The Diabetic Retinopathy Clinical Research Network Intravitreous AntiVEGF

The Diabetic Retinopathy Clinical Research Network Intravitreous Anti-VEGF vs. Vitrectomy for Vitreous Hemorrhage from PDR (Protocol AB) Protocol Chair: Andrew N. Antoszyk, MD

Background Ø Vitreous hemorrhage due to PDR is a frequent cause of vision loss in patients with diabetes. Ø No recent, definitive evidence to guide how to: § Optimize VA outcomes, or § Optimize time to maximal visual recovery Ø Major question of interest: § Once intervention is desired, what is the relative safety and efficacy of currently available therapies? 2

Anti-VEGF for VH in PDR Rationale Ø Data supporting anti-VEGF use for VH + PDR: • In DRCR. net Protocol N, a difference in rates of vitrectomy at 4 months with anti-VEGF compared with saline-treated eyes was not identified BUT • Ranibizumab treated eyes showed some biological effects suggesting benefits: § Higher probability of completing PRP (44% vs 31% by 16 weeks P = 0. 05) § Greater average VA improvement (22 vs. 16 letters, P = 0. 04) § Reduced rate of recurrent hemorrhage 3 (6% vs. 17%, P = 0. 01)

Vitrectomy for VH in PDR Rationale Ø Newer surgical techniques allow smaller incisions, faster operating times, and fewer associated adverse events Ø DRIVE UK study: Eyes with “simple VH” of at least 3 months duration had better outcomes than eyes with TRD or other diabetic pathology § 85% (51) had >3 line gain in VA § Mean gain of 42 ± 27 ETDRS letters § 8. 3% (5) had repeat vitrectomy • 3 for non-clearing VH; 2 for RD Gupta B, Sivaprasad S, Wong R, et al. Visual and anatomical outcomes following vitrectomy for 4 complications of diabetic retinopathy: the DRIVE UK study. Eye (Lond). 2012 Apr; 26(4): 510 -6.

Study Objectives 1. Evaluate and compare VA outcomes over time for the following regimens: A. Prompt vitrectomy + PRP B. Intravitreous anti-VEGF (Aflibercept) injections 2. Characterize the follow-up course for the two treatment regimens, including: § Post-operative complications for the vitrectomy group § Number of injections needed and percent requiring vitrectomy in the aflibercept group 5
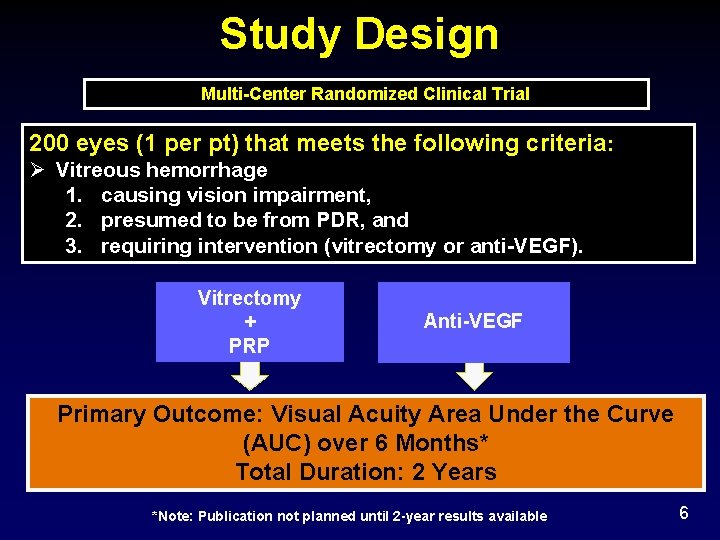
Study Design Multi-Center Randomized Clinical Trial 200 eyes (1 per pt) that meets the following criteria: Ø Vitreous hemorrhage 1. causing vision impairment, 2. presumed to be from PDR, and 3. requiring intervention (vitrectomy or anti-VEGF). Vitrectomy + PRP Anti-VEGF Primary Outcome: Visual Acuity Area Under the Curve (AUC) over 6 Months* Total Duration: 2 Years *Note: Publication not planned until 2 -year results available 6

Additional Key Outcomes ØAlthough 6 M VA AUC is “primary”, longterm outcomes are also important: • Mean VA at 1, 3 months and annual visits • VA AUC at annual visits • Percent 20/20 and 20/40 or better and 20/200 or worse at annual visits • Rates of recurrent VH on clinical exam • Productivity from WPAI questionnaire • Treatment and follow-up costs 7

Major Inclusion/Exclusion Criteria Ø VA 20/32 or worse but at least light perception Ø No anti-VEGF treatment within 2 months prior to vitreous hemorrhage onset Ø No prior vitrectomy • Note: Prior PRP is not a requirement nor an exclusion Ø No rhegmatogenous retinal detachment and no traction retinal detachment involving or threatening the fovea Ø Patient is able and willing to undergo vitrectomy within 2 weeks (no contraindication to surgery) 8

Follow-Up and Treatment Overview Ø Outcome assessment visits for both groups • Every 3 months in the first year • Every 4 months in the second year Ø Anti-VEGF Treatment • Visits every 4 weeks for 6 months and then as needed • Injections required in the Anti-VEGF Group through week 12 and then as needed according to criteria • Both groups may receive anti-VEGF for DME Ø Vitrectomy • Within 2 weeks of randomization in the Vitrectomy Group • If needed in anti-VEGF group according to criteria 9

Referrals We Need Your Help! Ø Please consider any eyes with vitreous hemorrhage due to diabetic retinopathy causing vision impairment Ø Study participants must be willing to be randomized to vitrectomy or aflibercept injections and continue follow-up for 2 years 10

Thank You on Behalf of Diabetic Retinopathy Clinical Research Network (DRCR. net) Dedicated to multicenter clinical research of diabetic retinopathy, macular edema and associated disorders. 11
- Slides: 11