The Dissolving Process Solute The substance being dissolved

The Dissolving Process

Solute • The substance being dissolved • The “dissolvee”

Solvent • The substance doing the dissolving • The “dissolver”

Solution • A solute dissolved in a solvent
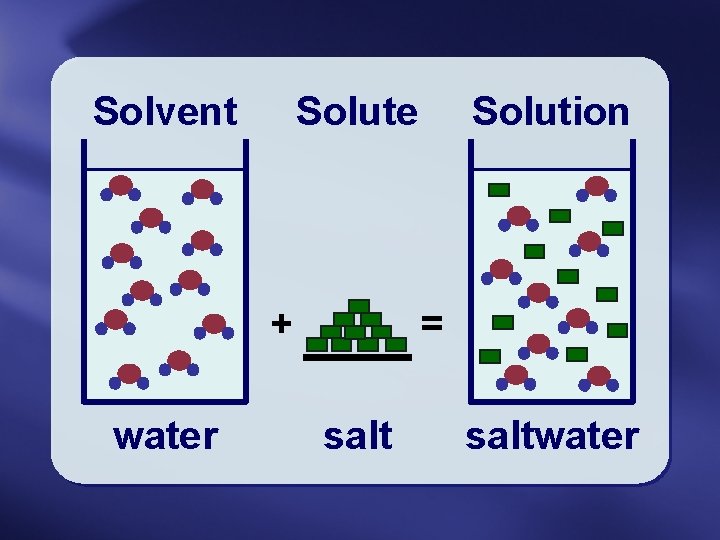
Solvent Solute + water Solution = saltwater

Question When the ____ is dissolved in the ____, a solution results. 1. solid, gas 2. liquid, gas 3. solvent, solute 4. solute, solvent

Types of Solutions • All phases can be the solute. • Solids and liquids can always be the solvent. • Gases can only be the solvent if a gas is the solute.

Types of Solutions Solvents gas Solutes gas does not liquid exist solid gas-in-gas (O 2 in N 2, air) does not exist liquid solid gas-in-liquid gas-in-solid (soft drink) (H 2 in Pd) liquid-inliquid (vinegar) liquid-insolid (amalgam) solid-inliquid (salt water) solid-insolid (alloys)

Question Which type of solution is not possible? 1. Solid in a gas 2. Liquid in a solid 3. Solid in a liquid 4. Gas in a gas 5. Gas in a liquid

The dissolving process consists of 3 aspects: 1. The solute separates. 2. The solvent attaches to the solute. 3. Diffusion moves the groups of particles around.

Solvation is the general term for this process.

Solvation • Hydration is solvation in water. • Dissociation is the solvation of ionic substances. • Ionization is the forming of ions from some covalent compounds such as acids.

Solvation • In any case, breaking solute-solute bonds requires energy—endothermic. • Then, the later formation of solute-solvent bonds releases energy— exothermic.
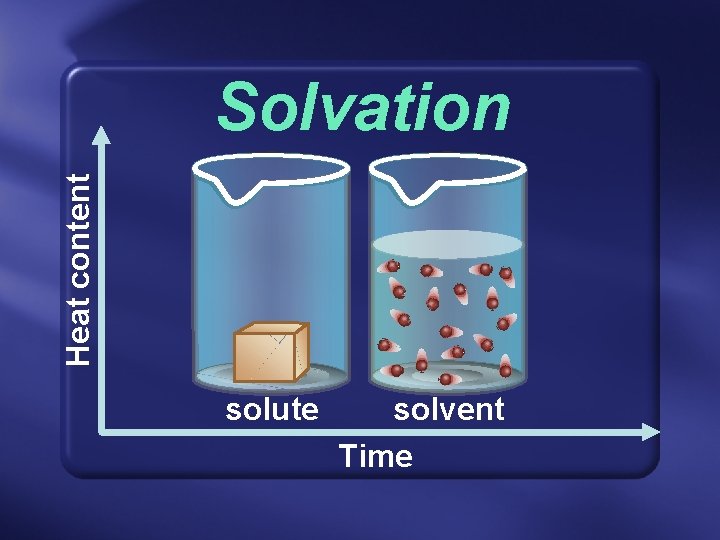
Heat content Solvation solute solvent Time
Heat content Solvation Time
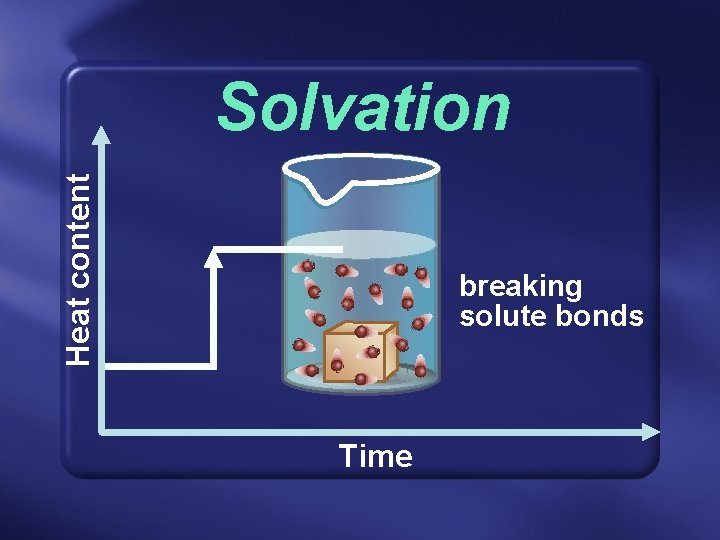
Heat content Solvation breaking solute bonds Time
Heat content Solvation moving solutesolvent away Time
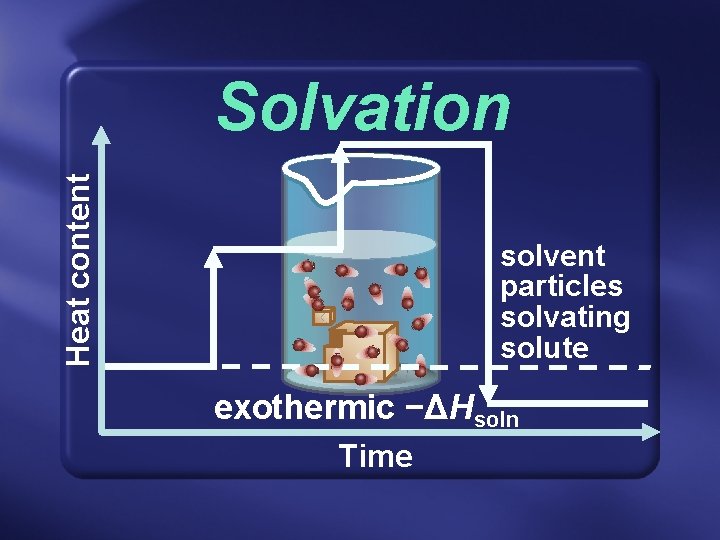
Heat content Solvation solvent particles solvating solute exothermic −ΔHsoln Time

Solvation • Net change in energy § Called the enthalpy (heat) of solution § Symbol: ΔHsoln § Units: kilojoules per mole (k. J/mol)

Solvent Selectivity • The general rule is “like dissolves like. ” § Polar dissolves polar. § Nonpolar dissolves nonpolar.

Question Breaking bonds ____ energy. 1. controls 2. releases 3. necessitates 4. requires 5. dissolves

Question “Like dissolves like” refers to _____. 1. hot and cold 2. strong and weak 3. good and bad 4. big and small 5. polar and nonpolar

To dissolve or not to dissolve … • Solubility is the maximum amount of solute that can be dissolved in an amount of solvent at specific conditions, such as temperature and pressure.

To dissolve or not to dissolve … • If solvation cannot occur, the solute is said to be insoluble in the solvent.

Equilibrium • A balance of two things § Static equilibrium: a balance of 2 forces § Dynamic equilibrium: a balance of 2 processes

Solution Equilibria • Balance exists between dissolving and precipitating. • The solution is saturated; it has the maximum amount of solute dissolved.

Not in Equilibrium • Unsaturated: Less than the maximum amount of solute is dissolved. • Supersaturated: More than the maximum amount of solute is dissolved.

Rate of Solution • Two factors: 1. Inherent solubility 2. Number of effective solute-solvent collisions

Rate of Solution To change the number of collisions: • Change temperature • Stir • Change surface area

Solubility & T • Dissolving liquids or solids increase in T = in solubility • Dissolving gases decrease in T = in solubility
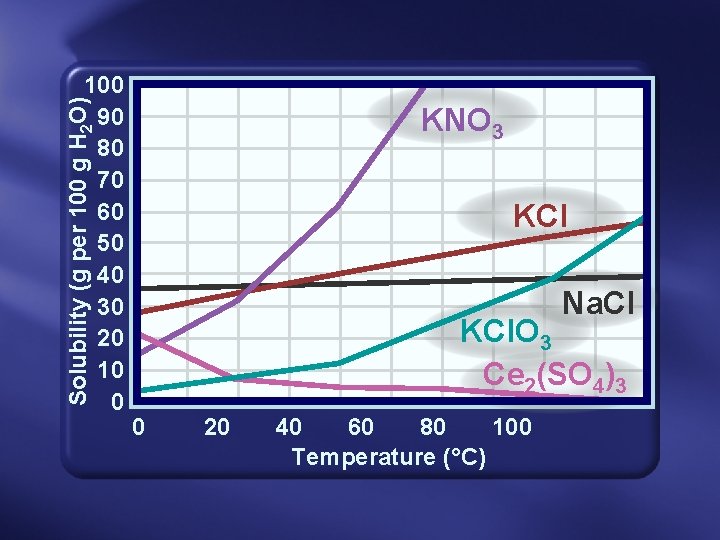
Solubility (g per 100 g H 2 O) 100 90 80 70 60 50 40 30 20 10 0 KNO 3 KCl Na. Cl KCl. O 3 Ce 2(SO 4)3 0 20 40 60 80 100 Temperature (°C)

Solubility & P • Dissolving liquids or solids P has no noticeable effect. • Dissolving gases increase in P = in solubility

Henry’s Law Solubility of gases increase with the partial pressure of the gas above the solution.

Henry’s Law S 1 S 2 = P 1 P 2

Effervescence the rapid escape of a gas from a liquid-gas solution
- Slides: 35