THE EFFECTS OF PARKINSONIAN POSTURE AND OBSTACLE CLEARANCE
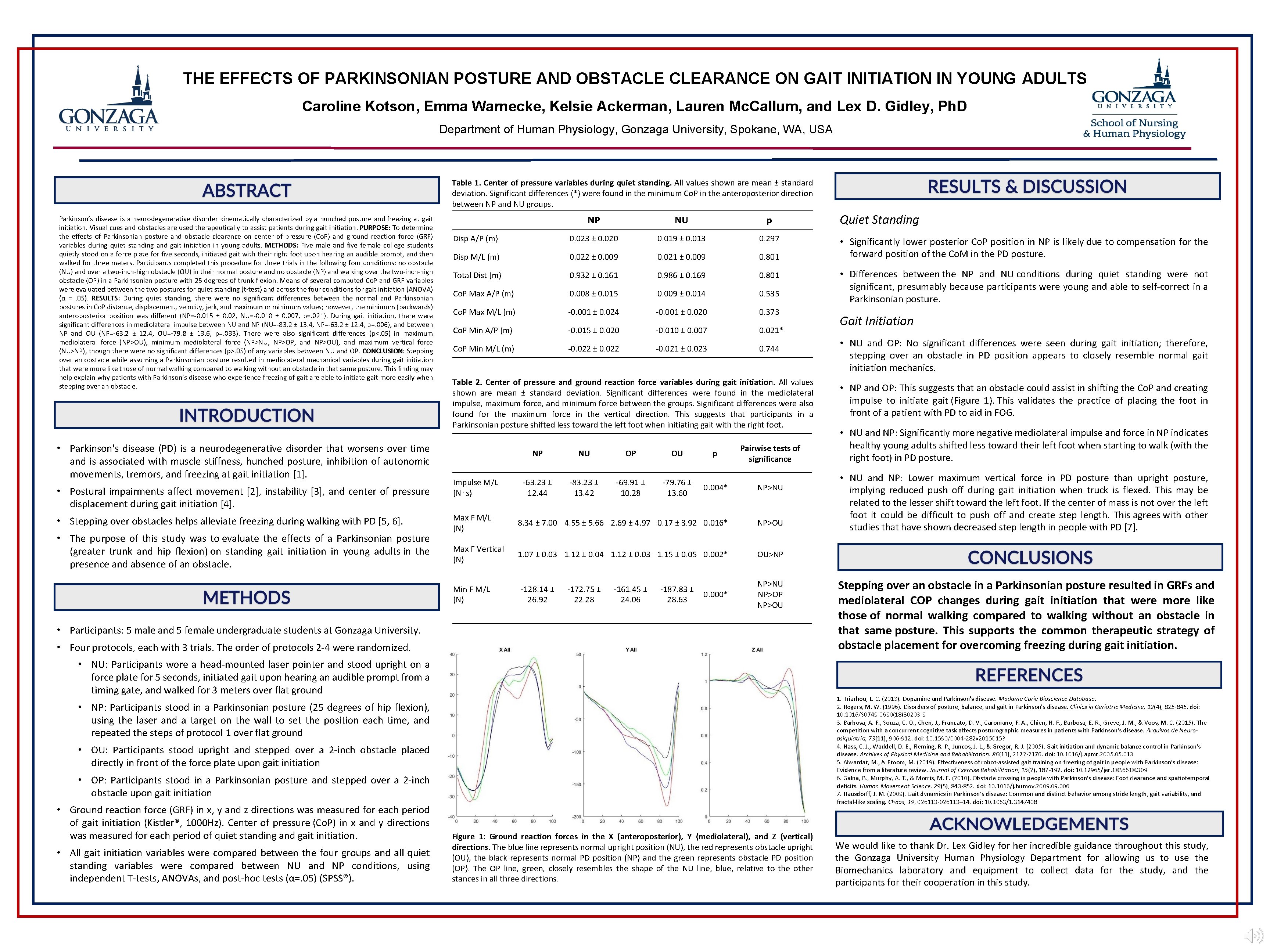
THE EFFECTS OF PARKINSONIAN POSTURE AND OBSTACLE CLEARANCE ON GAIT INITIATION IN YOUNG ADULTS Caroline Kotson, Emma Warnecke, Kelsie Ackerman, Lauren Mc. Callum, and Lex D. Gidley, Ph. D Department of Human Physiology, Gonzaga University, Spokane, WA, USA Table 1. Center of pressure variables during quiet standing. All values shown are mean ± standard deviation. Significant differences (*) were found in the minimum Co. P in the anteroposterior direction between NP and NU groups. Parkinson’s disease is a neurodegenerative disorder kinematically characterized by a hunched posture and freezing at gait initiation. Visual cues and obstacles are used therapeutically to assist patients during gait initiation. PURPOSE: To determine the effects of Parkinsonian posture and obstacle clearance on center of pressure (Co. P) and ground reaction force (GRF) variables during quiet standing and gait initiation in young adults. METHODS: Five male and five female college students quietly stood on a force plate for five seconds, initiated gait with their right foot upon hearing an audible prompt, and then walked for three meters. Participants completed this procedure for three trials in the following four conditions: no obstacle (NU) and over a two-inch-high obstacle (OU) in their normal posture and no obstacle (NP) and walking over the two-inch-high obstacle (OP) in a Parkinsonian posture with 25 degrees of trunk flexion. Means of several computed Co. P and GRF variables were evaluated between the two postures for quiet standing (t-test) and across the four conditions for gait initiation (ANOVA) (α =. 05). RESULTS: During quiet standing, there were no significant differences between the normal and Parkinsonian postures in Co. P distance, displacement, velocity, jerk, and maximum or minimum values; however, the minimum (backwards) anteroposterior position was different (NP=-0. 015 ± 0. 02, NU=-0. 010 ± 0. 007, p=. 021). During gait initiation, there were significant differences in mediolateral impulse between NU and NP (NU=-83. 2 ± 13. 4, NP=-63. 2 ± 12. 4, p=. 006), and between NP and OU (NP=-63. 2 ± 12. 4, OU=-79. 8 ± 13. 6, p=. 033). There were also significant differences (p<. 05) in maximum mediolateral force (NP>OU), minimum mediolateral force (NP>NU, NP>OP, and NP>OU), and maximum vertical force (NU>NP), though there were no significant differences (p>. 05) of any variables between NU and OP. CONCLUSION: Stepping over an obstacle while assuming a Parkinsonian posture resulted in mediolateral mechanical variables during gait initiation that were more like those of normal walking compared to walking without an obstacle in that same posture. This finding may help explain why patients with Parkinson’s disease who experience freezing of gait are able to initiate gait more easily when stepping over an obstacle. • Parkinson's disease (PD) is a neurodegenerative disorder that worsens over time and is associated with muscle stiffness, hunched posture, inhibition of autonomic movements, tremors, and freezing at gait initiation [1]. • Postural impairments affect movement [2], instability [3], and center of pressure displacement during gait initiation [4]. • Stepping over obstacles helps alleviate freezing during walking with PD [5, 6]. • The purpose of this study was to evaluate the effects of a Parkinsonian posture (greater trunk and hip flexion) on standing gait initiation in young adults in the presence and absence of an obstacle. NP NU p Disp A/P (m) 0. 023 ± 0. 020 0. 019 ± 0. 013 0. 297 Disp M/L (m) 0. 022 ± 0. 009 0. 021 ± 0. 009 0. 801 Total Dist (m) 0. 932 ± 0. 161 0. 986 ± 0. 169 0. 801 Co. P Max A/P (m) 0. 008 ± 0. 015 0. 009 ± 0. 014 0. 535 Co. P Max M/L (m) -0. 001 ± 0. 024 -0. 001 ± 0. 020 0. 373 Co. P Min A/P (m) -0. 015 ± 0. 020 -0. 010 ± 0. 007 0. 021* Co. P Min M/L (m) -0. 022 ± 0. 022 -0. 021 ± 0. 023 0. 744 Table 2. Center of pressure and ground reaction force variables during gait initiation. All values shown are mean ± standard deviation. Significant differences were found in the mediolateral impulse, maximum force, and minimum force between the groups. Significant differences were also found for the maximum force in the vertical direction. This suggests that participants in a Parkinsonian posture shifted less toward the left foot when initiating gait with the right foot. Impulse M/L (N⋅s) NP NU OP OU -63. 23 ± 12. 44 -83. 23 ± 13. 42 -69. 91 ± 10. 28 -79. 76 ± 13. 60 p Pairwise tests of significance 0. 004* NP>NU Max F M/L (N) 8. 34 ± 7. 00 4. 55 ± 5. 66 2. 69 ± 4. 97 0. 17 ± 3. 92 0. 016* NP>OU Max F Vertical (N) 1. 07 ± 0. 03 1. 12 ± 0. 04 1. 12 ± 0. 03 1. 15 ± 0. 05 0. 002* OU>NP Min F M/L (N) -128. 14 ± 26. 92 -172. 75 ± 22. 28 -161. 45 ± 24. 06 -187. 83 ± 28. 63 0. 000* NP>NU NP>OP NP>OU • Participants: 5 male and 5 female undergraduate students at Gonzaga University. • Four protocols, each with 3 trials. The order of protocols 2 -4 were randomized. • NU: Participants wore a head-mounted laser pointer and stood upright on a force plate for 5 seconds, initiated gait upon hearing an audible prompt from a timing gate, and walked for 3 meters over flat ground • OU: Participants stood upright and stepped over a 2 -inch obstacle placed directly in front of the force plate upon gait initiation • OP: Participants stood in a Parkinsonian posture and stepped over a 2 -inch obstacle upon gait initiation • All gait initiation variables were compared between the four groups and all quiet standing variables were compared between NU and NP conditions, using independent T-tests, ANOVAs, and post-hoc tests (α=. 05) (SPSS®). • Significantly lower posterior Co. P position in NP is likely due to compensation for the forward position of the Co. M in the PD posture. • Differences between the NP and NU conditions during quiet standing were not significant, presumably because participants were young and able to self-correct in a Parkinsonian posture. Gait Initiation • NU and OP: No significant differences were seen during gait initiation; therefore, stepping over an obstacle in PD position appears to closely resemble normal gait initiation mechanics. • NP and OP: This suggests that an obstacle could assist in shifting the Co. P and creating impulse to initiate gait (Figure 1). This validates the practice of placing the foot in front of a patient with PD to aid in FOG. • NU and NP: Significantly more negative mediolateral impulse and force in NP indicates healthy young adults shifted less toward their left foot when starting to walk (with the right foot) in PD posture. • NU and NP: Lower maximum vertical force in PD posture than upright posture, implying reduced push off during gait initiation when truck is flexed. This may be related to the lesser shift toward the left foot. If the center of mass is not over the left foot it could be difficult to push off and create step length. This agrees with other studies that have shown decreased step length in people with PD [7]. Stepping over an obstacle in a Parkinsonian posture resulted in GRFs and mediolateral COP changes during gait initiation that were more like those of normal walking compared to walking without an obstacle in that same posture. This supports the common therapeutic strategy of obstacle placement for overcoming freezing during gait initiation. 1. Triarhou, L. C. (2013). Dopamine and Parkinson's disease. Madame Curie Bioscience Database. 2. Rogers, M. W. (1996). Disorders of posture, balance, and gait in Parkinson's disease. Clinics in Geriatric Medicine, 12(4), 825 -845. doi: 10. 1016/S 0749 -0690(18)30203 -9 3. Barbosa, A. F. , Souza, C. O. , Chen, J. , Francato, D. V. , Caromano, F. A. , Chien, H. F. , Barbosa, E. R. , Greve, J. M. , & Voos, M. C. (2015). The competition with a concurrent cognitive task affects posturographic measures in patients with Parkinson's disease. Arquivos de Neuropsiquiatria, 73(11), 906 -912. doi: 10. 1590/0004 -282 x 20150153 4. Hass, C. J. , Waddell, D. E. , Fleming, R. P. , Juncos, J. L. , & Gregor, R. J. (2005). Gait initiation and dynamic balance control in Parkinson's disease. Archives of Physical Medicine and Rehabilitation, 86(11), 2172 -2176. doi: 10. 1016/j. apmr. 2005. 013 5. Alwardat, M. , & Etoom, M. (2019). Effectiveness of robot-assisted gait training on freezing of gait in people with Parkinson's disease: Evidence from a literature review. Journal of Exercise Rehabilitation, 15(2), 187 -192. doi: 10. 12965/jer. 1836618. 309 6. Galna, B. , Murphy, A. T. , & Morris, M. E. (2010). Obstacle crossing in people with Parkinson's disease: Foot clearance and spatiotemporal deficits. Human Movement Science, 29(5), 843 -852. doi: 10. 1016/j. humov. 2009. 006 7. Hausdorff, J. M. (2009). Gait dynamics in Parkinson’s disease: Common and distinct behavior among stride length, gait variability, and fractal-like scaling. Chaos, 19, 026113 -026113– 14. doi: 10. 1063/1. 3147408 • NP: Participants stood in a Parkinsonian posture (25 degrees of hip flexion), using the laser and a target on the wall to set the position each time, and repeated the steps of protocol 1 over flat ground • Ground reaction force (GRF) in x, y and z directions was measured for each period of gait initiation (Kistler®, 1000 Hz). Center of pressure (Co. P) in x and y directions was measured for each period of quiet standing and gait initiation. Quiet Standing Figure 1: Ground reaction forces in the X (anteroposterior), Y (mediolateral), and Z (vertical) directions. The blue line represents normal upright position (NU), the red represents obstacle upright (OU), the black represents normal PD position (NP) and the green represents obstacle PD position (OP). The OP line, green, closely resembles the shape of the NU line, blue, relative to the other stances in all three directions. We would like to thank Dr. Lex Gidley for her incredible guidance throughout this study, the Gonzaga University Human Physiology Department for allowing us to use the Biomechanics laboratory and equipment to collect data for the study, and the participants for their cooperation in this study.
- Slides: 1