The Dynamics of Ctermini of Microtubules in Dendrites

The Dynamics of C-termini of Microtubules in Dendrites: A possible clue for the role of neuronal cytoskeleton in the functioning of the brain Jack Tuszynski Department of Physics University of Alberta Edmonton, Canada

Acknowledgements Avner Priel, Bar Ilan U/Univ of Alberta Nancy Woolf, UCLA Eric Carpenter Tyler Luchko Horacio Cantiello, Harvard Med School Stephanie Portet, U Toronto Funding: NSERC, MITACS, Ye. Ta. Del, Technology Innovations LLC
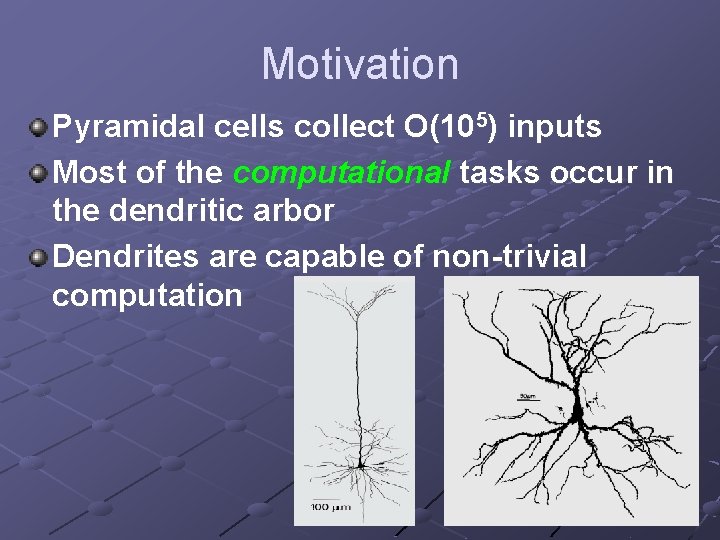
Motivation Pyramidal cells collect O(105) inputs Most of the computational tasks occur in the dendritic arbor Dendrites are capable of non-trivial computation
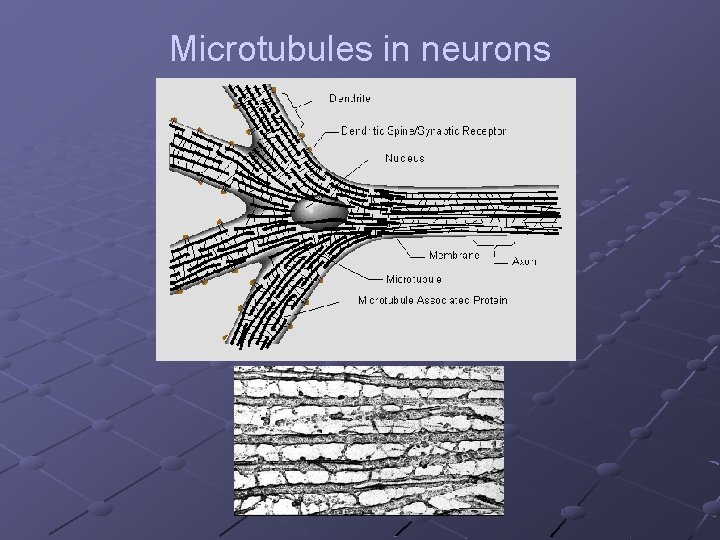
Microtubules in neurons
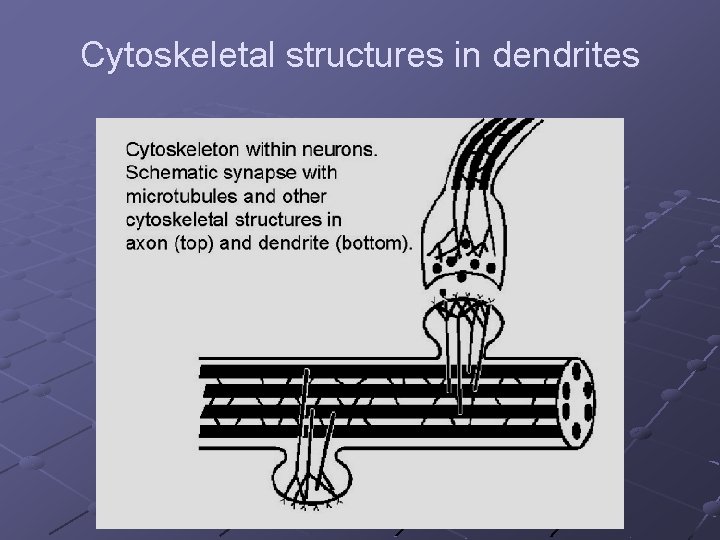
Cytoskeletal structures in dendrites
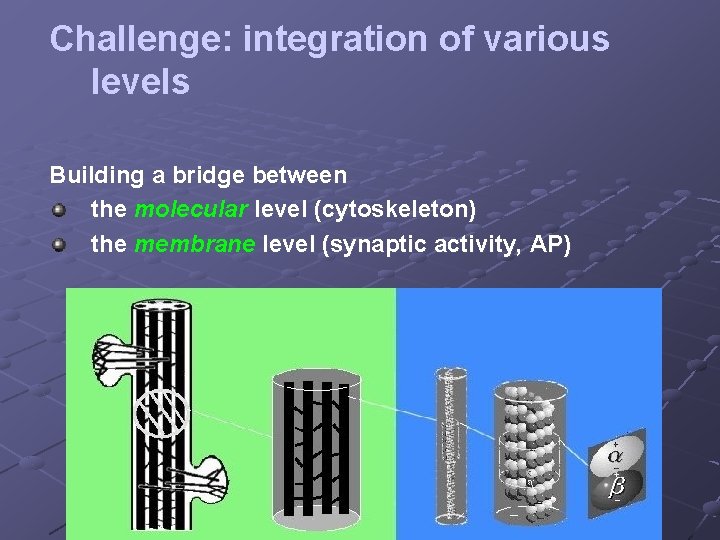
Challenge: integration of various levels Building a bridge between the molecular level (cytoskeleton) the membrane level (synaptic activity, AP)
Dendrites undergo significant structural plasticity during learning phases n n Spine level changes Cytoskeletal changes
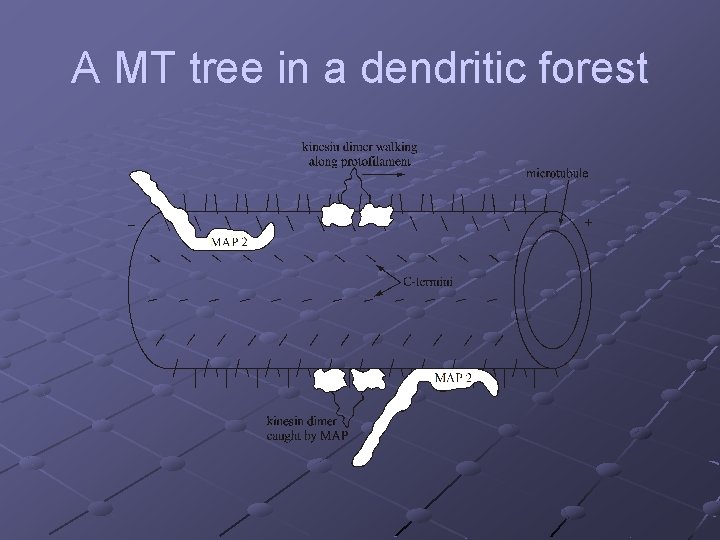
A MT tree in a dendritic forest
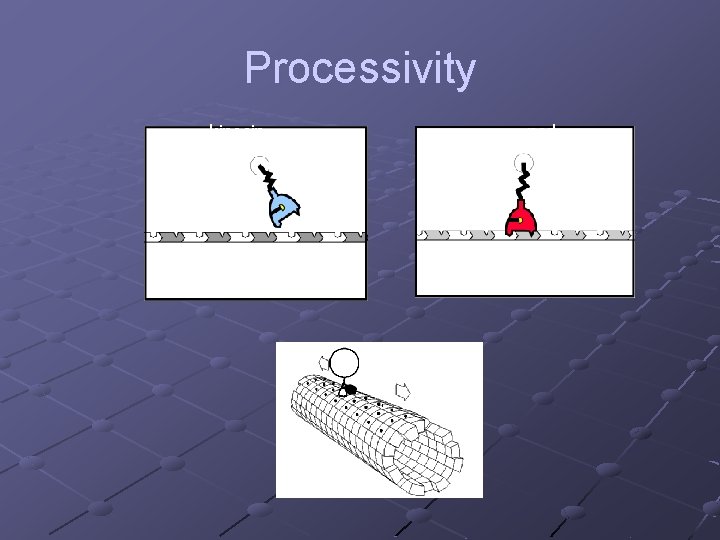
Processivity kinesin ncd
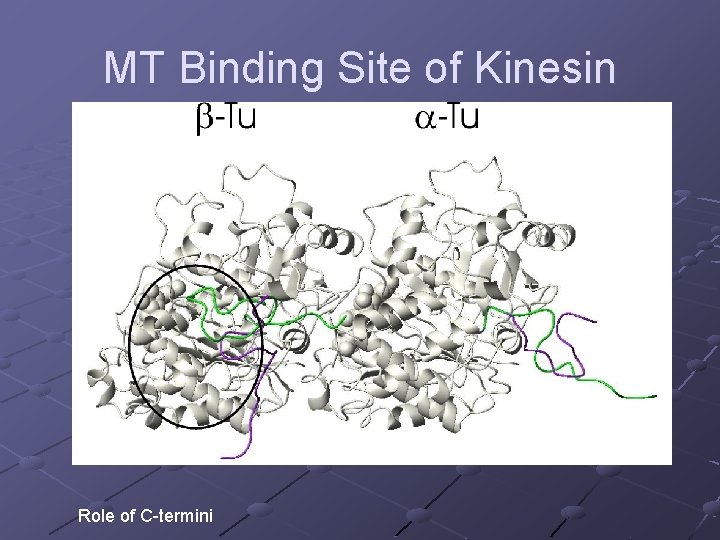
MT Binding Site of Kinesin Role of C-termini

The Model MTN – Microtubule Network A model of a single MT n Tubulin dimers interact electrically via dipole – dipole interactions A model of the MAP n Elastic rod connected at both ends to adjacent MTs MAP – MT interaction n Mechanical (elastic forces)
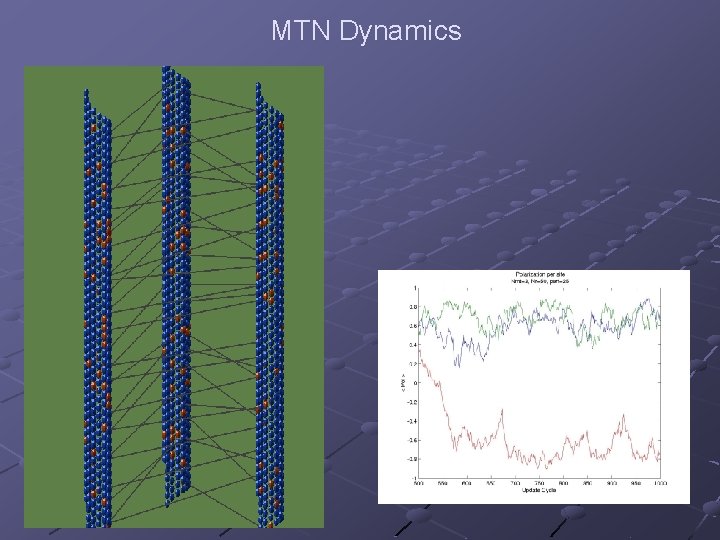
MTN Dynamics

Microtubules (MT) lengths vary, but commonly reach 5– 10 µm usually 13 protofilaments in vivo 12– 17 protofilaments when self-assembled in vitro protofilaments are strongly bound internally protein filaments of the cytoskeleton

Microtubules in dendrites : Self-assemble to extend dendrites (and axons) Form synaptic connections Linked to ion-channels and synaptic receptors Organized in parallel structures – interconnected by MAP-2 Appear in mixed polarity
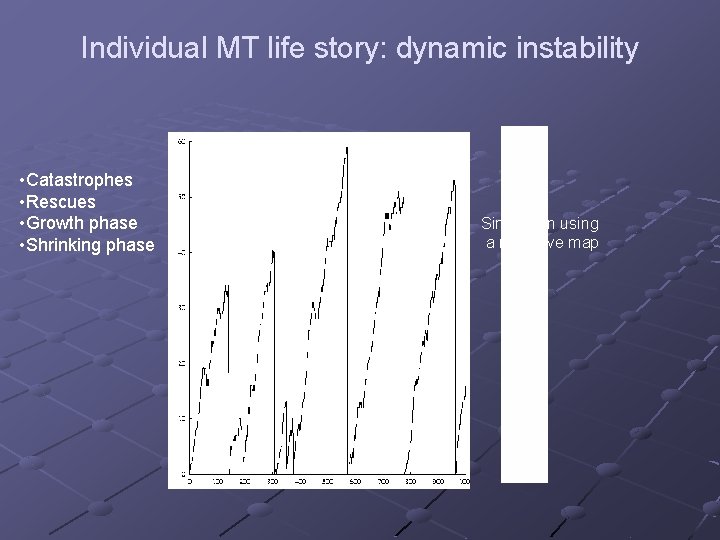
Individual MT life story: dynamic instability • Catastrophes • Rescues • Growth phase • Shrinking phase Simulation using a recursive map

Molecular Dynamics (MD) class of model system point masses (atoms) simple forces n n n bond stretching, Coulomb, van der Waals, etc. Newtonian integration over time
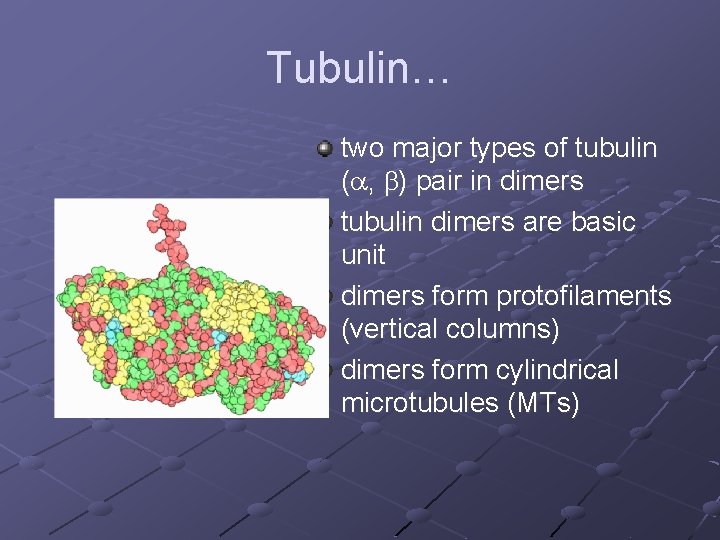
Tubulin… two major types of tubulin (a, b) pair in dimers tubulin dimers are basic unit dimers form protofilaments (vertical columns) dimers form cylindrical microtubules (MTs)

Electric Potential slice through dimer red/blue by potential strength green isopotential lines not well replaced with multipole expansion

Protein Surface Potential surface mix of positive (blue) and negative (red) charge regions isopotential surface in yellow note folded C-terminii
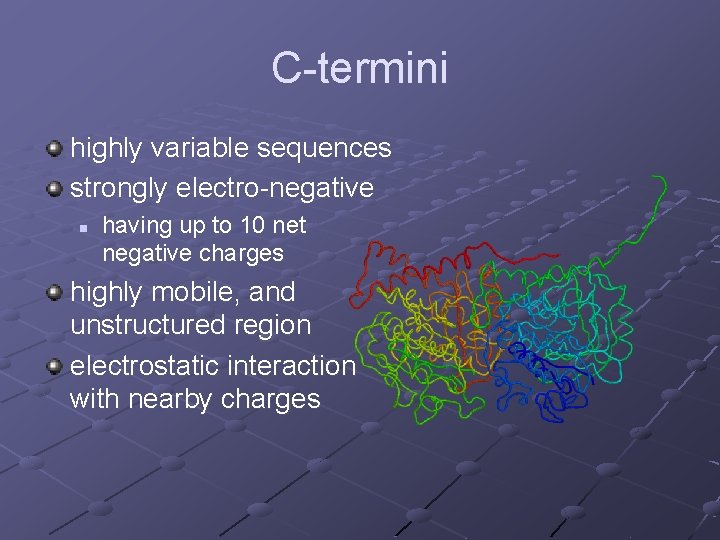
C-termini highly variable sequences strongly electro-negative n having up to 10 net negative charges highly mobile, and unstructured region electrostatic interaction with nearby charges

RMSF & RMSD (work in progress) RMS fluctuations are a measure of the flexibility of a structure. RMS deviations measure the change between two static structures. Created with VMD [Humphrey et al. , 1996]
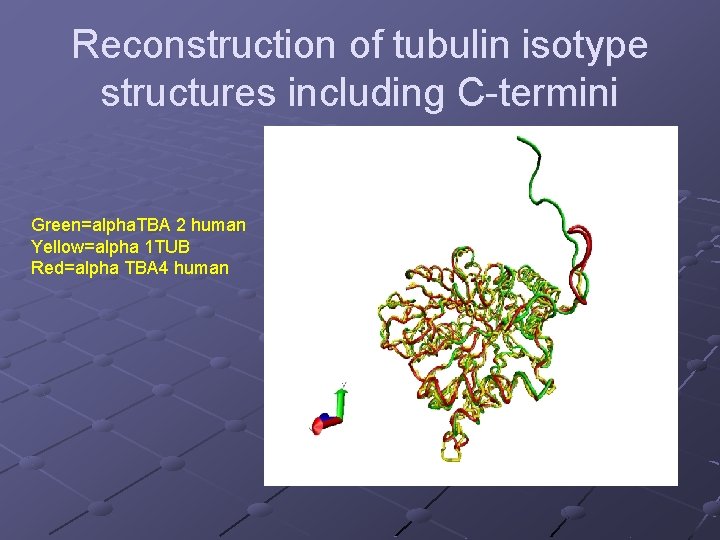
Reconstruction of tubulin isotype structures including C-termini Green=alpha. TBA 2 human Yellow=alpha 1 TUB Red=alpha TBA 4 human

Effects of p. H C-termini are antenna-like
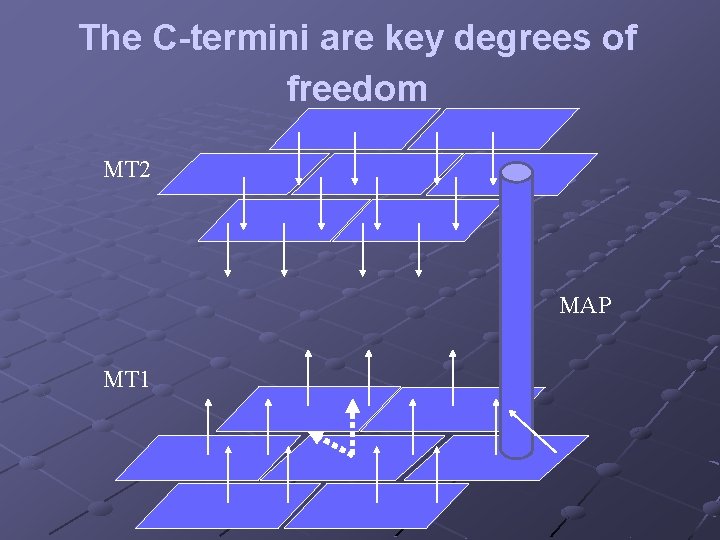
The C-termini are key degrees of freedom MT 2 MAP MT 1

Charge distribution of tubulin dimer including C-termini. Blue=negative Red=positive White=neutral Approx. 25 e per monomer or 50 per dimer
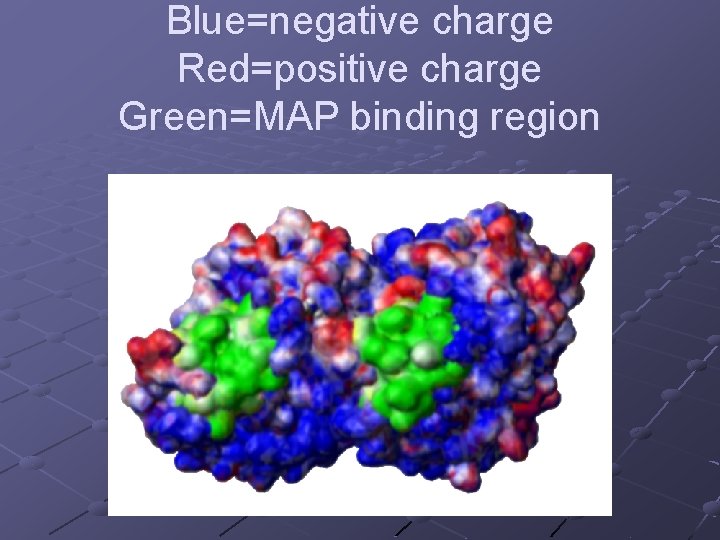
Blue=negative charge Red=positive charge Green=MAP binding region
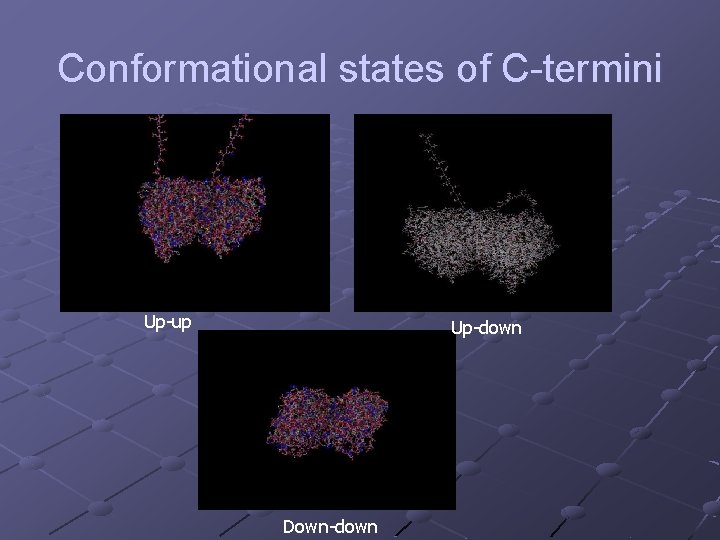
Conformational states of C-termini Up-up Up-down Down-down
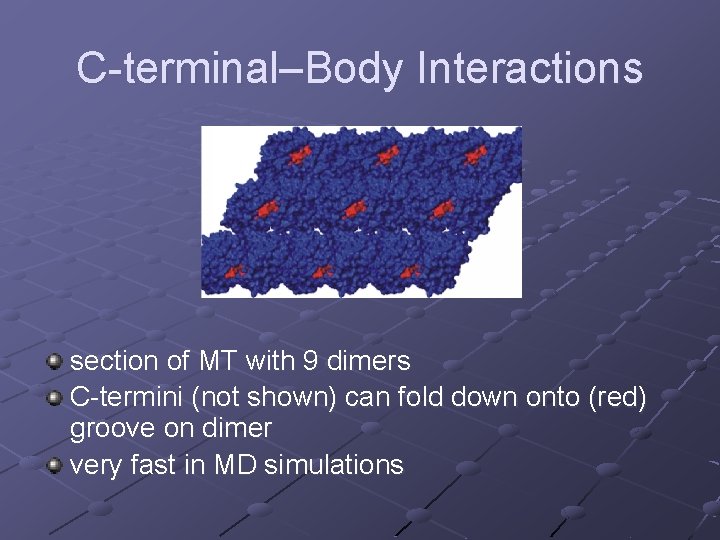
C-terminal–Body Interactions section of MT with 9 dimers C-termini (not shown) can fold down onto (red) groove on dimer very fast in MD simulations
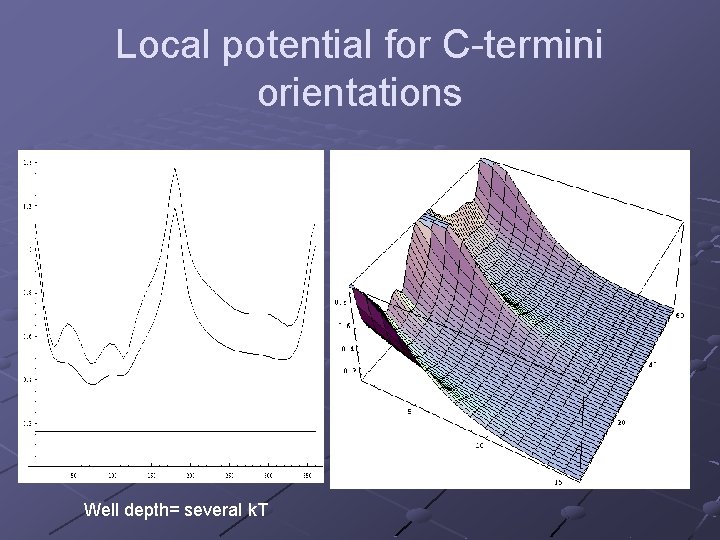
Local potential for C-termini orientations Well depth= several k. T

The proposed model to be developed: The C-termini as the key degrees of freedom Each C- terminus can be in one of several states: n up – extending outward from the surface n down – bound to the surface of a MT Transitions between the states are induced by thermal fluctuations, interaction with neighboring C-termini and interference from adjacent MT’s mediated by the MAP Transitions may induce ionic waves between MT’s and down the dendrite

C-terminal Interactions neighbouring C-termini adjacent protein including n n kinesin MAPs (microtubule associated proteins) adjacent surface of dimers
MD simulation of C-termini conformations

Collective states Condition for an ordered state: z. J<k. T => at T=300 K we get k. T=25 me. V Hence even J=5 me. V with z=6 neighbors (hexatic lattice) leads to an ordered state where pair-wise interactions are below thermal noise This corresponds to f=6 10 12 Hz (MW) Possibly dipole-dipole or H-bond interactions

Ionic Wave Propagation RLC representation of a biopolymer from the view point of counterions Speeds of 1 -100 m/s possible Localized traveling waves Singaling C-termini may collapse
- Slides: 36