The East Midlands Breathomics Pathology Node EMBER Salman

The East Midlands Breathomics Pathology Node (EMBER) Salman Siddiqui Professor of Airways Disease EMBER Chief Investigator and WS 4 lead

Work Strand 4 Clinical disease cohorts- 'breathomic' discovery and clinical validation & objective - ‘Breathomic' discovery and early clinical validation - Exemplar Clinical Project 'breathless' patient to include airways disease, heart failure and community acquired pneumonia in the acute setting in adults and children compared with healthy volunteers’

The Team • Clinical Operations Group • Prof Tim Coates • Dr Erol Gaillard • Dr Caroline Beardsmore • Dr Neil Greening • NIHR BRC • NIHR CRF • College of Life Sciences • Precision Medicine Institute

Burden of Acute Breathlessness • Shortness of breath is the chief complaint for about 8% of 999 calls to the ambulance service, and is the third most common type of emergency call [1] • Dyspnoea is a strong predictor of mortality in patients with COPD and heart failure and amongst the most common causes for emergency department visits [2] • Diagnostic uncertainty remains in ≈ 30% and is associated with adverse outcomes[3, 4]. • Commonest cardio-respiratory conditions that cause acute breathlessness are (i) exacerbations of asthma and COPD, (ii) acute heart failure and (i) community acquired pneumonia [4] [1] Wollard et al BMJ 2004, [2] Abidov et al, NEJM (2005), [3] Lam L et al, Ann Int Med 2010, [4] Green SM et al, Arch Intern Med 2008, [4] D Ponka et al Can Fam Physician (2007)
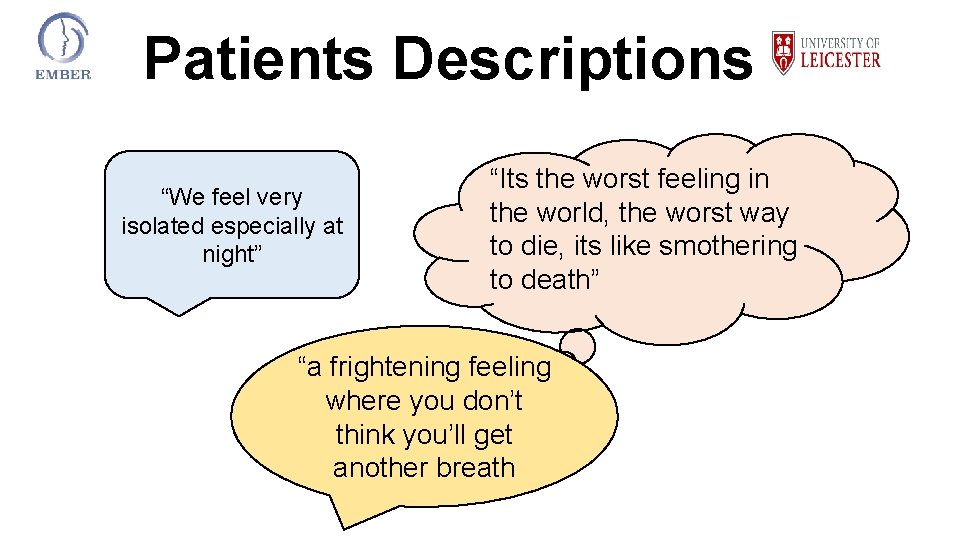
Patients Descriptions “We feel very isolated especially at night” “Its the worst feeling in the world, the worst way to die, its like smothering to death” “a frightening feeling where you don’t think you’ll get another breath
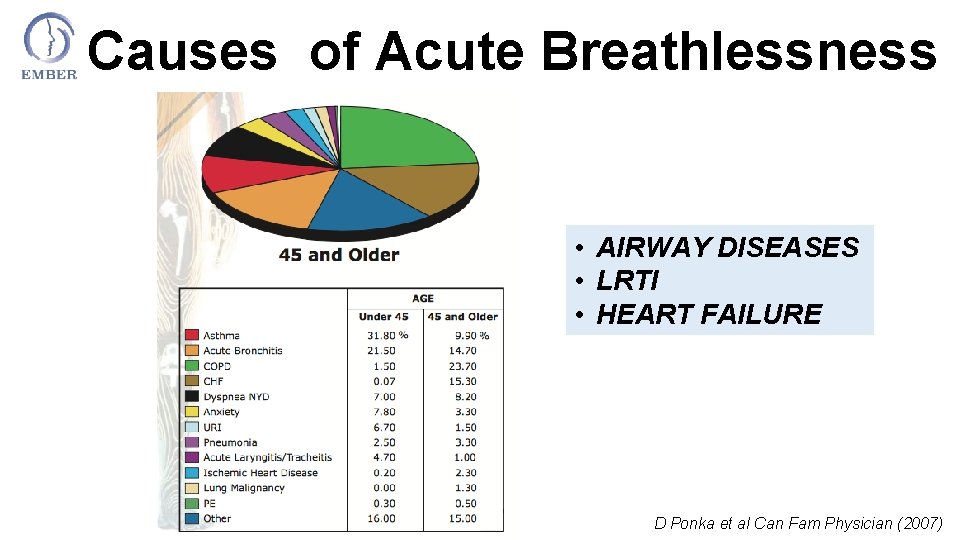
Causes of Acute Breathlessness • AIRWAY DISEASES • LRTI • HEART FAILURE D Ponka et al Can Fam Physician (2007)

Definition Acute Breathlessness - Patient Reported (binary classification) - A change in breathlessness of the e-MRC score of at least a unit from stable state - 100 mm visual analogue scale
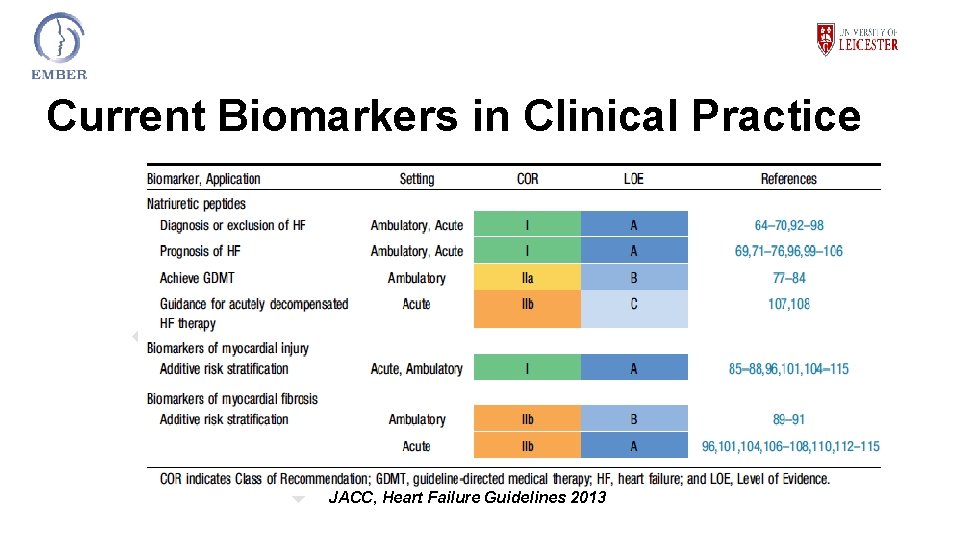
Current Biomarkers in Clinical Practice BNP CRP Eosb TNT • UHL pathology based markers used to make clinical decisions in acute breathlessness • BNP, TNT and CRP: well defined roles • EOSb – early POC guiding OCS therapy • Varying levels of turnaround • All have been adapted for point of care testing JACC, Heart Failure Guidelines 2013
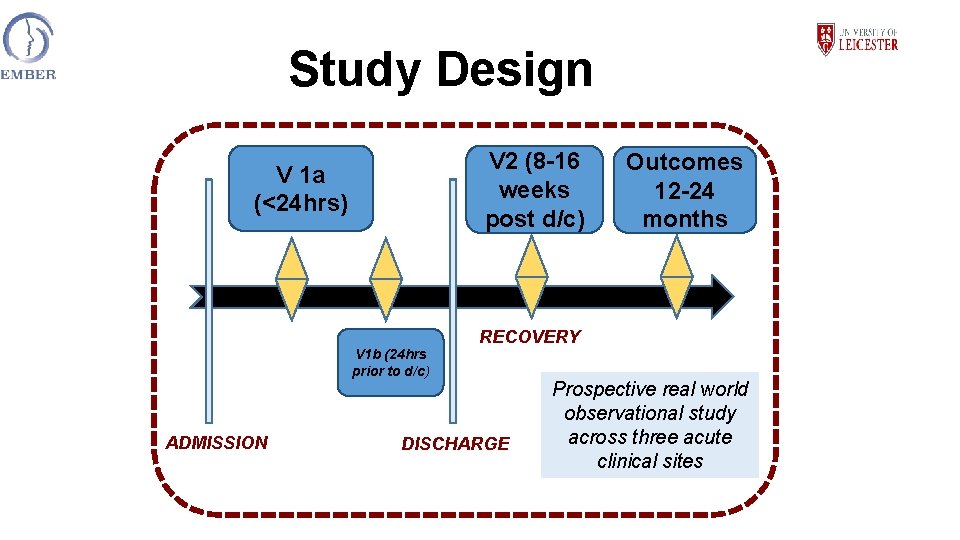
Study Design V 2 (8 -16 weeks post d/c) V 1 a (<24 hrs) V 1 b (24 hrs prior to d/c) ADMISSION Outcomes 12 -24 months RECOVERY DISCHARGE Prospective real world observational study across three acute clinical sites

Aims • Primary Objective • To evaluate the sensitivity, specificity, positive and negative predictive value of metabolomic biomarkers in exhaled breath samples to identify acute breathlessness • Secondary objectives • To discover and replicate metabolomic breath biomarkers • To discover and replicate (combined discovery and replication phase) metabolomic breath biomarkers that differentiate (i) Acute Heart failure (ii) Community acquired pneumonia, (iii) Adult exacerbations of asthma and COPD, (iv) paediatric exacerbations of asthma, (vi) stable state disease/health

Core Phenotyping • • SYSTEMATIC EVALUATION OF VIRUSES, MICROBIOME, MYCOBIOME PROTEOME …. . • • • Diet/Lifestyle/Environment Clinical Meta Data Co-morbidities Blood Biomarkers: BNP/CRP/Troponin T/blood eosinophils Medication Usage Imaging: CXR, ECG, Echo: between visit 1 a and 1 b, quadriceps USS (COPD/Heart failure) Lung Function: Spirometry/FENO/Airway Oscillometry Biobank: urine, serum, plasma, PAXGENE*2 (DNA/RNA) & sputum (supernatants/plugs) Frailty measures Psychological & Cognitive function Health Status
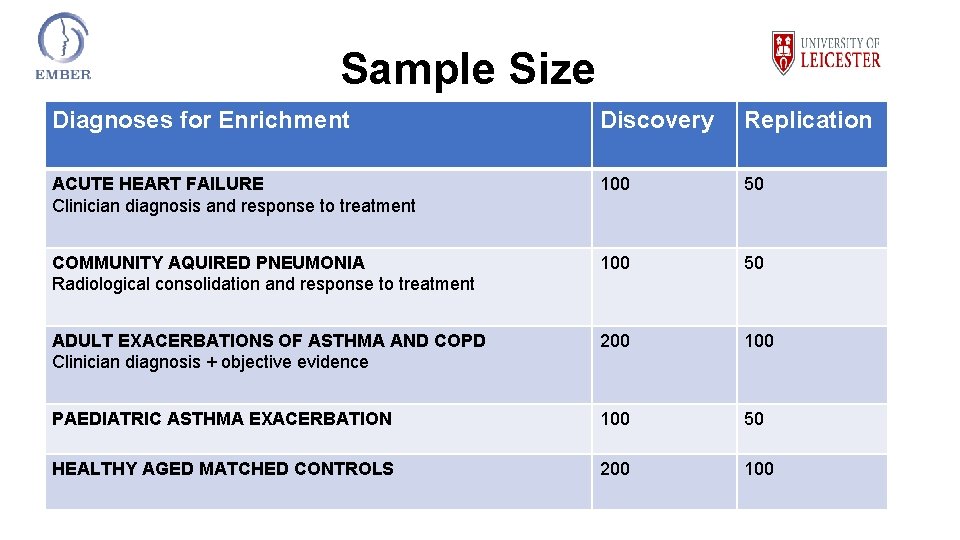
Sample Size Diagnoses for Enrichment Discovery Replication • Output ACUTE HEART FAILURE 100 50 Clinician diagnosis and response to treatment - Sensitvity, Specificity, PPV, NPV selected biomarkers in breath and blood (symptom & disease based classification) COMMUNITY AQUIRED PNEUMONIA 100 50 Radiological consolidation and response to treatment - Multinomial prediction models and derived ROC analyses • Estimates for upper an lower bound for PPV and NPV for ADULT EXACERBATIONS OF ASTHMA AND COPD 200 100 e. VOC unknown no preliminary data in acute disease Clinician diagnosis + objective evidence • Initial approach: empirical estimate of sample size per disease PAEDIATRIC ASTHMA EXACERBATION 100 50 class followed by planned interim analyses HEALTHY AGED MATCHED CONTROLS 200 100
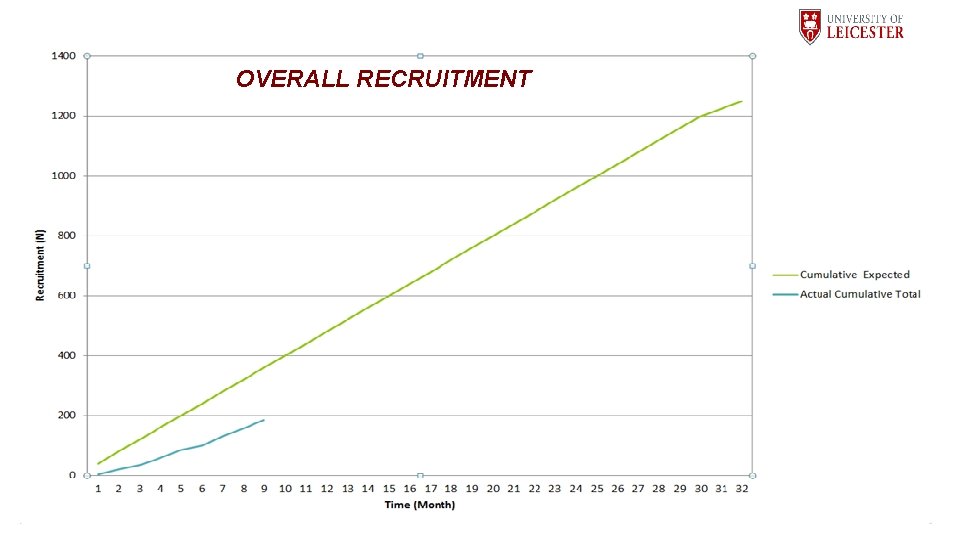
OVERALL RECRUITMENT
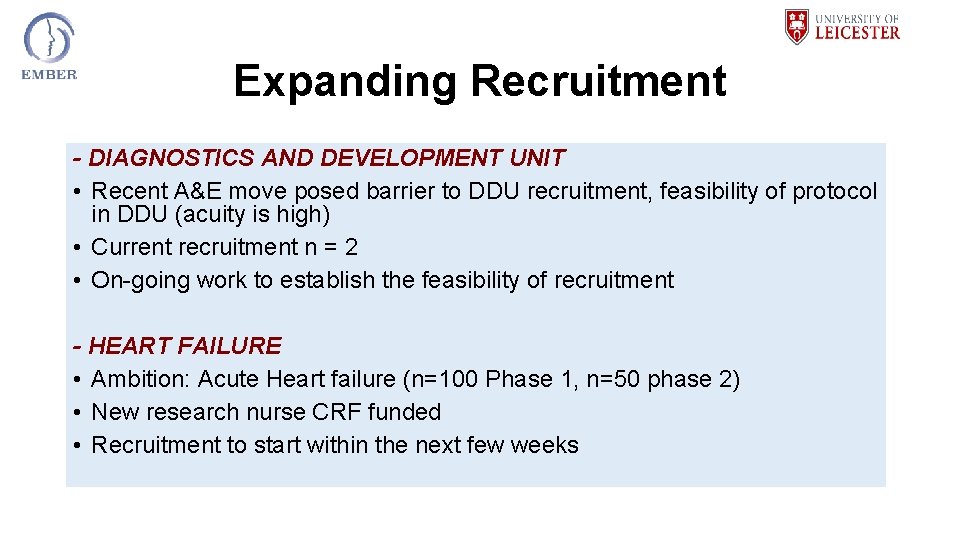
Expanding Recruitment - DIAGNOSTICS AND DEVELOPMENT UNIT • Recent A&E move posed barrier to DDU recruitment, feasibility of protocol in DDU (acuity is high) • Current recruitment n = 2 • On-going work to establish the feasibility of recruitment - HEART FAILURE • Ambition: Acute Heart failure (n=100 Phase 1, n=50 phase 2) • New research nurse CRF funded • Recruitment to start within the next few weeks
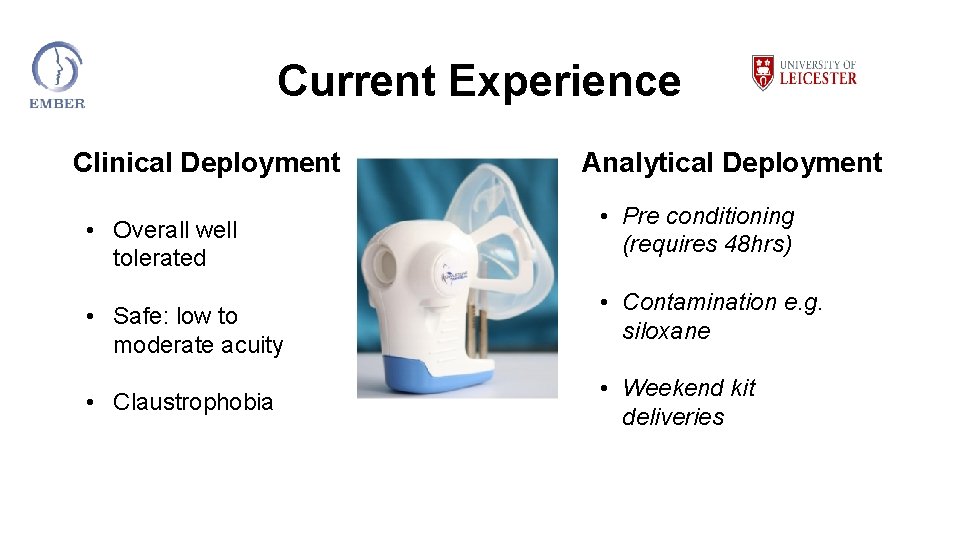
Current Experience Clinical Deployment Analytical Deployment • Overall well tolerated • Safe: low to moderate acuity • Claustrophobia • Pre conditioning (requires 48 hrs) • Contamination e. g. siloxane • Weekend kit deliveries
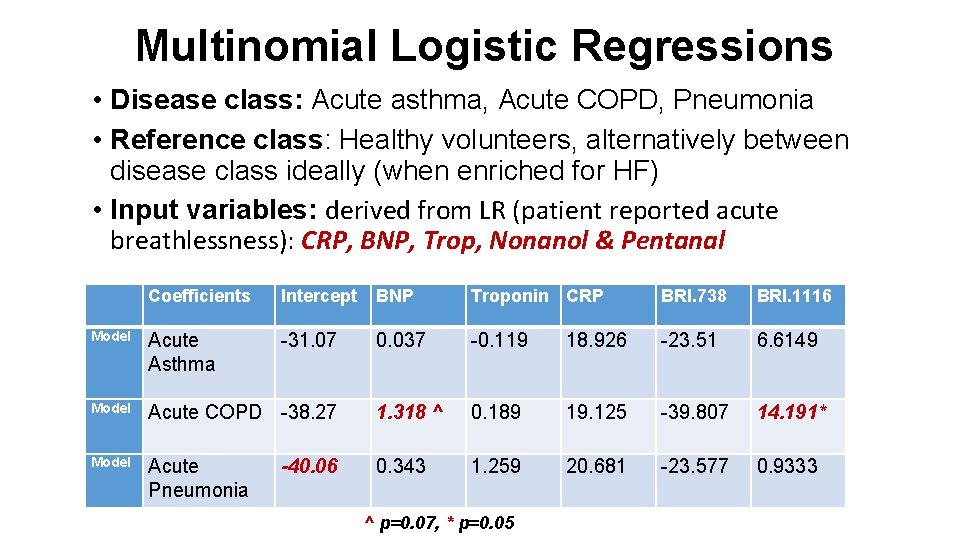
Multinomial Logistic Regressions • Disease class: Acute asthma, Acute COPD, Pneumonia • Reference class: Healthy volunteers, alternatively between disease class ideally (when enriched for HF) • Input variables: derived from LR (patient reported acute breathlessness): CRP, BNP, Trop, Nonanol & Pentanal Coefficients Model Intercept BNP Troponin CRP BRI. 738 BRI. 1116 Acute -31. 07 Asthma Acute COPD -38. 27 0. 037 -0. 119 18. 926 -23. 51 6. 6149 1. 318 ^ 0. 189 19. 125 -39. 807 14. 191* Acute Pneumonia 0. 343 1. 259 20. 681 -23. 577 0. 9333 -40. 06 ^ p=0. 07, * p=0. 05
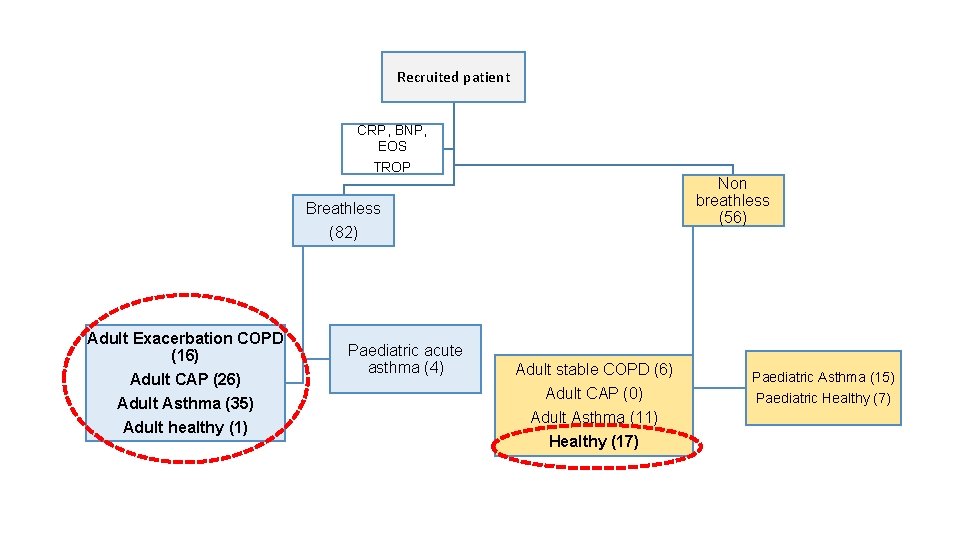
Recruited patient CRP, BNP, EOS TROP Non breathless (56) Breathless (82) Adult Exacerbation COPD (16) Adult CAP (26) Adult Asthma (35) Adult healthy (1) Paediatric acute asthma (4) Adult stable COPD (6) Adult CAP (0) Adult Asthma (11) Healthy (17) Paediatric Asthma (15) Paediatric Healthy (7)
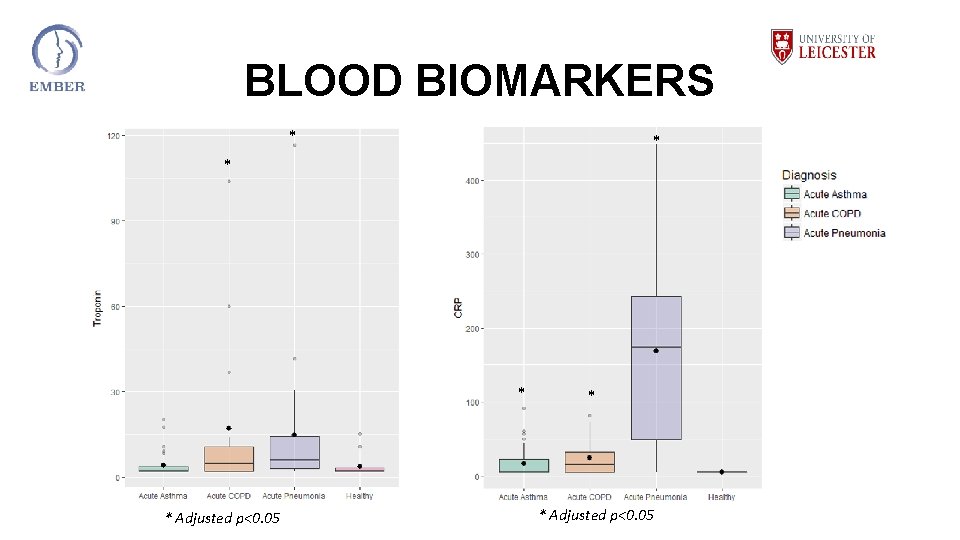
BLOOD BIOMARKERS * * * Adjusted p<0. 05
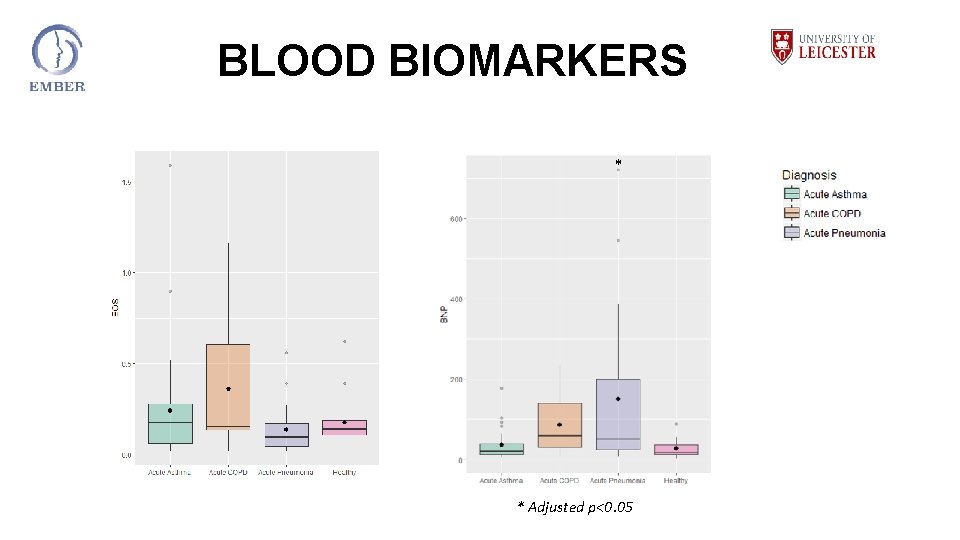
BLOOD BIOMARKERS * * Adjusted p<0. 05
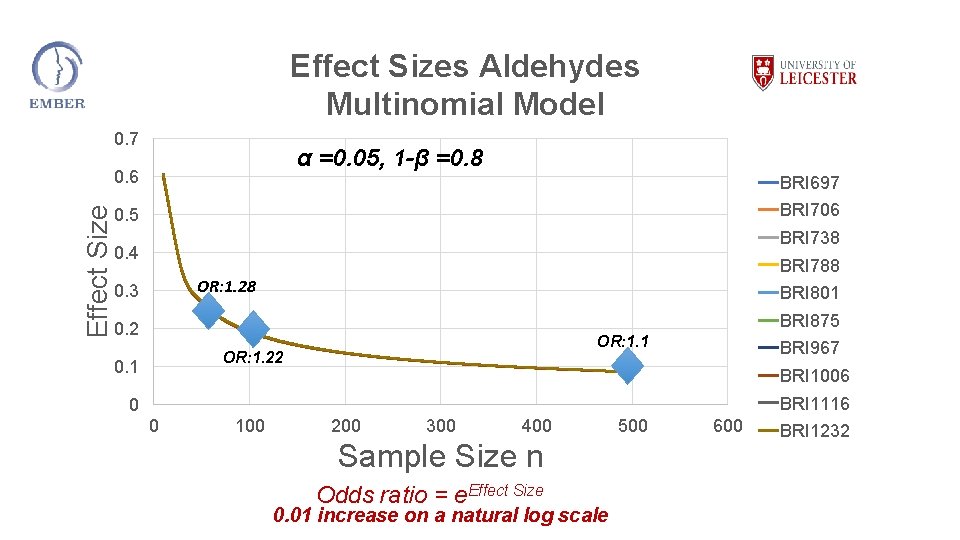
Effect Sizes Aldehydes Multinomial Model 0. 7 α =0. 05, 1 -β =0. 8 Effect Size 0. 6 BRI 697 BRI 706 0. 5 BRI 738 0. 4 BRI 788 OR: 1. 28 0. 3 BRI 801 BRI 875 0. 2 OR: 1. 1 OR: 1. 22 0. 1 BRI 967 BRI 1006 BRI 1116 0 0 100 200 300 400 Sample Size n Odds ratio = e. Effect Size 0. 01 increase on a natural log scale 500 600 BRI 1232

Conclusions • In the first year of recruitment promising levels of recruitment: asthma, COPD and pneumonia in adults • Demonstrated feasibility of recruitment in children acutely • Evaluating feasibility of heart failure and DDU recruitment with additional resource • Sample size estimates demonstrated that we will be well powered to detect relatively small effect sizes between acute classes and controls.
- Slides: 21