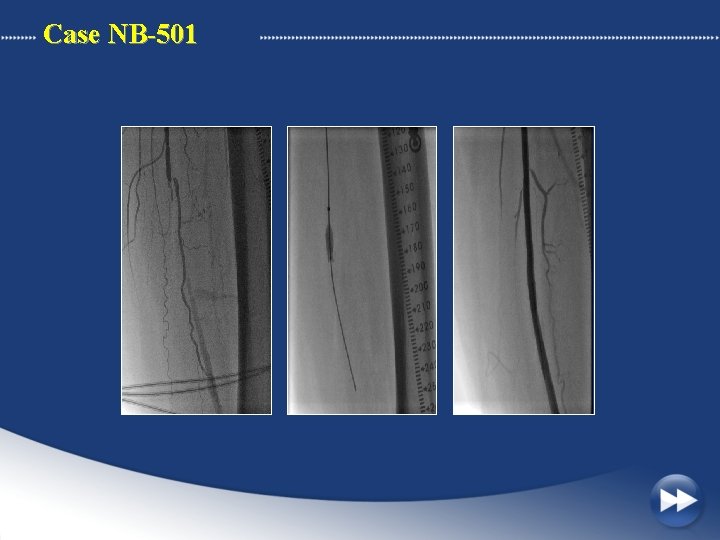
Case NB-501
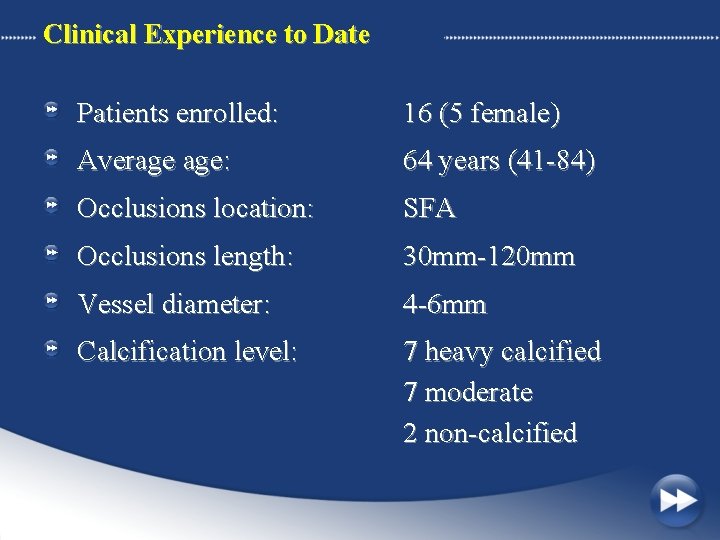
Clinical Experience to Date Patients enrolled: 16 (5 female) Average age: 64 years (41 -84) Occlusions location: SFA Occlusions length: 30 mm-120 mm Vessel diameter: 4 -6 mm Calcification level: 7 heavy calcified 7 moderate 2 non-calcified

Clinical Results n=16 Successful crossing 15 Serious adverse events 0 Device related adverse events 0 Procedure related adverse events 2* Successful revascularization 14 Average system operation time 2 min. * One Proximal dissection from initial vessel access resulting in guidewire placement into false lumen. One vessel perforation -performed with a dilatation balloon after the removal of the ENABLER-P Catheter.
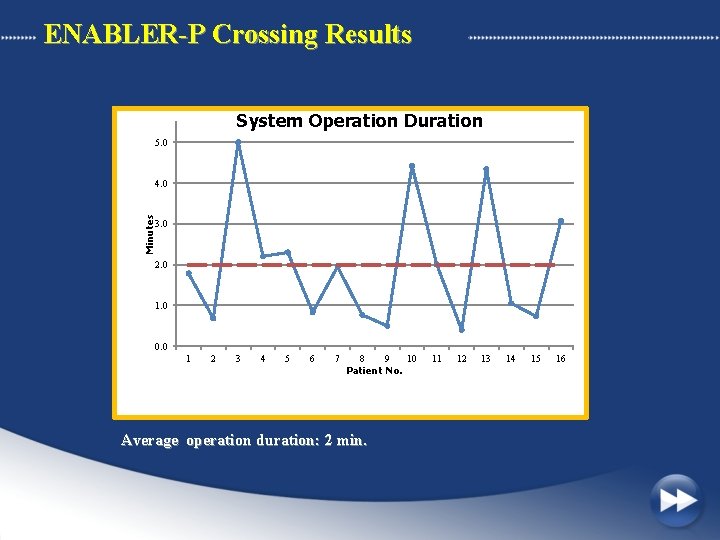
ENABLER-P Crossing Results System Operation Duration 5. 0 Minutes 4. 0 3. 0 2. 0 1. 0 0. 0 1 2 3 4 5 6 7 8 9 10 Patient No. Average operation duration: 2 min. 11 12 13 14 15 16

Conclusions ENABLER-P System is a promising novel device for treatment of Chronic Total Occlusions. In this early experience the Enabler appears to be safe and effective in crossing calcified lesions in the peripheral circulation. Ongoing evaluation continues to be extremely favorable. FDA Approved as of Jan 1, 2010
- Slides: 15