The Diabetic Retinopathy Clinical Research Network Expanded 2

The Diabetic Retinopathy Clinical Research Network Expanded 2 -year Follow-up of Ranibizumab Plus Prompt or Deferred Laser or Triamcinolone Plus Prompt Laser for Diabetic Macular Edema Sponsored by the National Eye Institute, National Institutes of Health, U. S. Department of Health and Human Services 1

Changes to Protocol I To Initiate After Primary Results Published Ø Study extended to 5 years from randomization Ø All study participants given opportunity to receive ranibizumab injections if edema present or develops Ø Triamcinolone+prompt laser (Group D) participants can continue to receive triamcinolone injections instead of ranibizumab at investigator discretion 2

Availability of 2 -Year Data Ø Completed prior to implementation of protocol change: N=642 (75%) Ø Unavailable: N=212 (25%) • • • death (35) withdrawals from the study (47) lost to follow (28) unavailable for the 2 -year visit (3) completed after the protocol change implemented (99) 3

Treatments 4
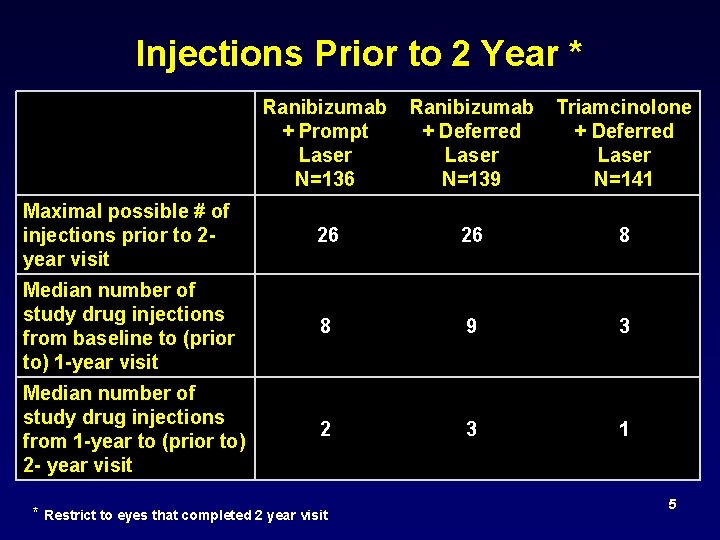
Injections Prior to 2 Year * Ranibizumab + Prompt Laser N=136 Ranibizumab Triamcinolone + Deferred Laser N=139 N=141 Maximal possible # of injections prior to 2 - year visit 26 26 8 Median number of study drug injections from baseline to (prior to) 1 -year visit 8 9 3 Median number of study drug injections from 1 -year to (prior to) 2 - year visit 2 3 1 * Restrict to eyes that completed 2 year visit 5
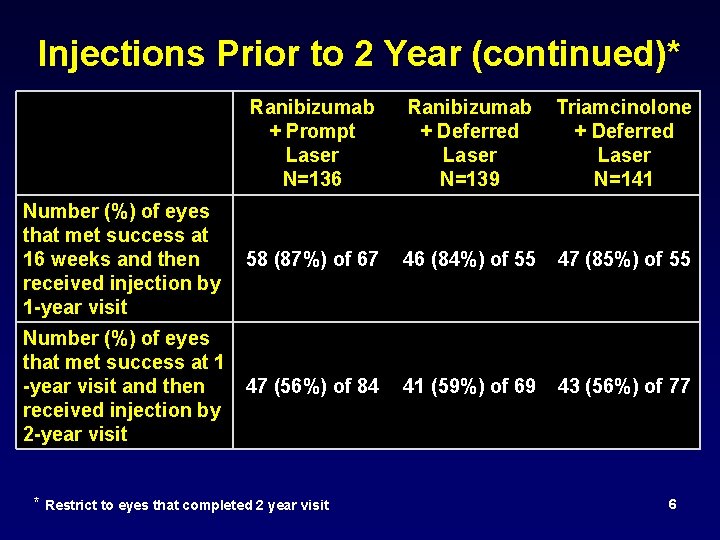
Injections Prior to 2 Year (continued)* Ranibizumab + Prompt Laser N=136 Ranibizumab Triamcinolone + Deferred Laser N=139 N=141 Number (%) of eyes that met success at 16 weeks and then 58 (87%) of 67 received injection by 1 -year visit 46 (84%) of 55 47 (85%) of 55 Number (%) of eyes that met success at 1 -year visit and then 47 (56%) of 84 received injection by 2 -year visit 41 (59%) of 69 43 (56%) of 77 * Restrict to eyes that completed 2 year visit 6
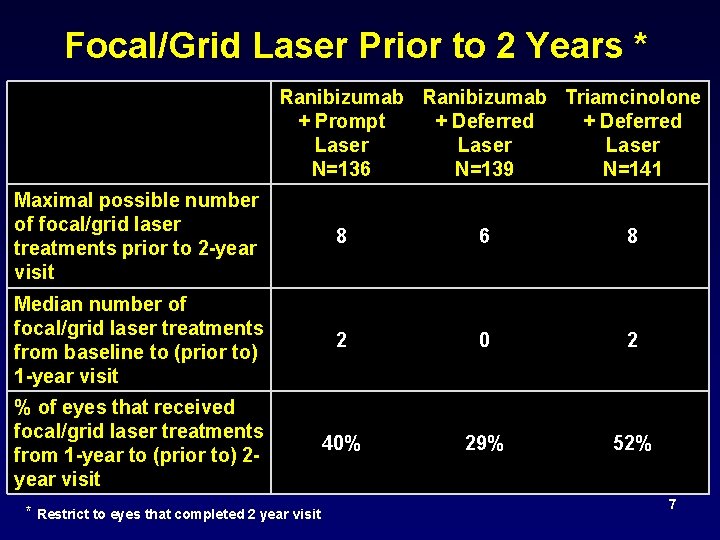
Focal/Grid Laser Prior to 2 Years * Ranibizumab Triamcinolone + Prompt + Deferred Laser N=136 N=139 N=141 Maximal possible number of focal/grid laser treatments prior to 2 -year visit 8 6 8 Median number of focal/grid laser treatments from baseline to (prior to) 1 -year visit 2 0 2 % of eyes that received focal/grid laser treatments from 1 -year to (prior to) 2 year visit 40% 29% 52% * Restrict to eyes that completed 2 year visit 7

Visual Acuity (includes all data up to and including the first visit when all investigators are unmasked to primary outcome results) 8
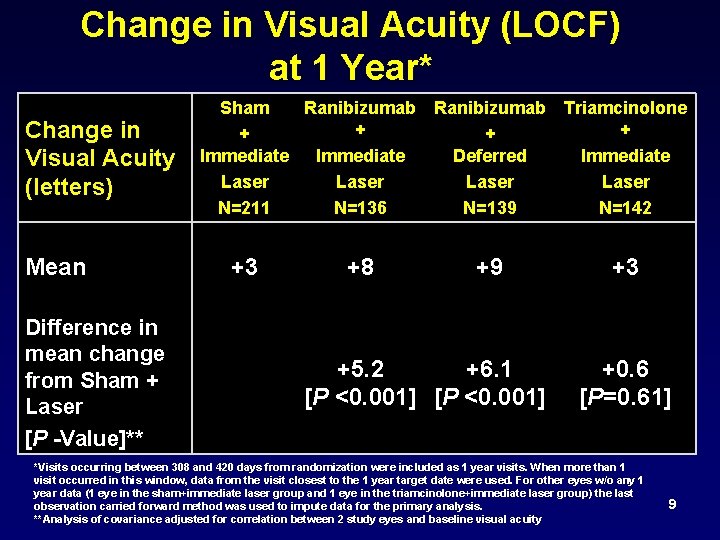
Change in Visual Acuity (LOCF) at 1 Year* Change in Visual Acuity (letters) Mean Difference in mean change from Sham + Laser [P -Value]** Sham Ranibizumab Triamcinolone + + Immediate Deferred Immediate Laser N=211 N=136 N=139 N=142 +3 +8 +9 +5. 2 +6. 1 [P <0. 001] +3 +0. 6 [P=0. 61] *Visits occurring between 308 and 420 days from randomization were included as 1 year visits. When more than 1 visit occurred in this window, data from the visit closest to the 1 year target date were used. For other eyes w/o any 1 year data (1 eye in the sham+immediate laser group and 1 eye in the triamcinolone+immediate laser group) the last observation carried forward method was used to impute data for the primary analysis. **Analysis of covariance adjusted for correlation between 2 study eyes and baseline visual acuity 9
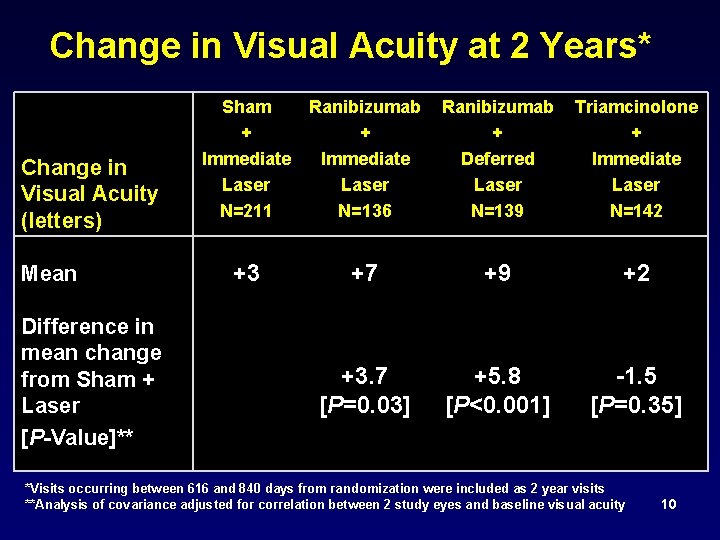
Change in Visual Acuity at 2 Years* Change in Visual Acuity (letters) Mean Difference in mean change from Sham + Laser [P-Value]** Sham + Immediate Laser N=211 Ranibizumab + Immediate Laser N=136 Ranibizumab Triamcinolone + + Deferred Immediate Laser N=139 N=142 +3 +7 +9 +2 +3. 7 [P=0. 03] +5. 8 [P<0. 001] -1. 5 [P=0. 35] *Visits occurring between 616 and 840 days from randomization were included as 2 year visits **Analysis of covariance adjusted for correlation between 2 study eyes and baseline visual acuity 10

Mean Change in Visual Acuity* at Follow-up Visits N = 626 (52 weeks) N = 600 (68 weeks) N = 600 (84 weeks) N = 628 (104 weeks) *Truncated to ± 30 letters P-values for difference in mean change in VA from sham+prompt laser at the 104 week visit: ranibizumab+prompt laser =0. 03; ranibizumab+deferred laser <0. 001; and triamcinolone+prompt laser=0. 35. 11
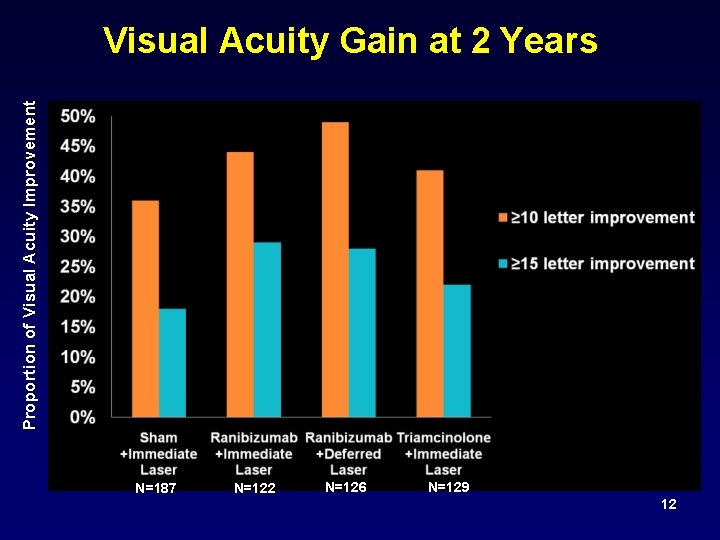
Proportion of Visual Acuity Improvement Visual Acuity Gain at 2 Years N=187 N=122 N=126 N=129 12
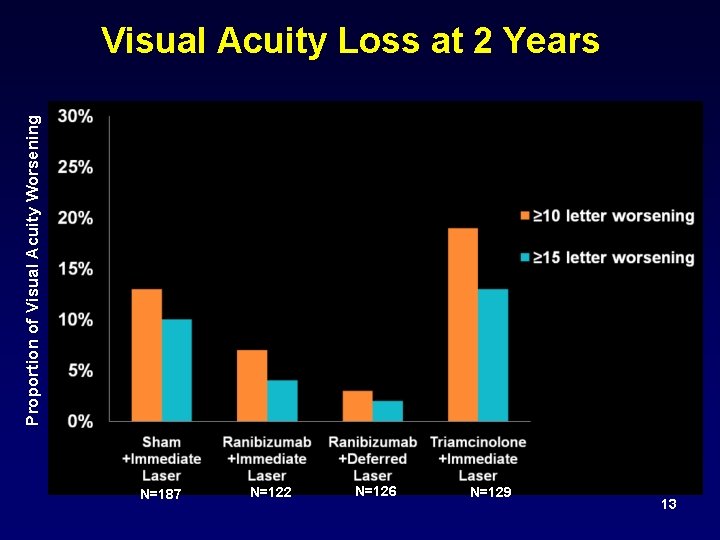
Proportion of Visual Acuity Worsening Visual Acuity Loss at 2 Years N=187 N=122 N=126 N=129 13
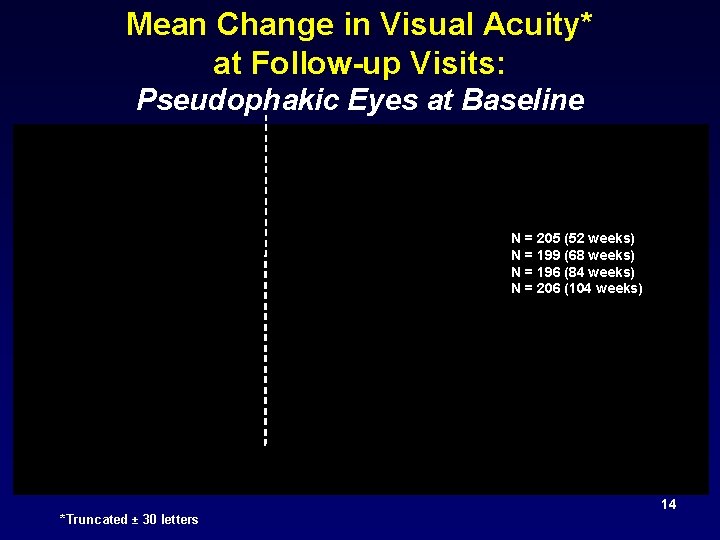
Mean Change in Visual Acuity* at Follow-up Visits: Pseudophakic Eyes at Baseline N = 205 (52 weeks) N = 199 (68 weeks) N = 196 (84 weeks) N = 206 (104 weeks) 14 *Truncated ± 30 letters

Retinal Thickening (includes all data up to and including the first visit when all investigators are unmasked to primary outcome results) 15
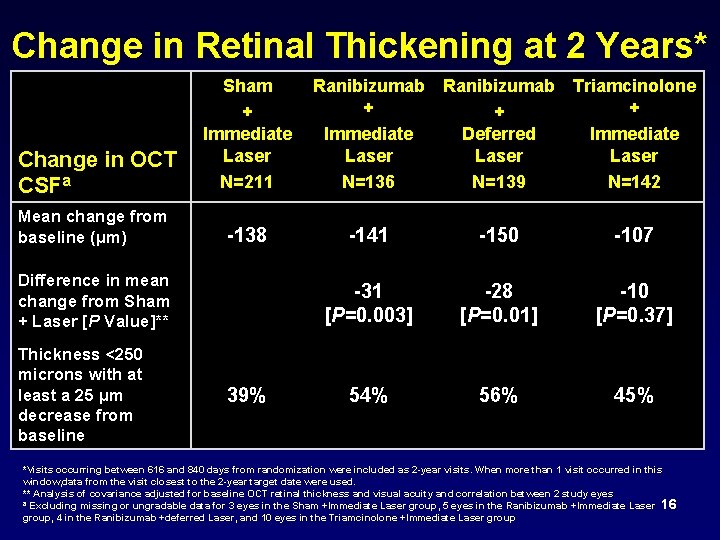
Change in Retinal Thickening at 2 Years* Change in OCT CSFa Sham + Immediate Laser N=211 Mean change from baseline (µm) -138 Difference in mean change from Sham + Laser [P Value]** Thickness <250 microns with at least a 25 µm decrease from baseline 39% Ranibizumab Triamcinolone + + + Immediate Deferred Immediate Laser N=136 N=139 N=142 -141 -150 -107 -31 [P=0. 003] -28 [P=0. 01] -10 [P=0. 37] 54% 56% 45% *Visits occurring between 616 and 840 days from randomization were included as 2 -year visits. When more than 1 visit occurred in this window, data from the visit closest to the 2 -year target date were used. ** Analysis of covariance adjusted for baseline OCT retinal thickness and visual acuity and correlation between 2 study eyes ª Excluding missing or ungradable data for 3 eyes in the Sham +Immediate Laser group, 5 eyes in the Ranibizumab +Immediate Laser 16 group, 4 in the Ranibizumab +deferred Laser, and 10 eyes in the Triamcinolone +Immediate Laser group

Mean Change in CSF Thickening at Follow-up Visits N = 617 (52 weeks) N = 581 (68 weeks) N = 568 (84 weeks) N = 606 (104 weeks) 17 *P values are for the difference in mean change in OCT CSF retinal thickness from Sham + Immediate Laser at the 52 week visit: ranibizumab+prompt laser =0. 003, ranibizumab+deferred laser =0. 01, and triamcinolone+prompt laser =0. 37.
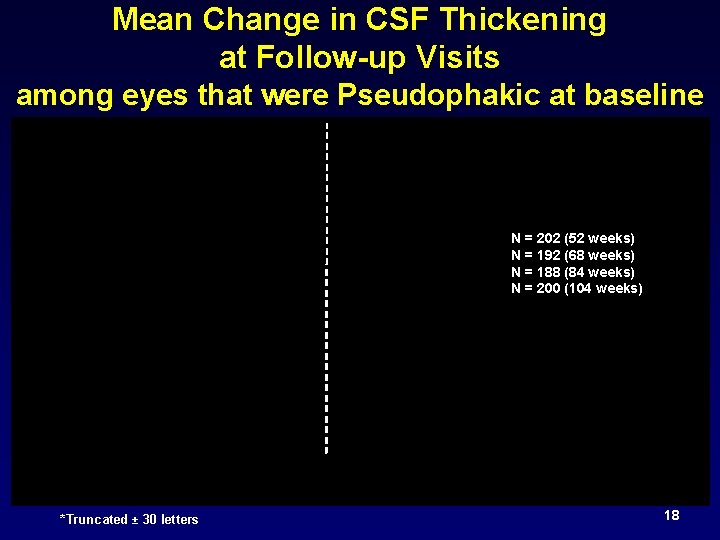
Mean Change in CSF Thickening at Follow-up Visits among eyes that were Pseudophakic at baseline N = 202 (52 weeks) N = 192 (68 weeks) N = 188 (84 weeks) N = 200 (104 weeks) *Truncated ± 30 letters 18

Safety (all available data) 19

Cardiovascular Events According to Antiplatelet Trialists’ Collaboration Shama Ranibizumab Triamcinoloneb N*=130 N*=375 N*=186 Non-fatal myocardial infarction 3% 1% 3% Non-fatal cerebrovascular accident-ischemic or hemorrhagic (or unknown) 6% 2% 2% Vascular death (from any potential vascular or unknown cause) 6% 3% 4% Any ATC cardiovascular event 13% 7% 8% * N=Number of Study Participants. Study participants with 2 study eyes are assigned to the nonsham group. Multiple events within a study participant are only counted once per event. aone participant began receiving ranibizumab prior to nonfatal stroke bone participant began receiving ranibizumab prior to nonfatal stroke 20
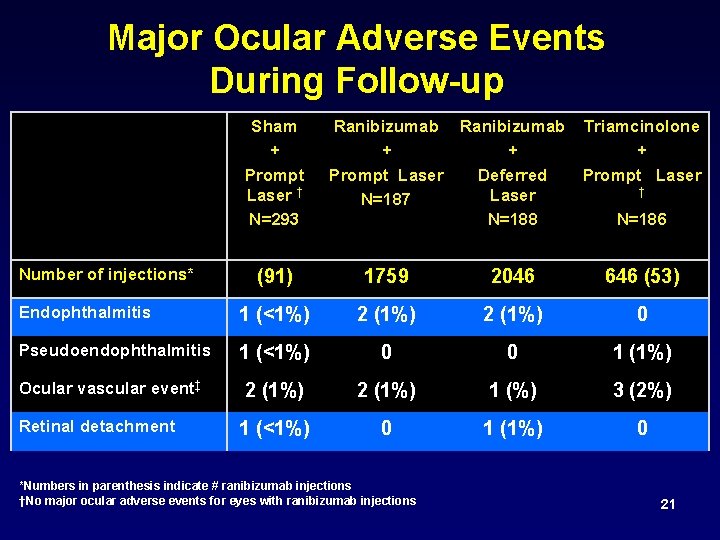
Major Ocular Adverse Events During Follow-up Sham + Prompt Laser † N=293 Number of injections* Ranibizumab Triamcinolone + + + Prompt Laser Deferred Prompt Laser † Laser N=187 N=188 N=186 (91) 1759 2046 646 (53) Endophthalmitis 1 (<1%) 2 (1%) 0 Pseudoendophthalmitis 1 (<1%) 0 0 1 (1%) Ocular vascular event‡ 2 (1%) 1 (%) 3 (2%) 1 (<1%) 0 1 (1%) 0 Retinal detachment *Numbers in parenthesis indicate # ranibizumab injections †No major ocular adverse events for eyes with ranibizumab injections 21
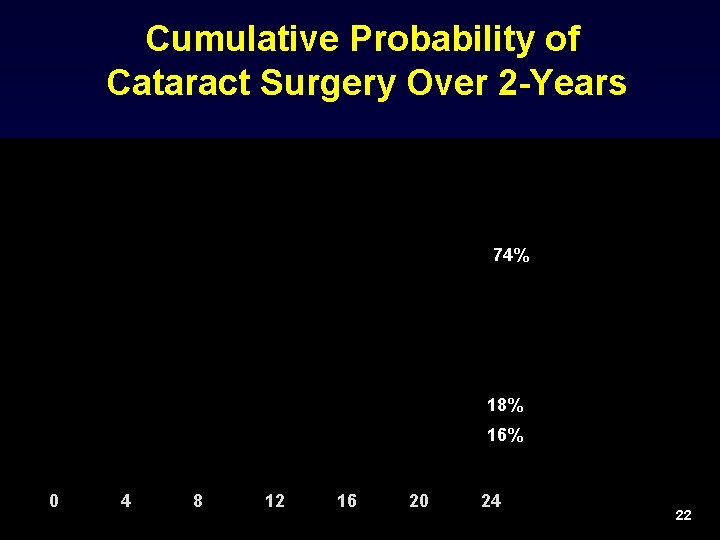
Cumulative Probability of Cataract Surgery Over 2 -Years 74% 18% 16% 0 4 8 12 16 20 24 22

Summary Ø Expanded 2 -year results are similar to results published previously and reinforce conclusions with respect to: • • • Visual acuity outcomes OCT outcomes Pseudophakic subgroup analyses Injections given Lasers given Safety results 23
- Slides: 23