The Diabetic Retinopathy Clinical Research Network Exploratory Analysis

The Diabetic Retinopathy Clinical Research Network Exploratory Analysis of Diabetic Retinopathy Progression through 3 Years in a Randomized Clinical Trial Comparing Intravitreal Triamcinolone with Focal/Grid Photocoagulation Sponsored by the National Eye Institute, National Institutes of Health, U. S. Department of Health and Human Services 1

Background Ø Current treatments for reducing the risk of progression of retinopathy: • Glycemic control • PRP – associated with side effects and potential complications Ø Identification of other treatments is desirable Ø Rationale for corticosteroids: • Down regulates VEGF and reduces breakdown of blood-retinal barrier • Anti-angiogenic properties • Intravitreal triamcinolone shown to prevent retinal neovascularization in animal and clinical studies 2

DRCR. net Protocol B: Laser vs. Intravitreal Triamcinolone for DME Ø 840 eyes (693 subjects) at 88 sites • Laser group: N = 330 • 1 mg IVT group: N = 256 • 4 mg IVT group: N = 254 Ø Major Eligibility Criteria: • VA 20/40 to 20/320 • CSF >= 250 microns on OCT Ø Visits (and retreatment assessment) every 4 months through 3 years Ø Primary Outcome: VA at 2 years 3

Primary Results Ø Focal/Grid Laser was more effective (VA and CSF) with fewer side effects (IOP and cataract) than 1 mg or 4 mg IVT at 2 years Ø Longer term 3 year results were consistent with 2 year results Ø Change in retinopathy level was a planned secondary outcome • An exploratory analysis was pursued to evaluate effect of IVT on a multilevel definition of 4 progression of retinopathy

Outcome Definition Ø Progression of retinopathy up to 3 years (any of the following): • Progressed from NPDR at baseline to PDR during follow-up* • Received PRP between baseline and followup • Reported a vitreous hemorrhage between baseline and follow-up • Worsened 2 or more levels on the ETDRS diabetic retinopathy scale* *Based on Reading Center grading of annual fundus photos 5
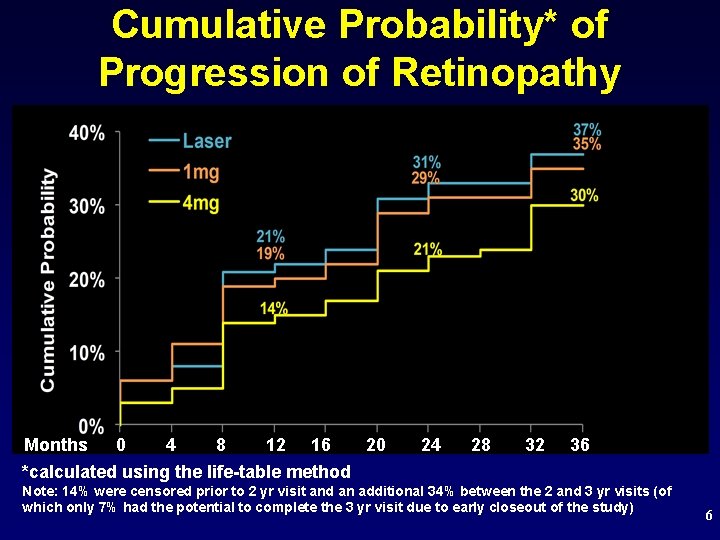
Cumulative Probability* of Progression of Retinopathy Months 0 4 8 12 16 20 *calculated using the life-table method 24 28 32 36 Note: 14% were censored prior to 2 yr visit and an additional 34% between the 2 and 3 yr visits (of which only 7% had the potential to complete the 3 yr visit due to early closeout of the study) 6
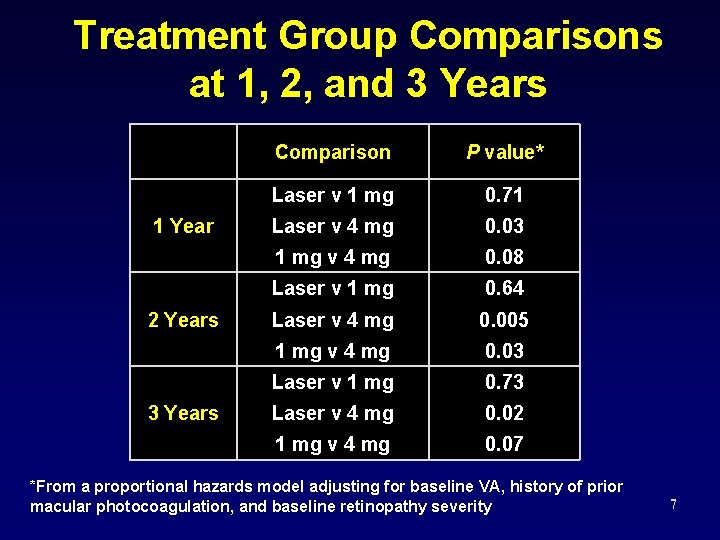
Treatment Group Comparisons at 1, 2, and 3 Years 1 Year 2 Years 3 Years Comparison P value* Laser v 1 mg 0. 71 Laser v 4 mg 0. 03 1 mg v 4 mg 0. 08 Laser v 1 mg 0. 64 Laser v 4 mg 0. 005 1 mg v 4 mg 0. 03 Laser v 1 mg 0. 73 Laser v 4 mg 0. 02 1 mg v 4 mg 0. 07 *From a proportional hazards model adjusting for baseline VA, history of prior macular photocoagulation, and baseline retinopathy severity 7

Cumulative Probability* of Progression of Retinopathy up to 2 Years Additional Probability (not counted in prior row) Laser N=330 1 mg N=256 4 mg N=254 NPDR to PDR 15% 11% 8% PRP 9% 6% 5% Vitreous hemorrhage 6% 9% 5% Worsened 2 levels 1% 3% 3% Combined definition 31% 29% 21% *calculated using the life-table method 8

Cumulative Probability* of Progression of Retinopathy up to 2 Years Total Probability for Each Criteria Laser N=330 1 mg N=256 4 mg N=254 NPDR to PDR 15% 11% 8% PRP 14% 10% 9% Vitreous hemorrhage 16% 11% Worsened 2 levels 15% 11% Combined definition 31% 29% 21% *calculated using the life-table method 9

Cumulative Probability* of Progression of Retinopathy up to 3 Years Additional Probability (not counted in prior row) Laser N=330 1 mg N=256 4 mg N=254 NPDR to PDR 19% 14% 16% PRP 10% 7% 6% Vitreous hemorrhage 6% 12% 5% Worsened 2 levels 2% 2% 3% Combined definition 37% 35% 30% *calculated using the life-table method 10

Cumulative Probability* of Progression of Retinopathy up to 3 Years Total Probability for Each Criteria Laser N=330 1 mg N=256 4 mg N=254 NPDR to PDR 19% 14% 16% PRP 18% 11% 12% Vitreous hemorrhage 19% 20% 15% Worsened 2 levels 17% 15% 17% Combined definition 37% 35% 30% *calculated using the life-table method 11

Discussion Ø 4 mg IVT can reduce the risk of progression of retinopathy at 1, 2, and 3 years Ø Difference cannot be explained by an increase in retinopathy caused by focal/grid laser • Similar risk in laser group and 1 mg group • Similar risk of development of PDR in ETDRS immediate laser group (11. 1%) and deferred laser group (10. 8%) Ø Results in Fluocinolone Acetonide Implant Study Group trial support DRCRnet results • Lower rate of retinopathy worsening compared to standard of care group (P<0. 002, P=0. 012, and 12 P<0. 015 at 1, 2, and 3 years, respectively)

Conclusions Ø Use of IVT to reduce risk of progression of retinopathy is not warranted at this time • IVT is associated with cataract and elevated IOP • PDR can be treated relatively safely with PRP Ø Further investigation of treatments to reduce risk of progression of retinopathy safely is needed 13
- Slides: 13