The electronic structure of biphenylene Cesare Grazioli Self

The electronic structure of biphenylene Cesare Grazioli

Self assembly – self organization Advantages parallel production, molecular dimension control, defect tolerance, self-repair, and increased life-time. The modularity offers synthetic simplicity and rapid adaption to specific needs. Judicious design of the building blocks and interactions. from readily accessible components, tailoring the final material architecture material science is still in progress of borrowing from Nature the control of hierarchical architectures. “DELICATE CONTROL” based on weak interactions, • hydrogen bonding, • metal ion coordination, • van der Waals interactions • reversible covalent bonds (esters, disulfides, cycloadditions etc)

Biphenylene (C 12 H 8) cyclic hydrocarbon formed by two benzene rings and a cyclobutadiene ring. stable synthesizable molecule 4 nπ-electrons - expected antiaromaticity (Hückel rule) pronounced aromatic character, which make it a very interesting molecule from a fundamental physics point of view. We performed synchrotron radiation experiments Precursor of a 2 D porous molecular • Results from GAS graphite-like PHASE + DFT calculations, could distinguish networkcharacteristic called biphenyl carbon (BPC) contributions of chemically non-equivalent carbon and correctly assign them. A. T. Balaban, et al. Rev. Roum. Chim. 13 (1968) • Characterization of FILMS on Cu(111) + DFT calculations
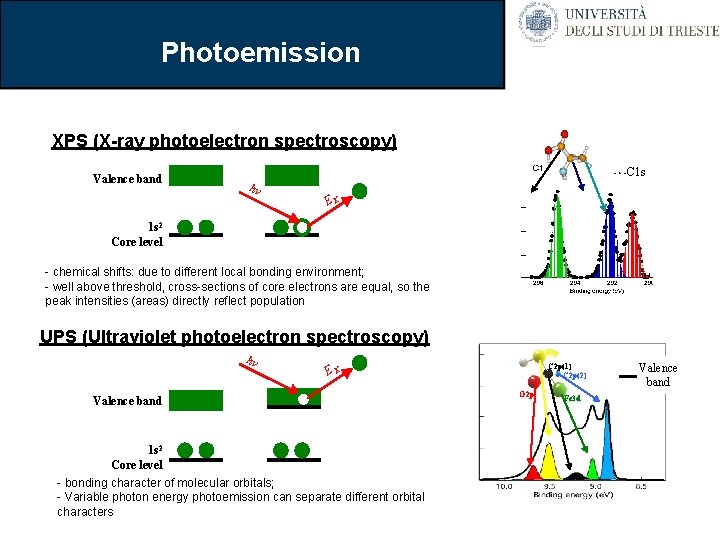
Photoemission XPS (X-ray photoelectron spectroscopy) Valence band C 1 s hv EK 1 s 2 Core level - chemical shifts: due to different local bonding environment; - well above threshold, cross-sections of core electrons are equal, so the peak intensities (areas) directly reflect population UPS (Ultraviolet photoelectron spectroscopy) hv N 2 p C 2 p(1) C 2 p(2) EK Valence band 1 s 2 Core level - bonding character of molecular orbitals; - Variable photon energy photoemission can separate different orbital characters O 2 p Fe 3 d Valence band
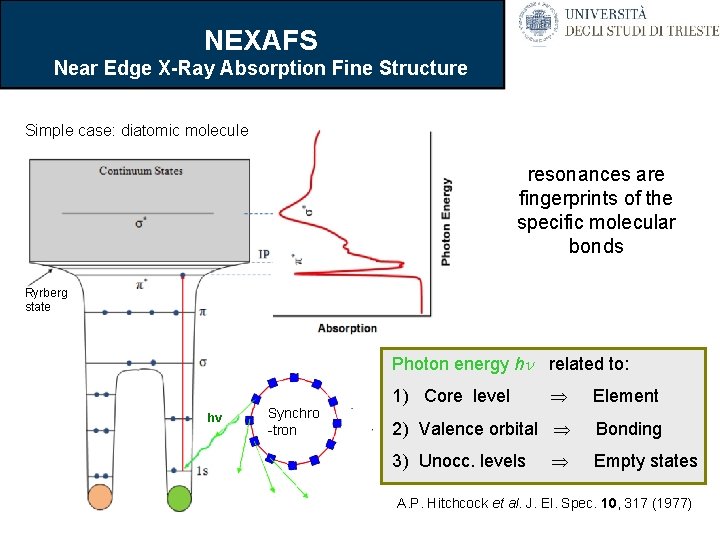
NEXAFS Near Edge X-Ray Absorption Fine Structure Simple case: diatomic molecule resonances are fingerprints of the specific molecular bonds Ryrberg state Photon energy h related to: hv Synchro -tron Element 2) Valence orbital Bonding 1) Core level 3) Unocc. levels Empty states A. P. Hitchcock et al. J. El. Spec. 10, 317 (1977)

Gas phase: C 1 s XPS hv=332 e. V Rather narrow XPS peak. Asimmetric tail due to vibrations Asymmetric skew gaussians at the calculated binding energies. Gaussian width = 70 me. V (resolution) Skew factor = fit C 1 s The contributions by the different carbons Cα Cβ Cγ can be separated Binding energy (e. V) J. Lüder, M. de Simone, R. Totani, M. Coreno, C. Grazioli, B. Sanyal, O. Eriksson, B. Brena, C. Puglia, J. of Chemical Physics, 142, 2015
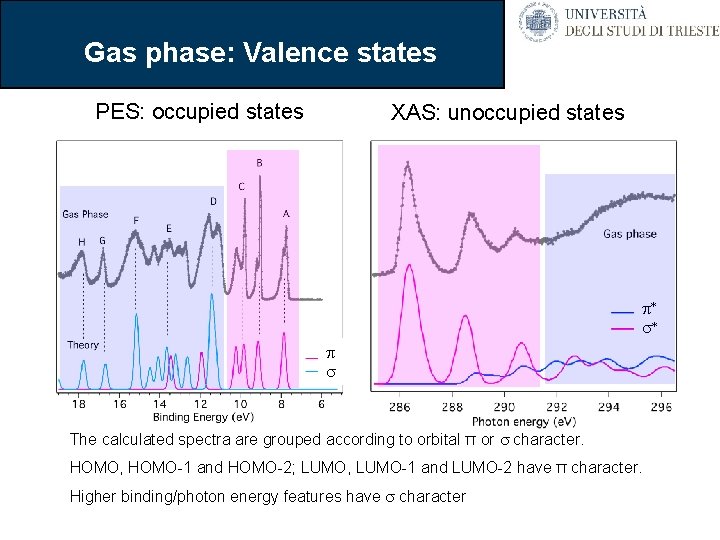
Gas phase: Valence states PES: occupied states XAS: unoccupied states p* s* p s The calculated spectra are grouped according to orbital π or s character. HOMO, HOMO-1 and HOMO-2; LUMO, LUMO-1 and LUMO-2 have π character. Higher binding/photon energy features have s character
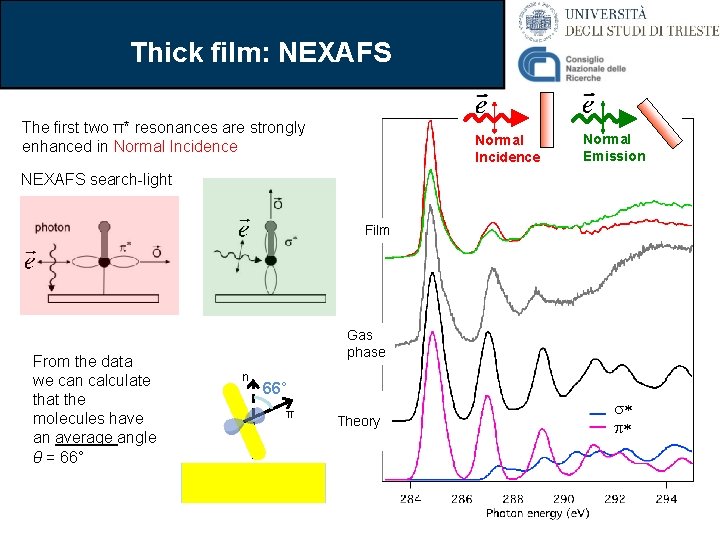
Thick film: NEXAFS The first two π* resonances are strongly enhanced in Normal Incidence Normal Emission NEXAFS search-light Film From the data we can calculate that the molecules have an average angle θ = 66° Gas phase n 66° π Theory s* p*
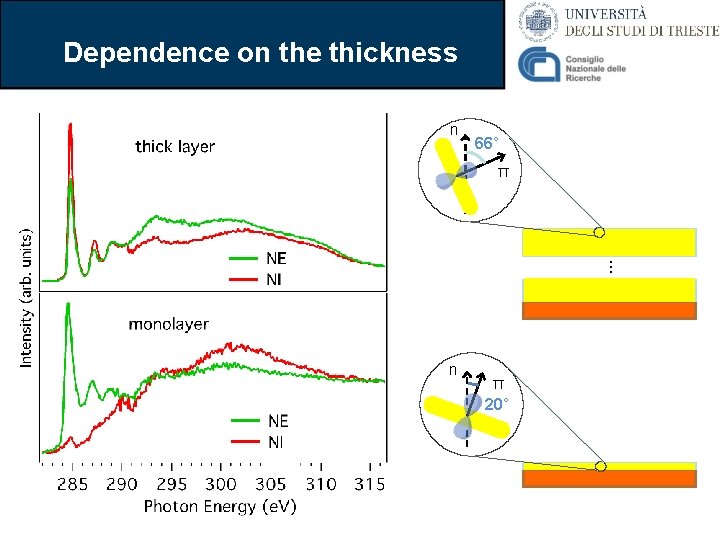
Dependence on the thickness n 66° π . . . n π 20°
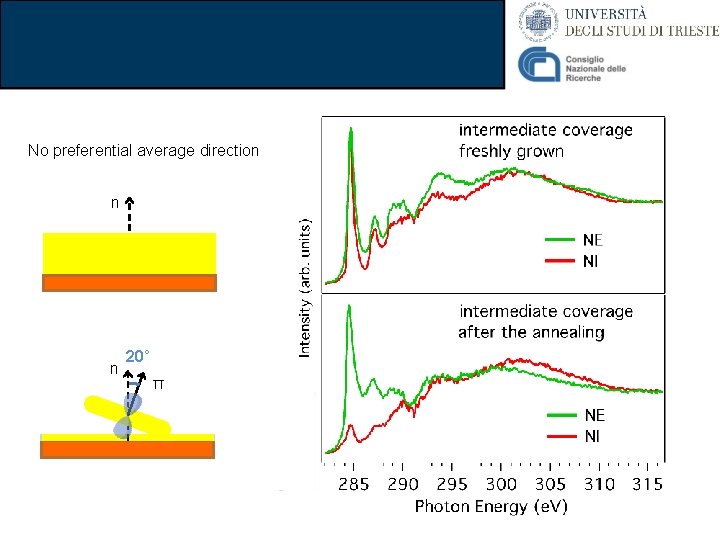
No preferential average direction n n 20° π

Conclusions on biphenylene ü Isolated biphenylene molecule has been characterized in detail • (Photoemission, NEXAFS, DFT) ü The growth on Cu(111) has been characterized • They preserves a molecular character • General broadening of the spectrum ü The molecules are oriented depending on the film thickness • The mechanism is reversible
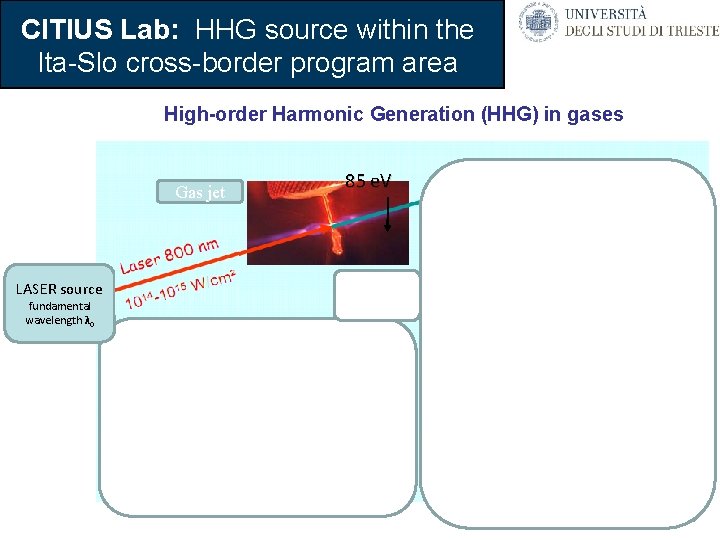
CITIUS Lab: HHG source within the Ita-Slo cross-border program area High-order Harmonic Generation (HHG) in gases Gas jet Grating 85 e. V LASER source fundamental wavelength λ 0 • high-order harmonics • very short optical pulses: tens of fs, good coherence • significant energy per pulse Ultrafast spectroscopy and femtochemisty Number of photons Photons/pulse 7 10 « plateau » HHG in neon 31 15 « cutoff » 6 10 65 5 10 4 10 50 40 30 20 Harmonic order n Wavelength (nm) LASER output harmonics λ 0/n wavelengths λ 0/nof the fundamental (Saclay & Chicago, 1988)
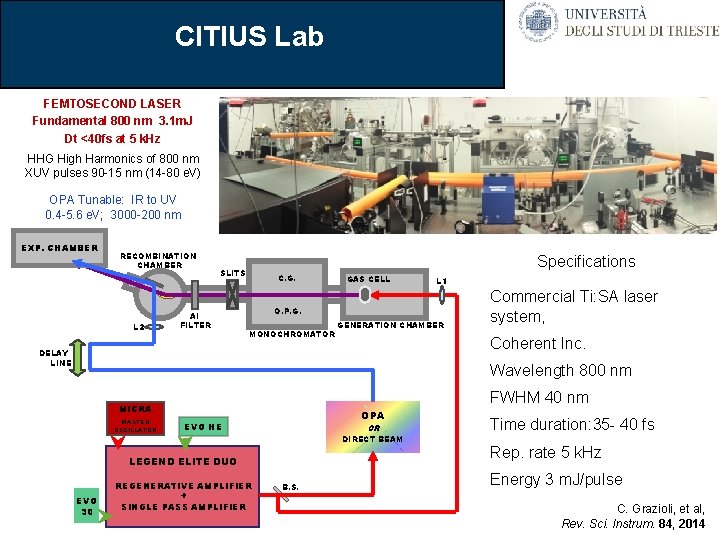
CITIUS Lab FEMTOSECOND LASER Fundamental 800 nm 3. 1 m. J Dt <40 fs at 5 k. Hz HHG High Harmonics of 800 nm XUV pulses 90 -15 nm (14 -80 e. V) OPA Tunable: IR to UV 0. 4 -5. 6 e. V; 3000 -200 nm EXP. CHAMBER RECOMBINATION CHAMBER L 2 Specifications SLITS Al FILTER C. G. GAS CELL L 1 O. P. G. MONOCHROMATOR GENERATION CHAMBER Commercial Ti: SA laser system, Coherent Inc. DELAY LINE Wavelength 800 nm FWHM 40 nm MICRA MASTER OSCILLATOR OPA EVO HE OR DIRECT BEAM LEGEND ELITE DUO EVO 30 REGENERATIVE AMPLIFIER + SINGLE PASS AMPLIFIER B. S. Time duration: 35 - 40 fs Rep. rate 5 k. Hz Energy 3 m. J/pulse C. Grazioli, et al, Rev. Sci. Instrum. 84, 2014
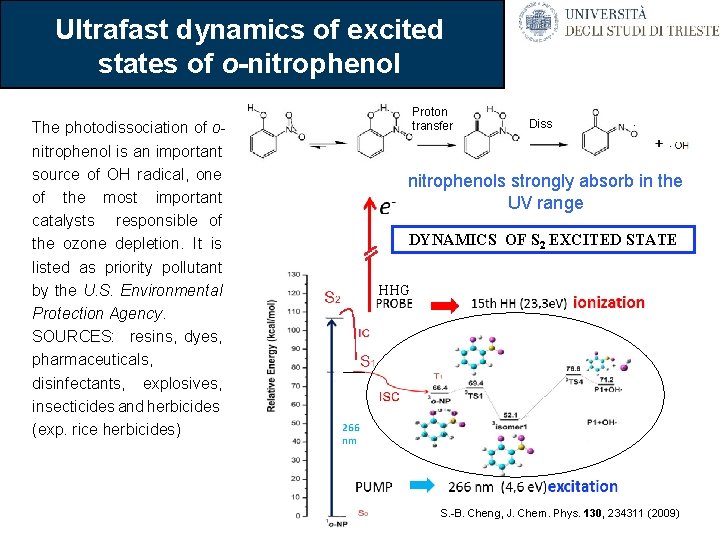
Ultrafast dynamics of excited states of o-nitrophenol The photodissociation of onitrophenol is an important source of OH radical, one of the most important catalysts responsible of the ozone depletion. It is listed as priority pollutant by the U. S. Environmental Protection Agency. SOURCES: resins, dyes, pharmaceuticals, disinfectants, explosives, insecticides and herbicides (exp. rice herbicides) Proton transfer Diss nitrophenols strongly absorb in the UV range DYNAMICS OF S 2 EXCITED STATE HHG S. -B. Cheng, J. Chem. Phys. 130, 234311 (2009)
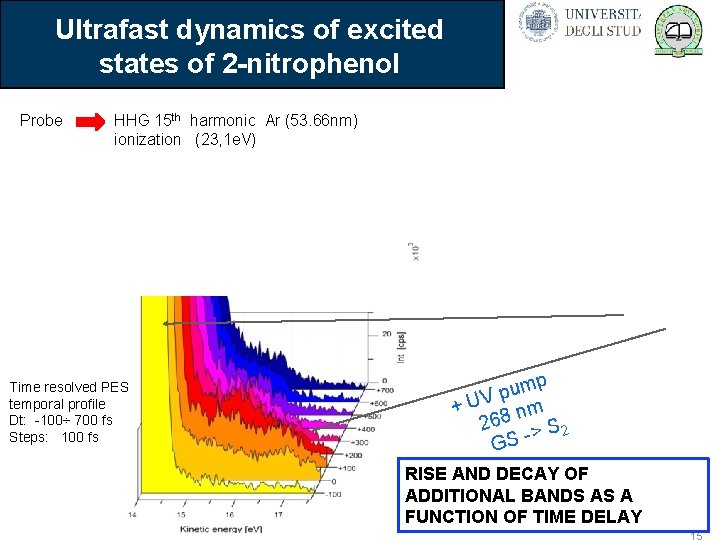
Ultrafast dynamics of excited states of 2 -nitrophenol Probe HHG 15 th harmonic Ar (53. 66 nm) ionization (23, 1 e. V) Pump OPA (368 nm) / Ti: Sa 3 rd (268 nm) excitation (3, 4 e. V or 4, 6 e. V) Time resolved PES temporal profile Dt: -100÷ 700 fs Steps: 100 fs Valence PES of 2 -np Static PES mp u p + UV 8 nm 26 -> S 2 S G RISE AND DECAY OF ADDITIONAL BANDS AS A FUNCTION OF TIME DELAY 15

Thank you! BIPHENYLENE M. de Simone, M. Coreno R. Totani L. Lozzi Uppsala University of L’Aquila T. Zhang, V. Lanzillotto, I. Bidermane, H. Ottosson, J. Lu der, B. Brena, C. Puglia Uppsala University Trieste L’Aquila Lead G. De Ninno Partner B. Ressel A. Ciavardini PP 1 M. Coreno, M. de Simone, A. Kivimäki A. L. Poletto P. Miotti F. Frassetto D. Gauthier Univerza v Novi Gorici Elettra Sincrotrone (Ts) R. Ivanov PP 2 Univerza v Novi Gorici PP 3 o-nitrophenol CITIUS Lab CNR - IFN, Luxor (Pd) CNR - ISOF (Fe) M. Pezzella, PP 5 PP 6 S. Fornarini Kontrolni Sistemi (Sežana) Univerza v Ljubljani C. Spezzani E. Bodo S. Piccirillo funding from FP 7: Italo-Slovenian cross border University La program) Sapienza and Tor Vergata cooperation Elettra Sincrotrone
- Slides: 16