The Early History of Chemistry 4 Greeks 400




















- Slides: 29

The Early History of Chemistry 4 Greeks 400 B. C. Four fundamental substances – fire, earth, water, a air. - 4 Democritus – uses term “atomos” (atoms) to describe small, indivisible matter. No experiments to support the idea, so it is dropped. Before 16 th Century – Alchemy: Attempts (scientific or otherwise) to change cheap metals into gold Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 1

The Early History of Chemistry 4 17 th Century – Robert Boyle: First “chemist” to perform quantitative experiments (pressure/volume) –- Incorrectly believed that the alchemist’s view that metals were not true elements and that a way would eventually be found to change one metal into another. 4 18 th Century – George Stahl: Phlogiston flows out of a burning material. – Joseph Priestley: Discovers oxygen gas, “dephlogisticated air. ” Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 2

Law of Conservation of Mass 4 Discovered 4 Mass by Antoine Lavoisier is neither created nor destroyed 4 Combustion involves oxygen, not phlogiston Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 3

Other Fundamental Chemical Laws Law of Definite Proportion 4 A given compound always contains exactly the same proportion of elements by mass. 4 Na. Cl – always 39. 34% Na and 60. 66% Cl (mass) 4 Carbon tetrachloride is always 1 atom carbon per 4 atoms chlorine. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 4

Other Fundamental Chemical Laws Law of Multiple Proportions 4 When two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. 4 The ratio of the masses of oxygen in H 2 O and H 2 O 2 will be a small whole number (“ 2”). Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 5

Law of Multiple Proportions Mass of oxygen that combines with 1 g of Carbon Compound 1 (CO) 1. 33 g Compound 2 (CO 2) 2. 66 g Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 6

Dalton’s Atomic Theory (1808) Ê Each element is made up of tiny particles called atoms. Ë The atoms of a given element are identical; the atoms of different elements are different in some fundamental way or ways. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 7

Dalton’s Atomic Theory (continued) Ì Chemical compounds are formed when atoms combine with each other. A given compound always has the same relative numbers and types of atoms. Í Chemical reactions involve reorganization of the atoms - changes in the way they are bound together. The atoms themselves are not changed in a chemical reaction. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 8

Dalton’s Atomic Theory (continued) 5. Atoms cannot be subdivided, created, or destroyed. 6. Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 9

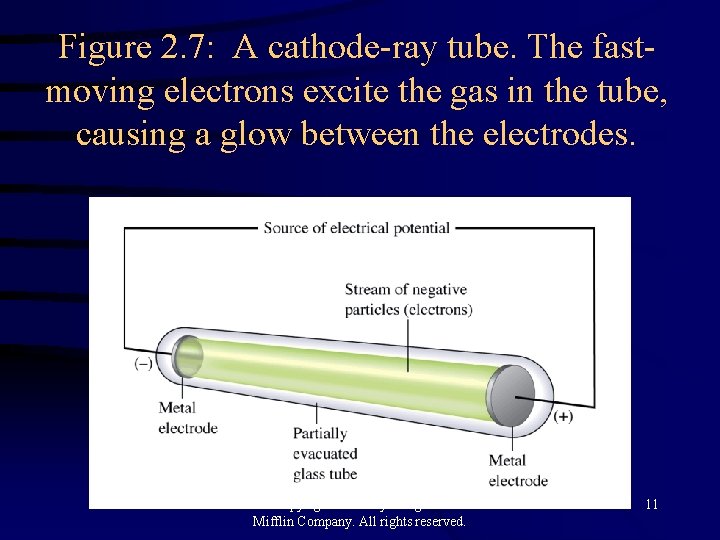
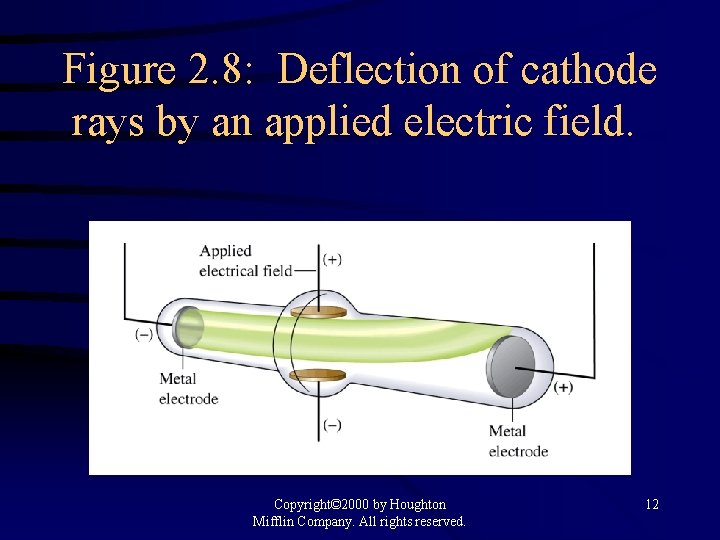
Early Experiments to Characterize the Atom H J. J. Thomson - postulated the existence of electrons using cathode ray tubes. H Ernest Rutherford - explained the nuclear atom, containing a dense nucleus with electrons traveling around the nucleus at a large distance. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 10

Figure 2. 7: A cathode-ray tube. The fastmoving electrons excite the gas in the tube, causing a glow between the electrodes. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 11

Figure 2. 8: Deflection of cathode rays by an applied electric field. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 12

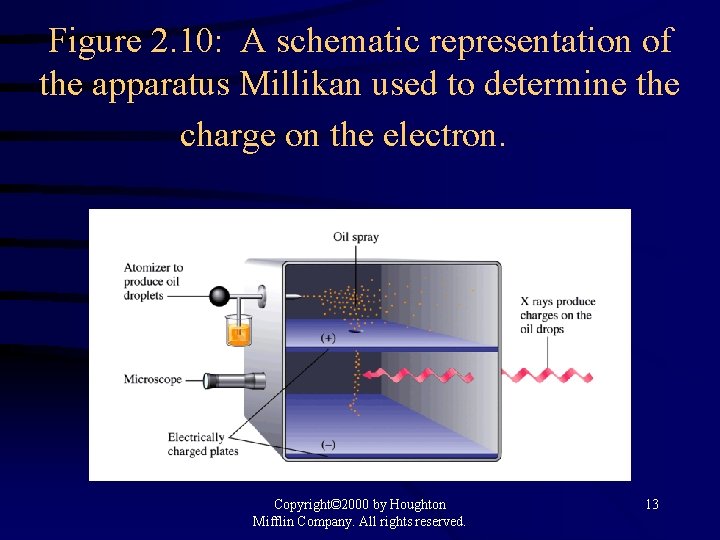
Figure 2. 10: A schematic representation of the apparatus Millikan used to determine the charge on the electron. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 13

The Electron Tiny, negatively charged particle H Very light compared to the mass of an atom – 1/1837 th the mass of a H atom H Move extremely rapidly within the atom. H Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 14

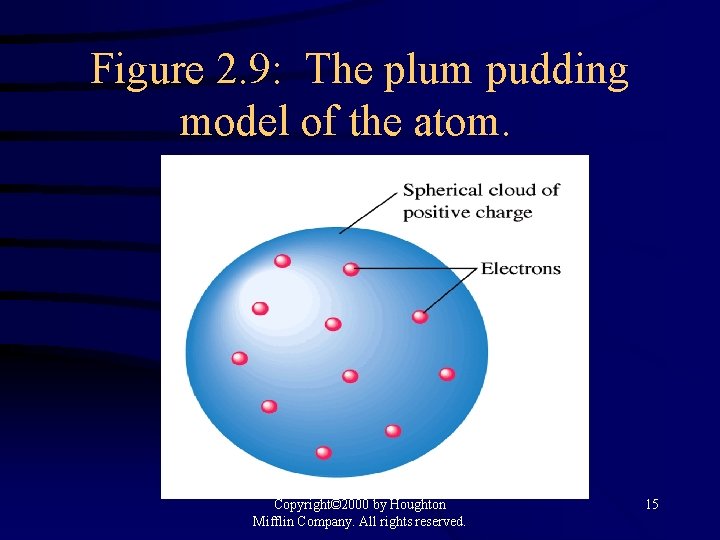
Figure 2. 9: The plum pudding model of the atom. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 15

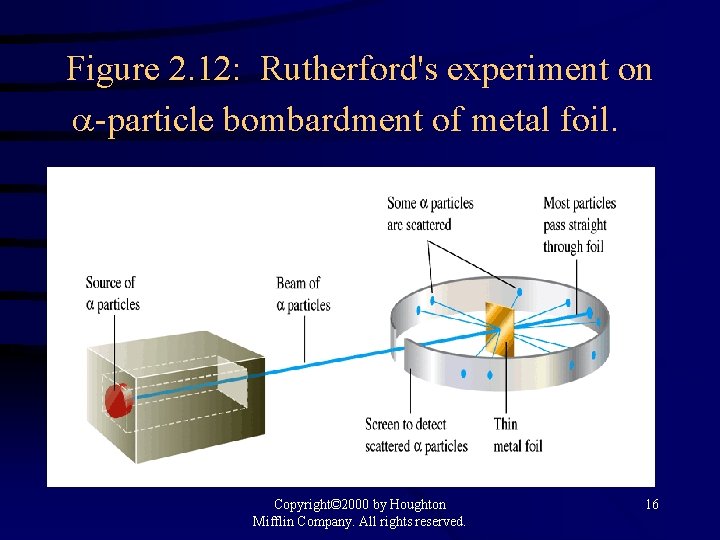
Figure 2. 12: Rutherford's experiment on -particle bombardment of metal foil. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 16

Figure 2. 13: (a) The expected results of the metal foil experiment if Thomson's model were correct. (b)Actual results. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 17

The Modern View of Atomic Structure l : Electrons electron cloud l Protons: found in the nucleus, they have a positive charge equal in magnitude to the electron’s negative charge. l Neutrons: l The nucleus contains: protons and neutrons. found in the nucleus, virtually same mass as a proton but no charge. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 18

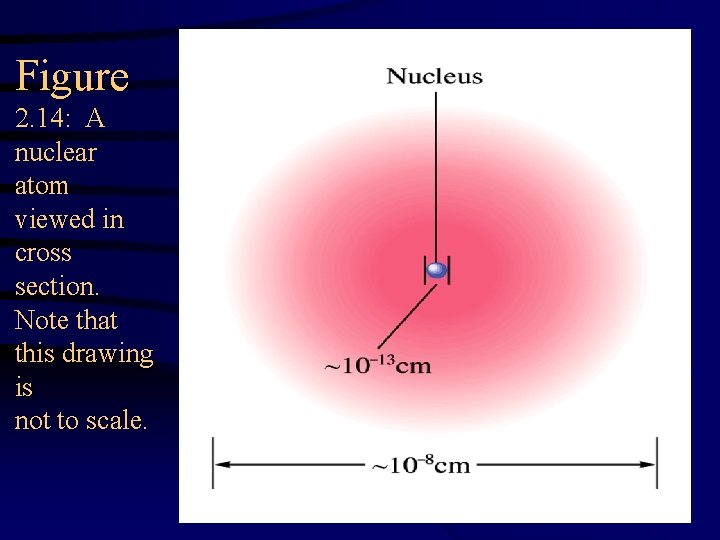
Figure 2. 14: A nuclear atom viewed in cross section. Note that this drawing is not to scale.

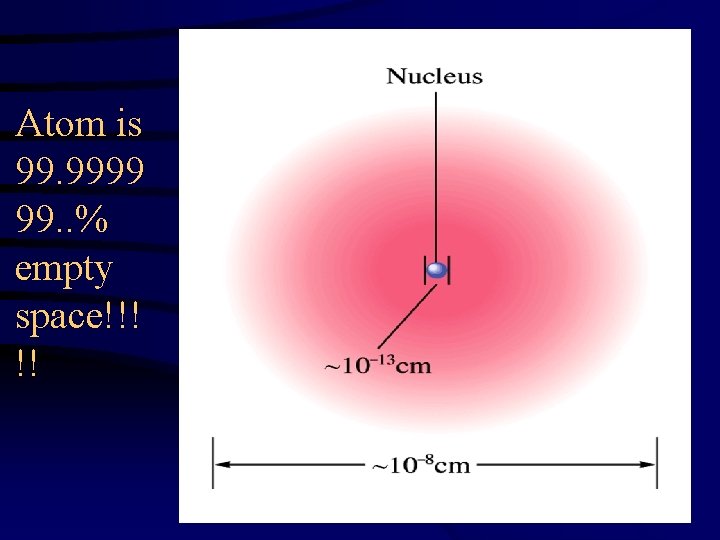
Atom is 99. 9999 99. . % empty space!!! !!

The Mass and Charge of the Electron, Proton, and Neutron Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 21

The Mass and Charge of the Electron, Proton, and Neutron Particle Electron Relative Mass 0 Relative Charge -1 Proton 1 +1 Electron cloud Nucleus Neutron 1 0 Nucleus Copyright© 2000 by Houghton Mifflin Company. All rights reserved. Location 22

Atomic Number / Atomic Mass H The atomic number is equal to the number of protons. H The atomic mass is equal to the number of protons + neutrons Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 23

Atomic Number / Atomic Mass H Essentially, all of the mass of the atom is considered to reside in the nucleus. H In a neutral atom, the number of protons equals the number of electrons. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 24

Isotopes All atoms of an element have the same number of protons. H The number of protons = the atomic number H Atoms of an element with different numbers of neutrons are called isotopes. H Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 25

Isotopes continued… All isotopes of an element are chemically identical H Isotopes have different masses H Isotopes are identified by their mass number. H Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 26

The Chemists’ Shorthand: Atomic Symbols Mass number Atomic number 39 K 19 Element Symbol Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 27

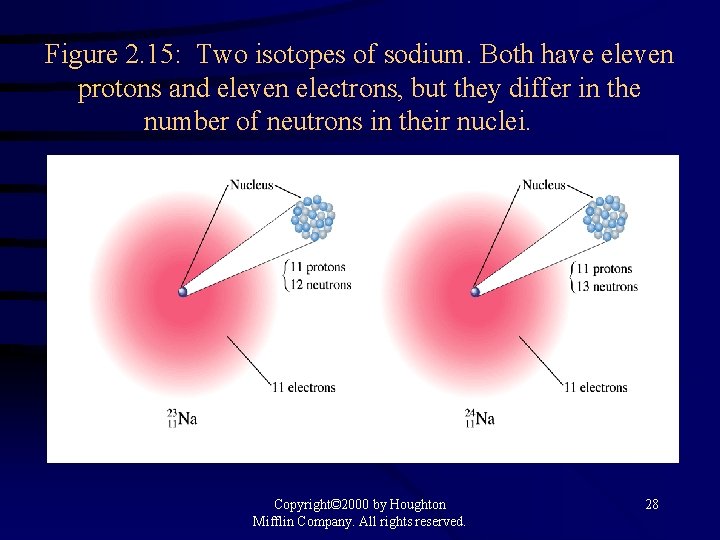
Figure 2. 15: Two isotopes of sodium. Both have eleven protons and eleven electrons, but they differ in the number of neutrons in their nuclei. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 28

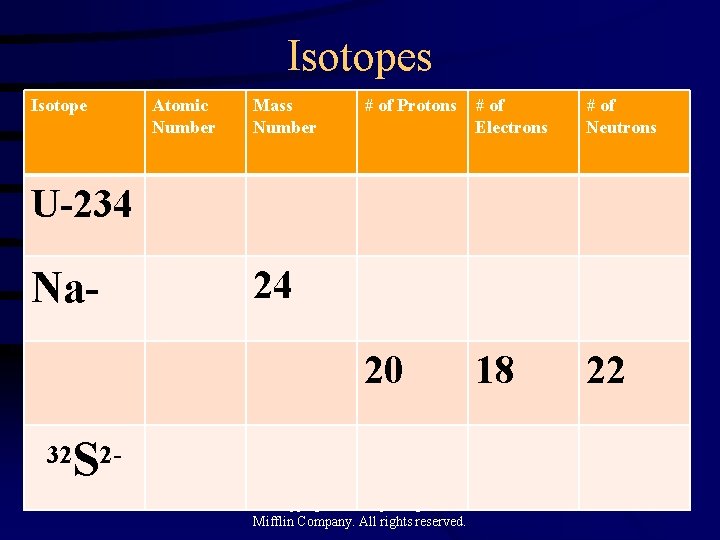
Isotopes Isotope Atomic Number Mass Number # of Protons # of Electrons # of Neutrons 20 18 22 U-234 Na- 24 S 2 - 32 Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 29