The Diabetes Download Diabetes Update 2017 KIM NEFF

The Diabetes Download: Diabetes Update 2017 KIM NEFF, PHARMD, PHC CLINICAL PHARMACY SPECIALIST, AMBULATORY CARE NEW MEXICO VA HEALTHCARE SYSTEM

Objectives Pharmacist Objectives: Explain the 2016 FDA safety alerts and labeling changes for diabetes medications Counsel patients on diabetic medications incorporating the information from the FDA safety alerts Describe the 2017 updates to the American Diabetes Association standards of care in diabetes Technician Objectives Identify medications affected by recent FDA safety alerts Describe the 2016 safety alerts and labeling changes for diabetes medications Describe the 2017 updates to the American Diabetes Association standards of care in diabetes

https: //www. pinterest. com/kascott 29/rx/

Safety Studies for Diabetes Medications FDA change in 2008 regarding expectation of drug studies for new diabetic medications to ensure there is no unacceptable cardiovascular risk 1 Prompted by 1 -2 Recommendations of the 2008 Endocrinologic and Metabolic Drugs Advisory Committee High cardiovascular risk in patients with diabetes Safety issues with Rosiglitazone Muraglitizar Intense glucose lowering in the ACCORD trial

Safety Studies for Diabetes Medications FDA expectations 1 Independent cardiovascular endpoints committee Evaluation of Cardiovascular mortality Stroke Myocardial infarction Design studies so can be easily and clearly analyzed via a meta-analysis at study completion Should include patients with high cardiovascular risk Elderly Renal impairment Advanced cardiovascular disease Pre and post-marketing trials may be required for new drugs depending upon estimated risk of pre-marketing studies
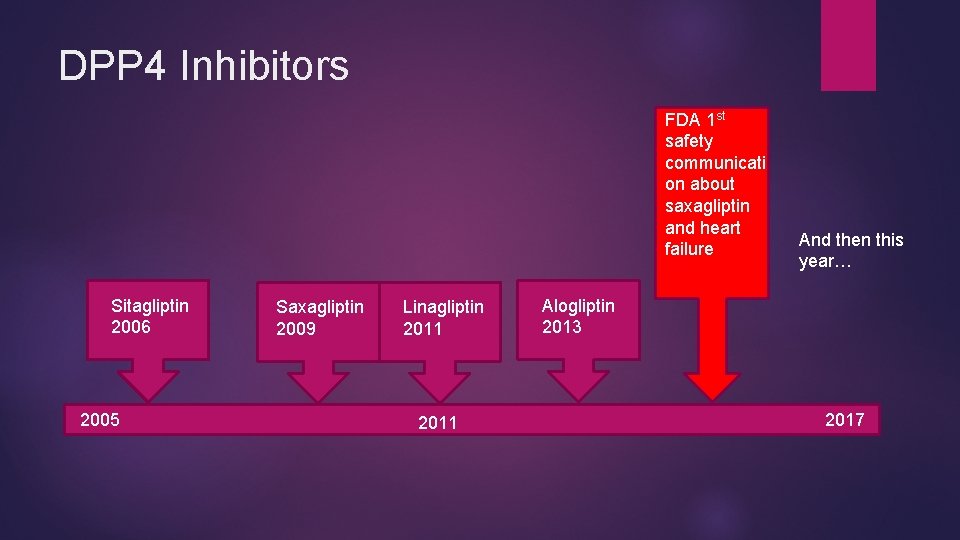
DPP 4 Inhibitors FDA 1 st safety communicati on about saxagliptin and heart failure Sitagliptin 2006 2005 Saxagliptin 2009 Linagliptin 2011 And then this year… Alogliptin 2013 2017

DPP 4 Inhibitors and Heart failure 4/5/2016 “FDA Drug Safety communication: FDA adds warnings about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin” 3 A tale of three studies…
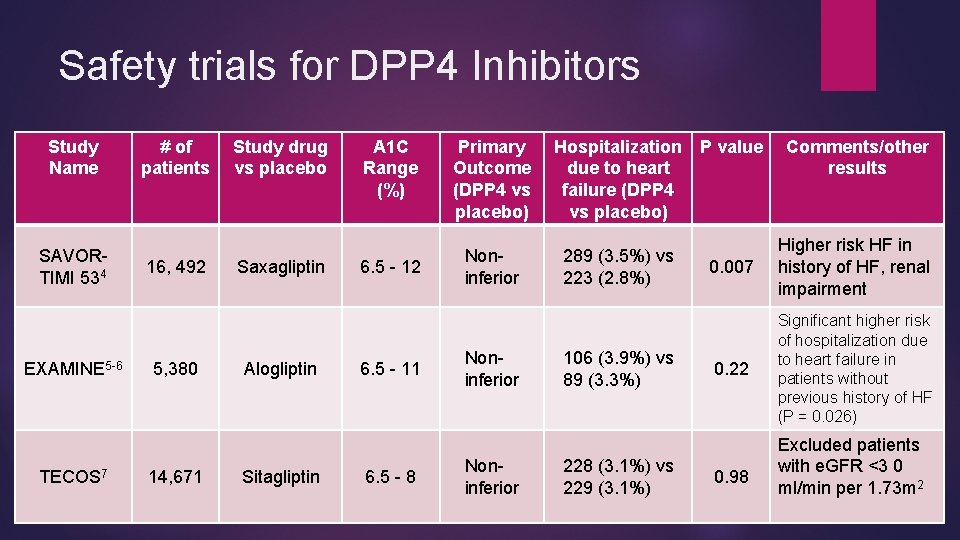
Safety trials for DPP 4 Inhibitors Study Name SAVORTIMI 534 EXAMINE 5 -6 TECOS 7 # of patients 16, 492 5, 380 14, 671 Study drug vs placebo Saxagliptin Alogliptin Sitagliptin A 1 C Range (%) 6. 5 - 12 6. 5 - 11 6. 5 - 8 Primary Outcome (DPP 4 vs placebo) Noninferior Hospitalization P value due to heart failure (DPP 4 vs placebo) 289 (3. 5%) vs 223 (2. 8%) 106 (3. 9%) vs 89 (3. 3%) 228 (3. 1%) vs 229 (3. 1%) Comments/other results 0. 007 Higher risk HF in history of HF, renal impairment 0. 22 Significant higher risk of hospitalization due to heart failure in patients without previous history of HF (P = 0. 026) 0. 98 Excluded patients with e. GFR <3 0 ml/min per 1. 73 m 2

DPP 4 Inhibitors and Heart failure: Retrospective Study 8 Evaluated new users of Saxagliptin Sitagliptin Second generation sulfonylureas Pioglitazone Long acting insulin Follow up time <1 year No overall increase in incidence of hospitalization due to HF in new users of saxagliptin and sitagliptin compared to pioglitazone, sulfonylureas, or long acting insulin
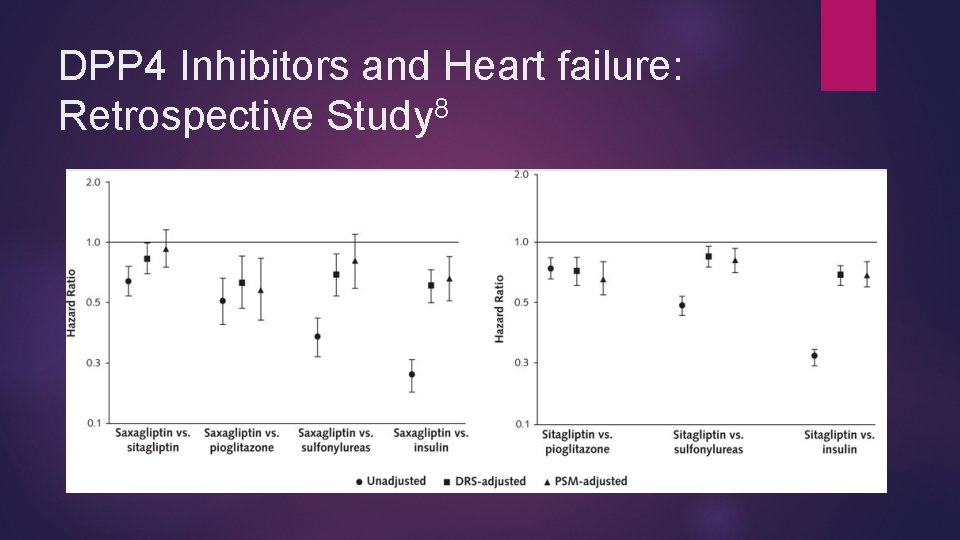
DPP 4 Inhibitors and Heart failure: Retrospective Study 8

DPP 4 Inhibitors and Heart failure: Retrospective Study 8 Recommend cautious interpretation Retrospective study design Short follow up time Re-enforces lack of association of sitagliptin with heart failure BUT, 2/3 cohort studies found a positive association of sitagliptin with heart failure 9 -11 And…

Results from the FDA Adverse Event Reporting System (FAERS)12

Results from the FDA Adverse Event Reporting System (FAERS)12

What About Linagliptin? Two large trials ongoing, results not out yet CARMELINA– cardiovascular CAROLINA—cardiovascular and renal outcomes Pooled study of 19 study results and ADEs 13 Increased heart failure incidence but not clear if significant Must interpret w/caution since pooled data

DPP 4 Inhibitors and Heart Failure: Clinical Considerations Conflicting evidence regarding HF risk Avoid DPP 4 I if has HF and other treatment options available If to use DPP 4 I, sitagliptin likely safest option Careful w/renal impairment Still may have some HF risk May consider alternate option even if any risk factors for HF Do not recommend with very poor blood sugar control Low level of A 1 C lowering Possibility that poorer control may be more associated with higher HF risk

Counseling on DPP 4 Inhibitors Possibility of heart failure Patients need to contact their provider right away if: Difficulty breathing w/laying down Edema/swelling in ankles, feet, legs, stomach Unusual SOB Dyspnea on exertion If on a renally adjusted dose, HF education may be more important Other possible ADEs to educate on Arthralgias Pancreatitis hypoglycemia

The New Kids on the Block http: //images 4. fanpop. com/image/photos/22100000/nkotb-new-kids-on-the-block-22146585 -1152 -1156. jpg http: //www. mims. co. uk/sglt 2 -inhibitors-risk-diabetic-ketoacidosis/diabetes/article/1353980

Empagliflozin and CV Protection 12/2/2016 Jardiance® (Empagliflozin) gains new FDA approval 14 To reduce cardiac death in patients with type 2 DM Approval based on EMPA-REG study 15 Significant improvement in various cardiovascular endpoints Composite cardiovascular outcome Death from cardiovascular causes Non-fatal MI* Non-fatal stroke* Death from any cause Hospitalization from heart failure * defines not independently significant difference
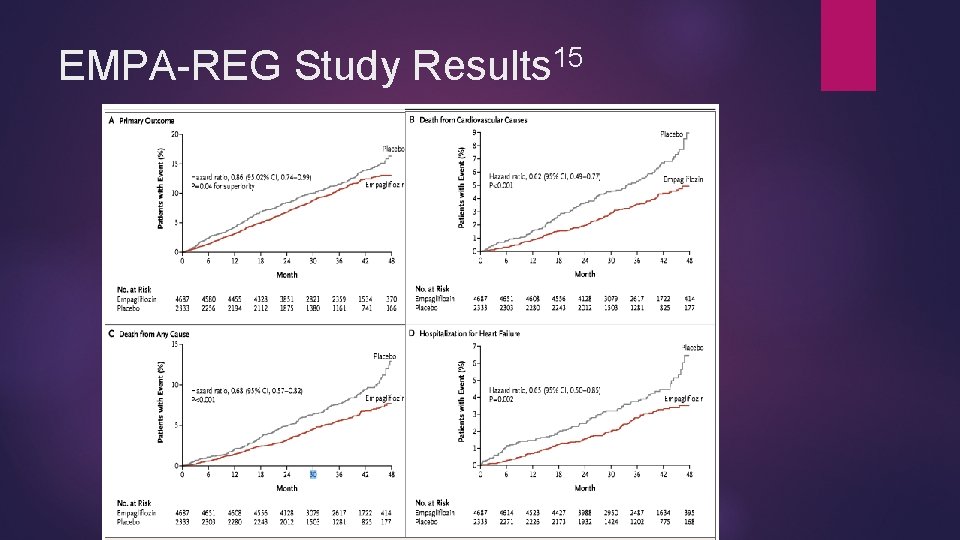
15 EMPA-REG Study Results
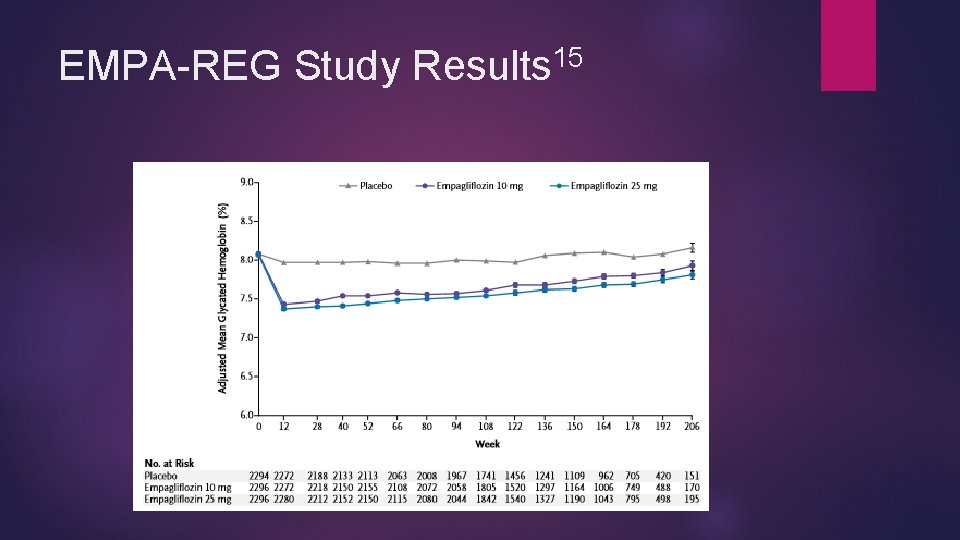
15 EMPA-REG Study Results

15 Empagliflozin and CV Protection CV benefit thought to be due to mechanisms other than just A 1 C lowering Changes in arterial stiffness, cardiac function and cardiac oxygen demand Cardiorenal effects Reduction in Albuminuria uric acid Weight visceral adiposity Blood pressure Improved blood sugar

15 Empagliflozin and CV Protection Significant safety outcomes Less than placebo UTI in women than placebo Acute renal failure Acute kidney injury ADEs leading to study discontinuation More than placebo Genital infections in both men and women Non-significant but more urosepsis

Empagliflozin For Everyone? https: //koshersamurai. files. wordpress. com/2012/12/monopoly-man-rich-guy. jpg http: //www. multivu. com/players/English/7617351 -bi-lilly-empa-reg-cv-results/

Moving Forward with Empagliflozin CV benefit impressive Prevents death in 1 in 45 patients over 3 years 16 Prevents overall mortality in 1 in 39 patients 16 Points to keep in mind Benefit was shown in patients with cardiovascular disease In addition to optimal treatment Patients without cardiovascular disease were not a part of the study No significant reduction in stroke or heart attack Empagliflozin isn’t cheap Although less than other SGLT 2 s, empagliflozin has side effects

Canagliflozin and Amputations 5/18/2016 FDA warns of increase risk of leg and foot amputations, mostly affecting the toes, with the diabetes medicine canaglfilozin 17 Canagliflozin Cardiovascular Assessment Study (CANVAS) Investigating cardiovascular safety Interim analysis showed higher risk of amputation (particularly toe) in canagliflozin than placebo 17 7/1000 with canagliflozin 100 mg daily 5/1000 with canagliflozin 300 mg daily 3/1000 with placebo daily Study was permitted to continue

Canagliflozin and Amputations European Medicine’s Agency (EMA) has also started to review canagliflozin after this interim result from CANVAS 18 CANVAS R trial (A study of the effects of canagliflozin on renal endpoints in adult participants with type 2 diabetes mellitus) Hasn’t shown same results 17 Non-statistically significant difference between canagliflozin and placebo for amputations 19 16 amputations in canagliflozin group, 12 in placebo Estimated incidence 7/1000 compared to 5/1000 Inagaki et al 20 Didn’t show increased amputation risk

Canagliflozin and Amputations Mechanism– not clear Dehydration? Volume depletion?

Application to Clinical Practice Noteworthy possible risk but shouldn’t significantly affect prescribing Avoid and/or discontinue in patient with lower limb complications Closely monitor patients with risk factors for amputation Peripheral vascular disease Neuropathy Previous amputations Education on preventative foot care and good hydration important
SGLT 2 and Renal Impairment 6/14/2016 “FDA strengthens kidney warnings for diabetes medicines canagliflozin (Invokana®, Invokamet®) and dapagliflozin (Farxiga®, Xigduo XR®)” 21 Prompted by case reports after drug approval

Canagliflozin/Dapagliflozin and Renal Impairment 21 Oct 2014 -sept 2015, ~1. 5 million got prescription for dapagliflozin or canagliflozin In 2. 5 years (March 2013 -Oct 2015), 101 confirmed cases of acute kidney injury (AKI) with dapagliflozin or canagliflozin use 96 required hospitalization 22 admitted to intensive care units 4 deaths (2 cardiac-related) 15 patients received dialysis 3 had underlying CKD 6 were also on ACEI and diuretic ~50% happened within 1 month of drug start

Canagliflozin/Dapagliflozin and Renal Impairment 21 Patient characteristics 28 -79 yo, median age 57 Over 50% <60 yo 51 on ACEI 26 diuretic 6 NSAID 10 previous CKD

Canagliflozin/Dapagliflozin and Renal Impairment 21 Change in renal function Median reported increase of SCr = 1. 5 mg/d. L Median decrease in e. GFR 46 ml/in/1. 73 m 2 Most cases reversible but not all (76 reported discontinuation) 56 reported improvement 11 didn’t improve (including 4 deaths) 3 recovered with sequela

22 Canagliflozin and Renal Effects Generally a drop in GFR in the first 3 -6 weeks of canagliflozin initiation ~26 m. L/min/1. 73 m 2 across studies Trends of e. GFR similar across most subsets Degree of e. GFR slightly higher in elderly and worsened renal function but clinical significance questionable Most e. GFR drops stabilize or are attenuated by 26 -104 weeks >30% reductions in e. GFR more common in CKD patients with canagliflozin vs placebo Canagliflozin 100 mg - 31/332 (9. 3%) Canagliflozin 300 mg - 43/352 (12. 2%) Placebo - 18/367 (4. 9%)

22 Canagliflozin and Renal Effects >50% reduction not significantly different across groups with normal renal function and moderate renal impairment (all less than 1%) Normal renal impairment Canagliflozin 100 mg - none Canagliflozin 300 mg - 1/805 (0. 1%) Placebo- 1/624 (0. 2%) Stage 3 CKD pts: canagliflozin 1. 5% w/>50% drop in SCr, 0. 9% of 300, none for placebo Canagliflozin 100 mg – 5/332 (1. 5%) Canagliflozin 300 mg – 3/352 (0. 9%) Placebo- none In all cases, fewer patients had significant reductions at last post-baseline assessment
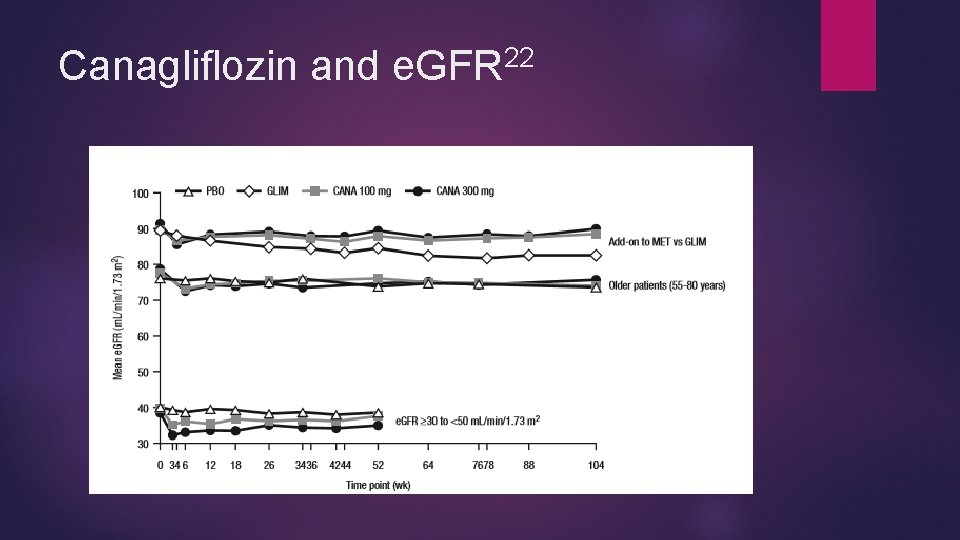
22 Canagliflozin and e. GFR
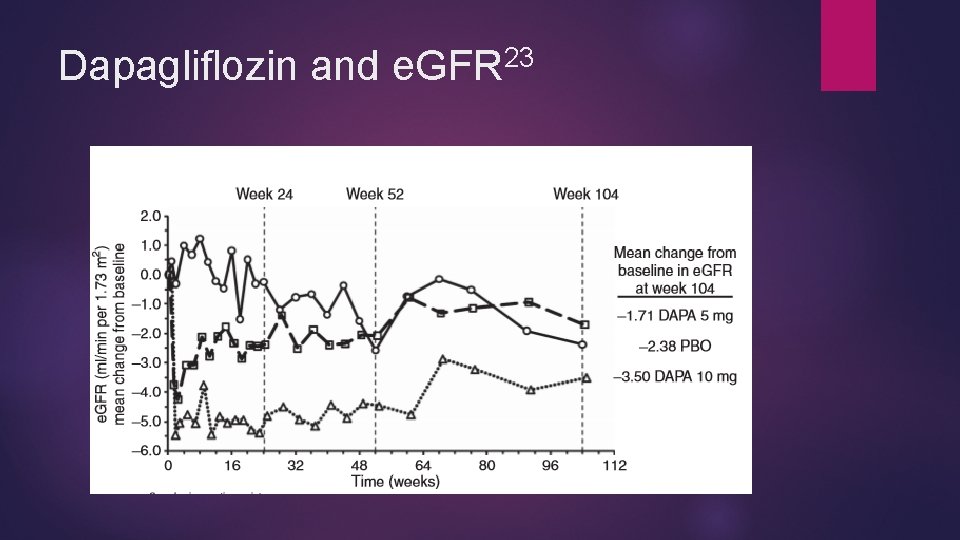
23 Dapagliflozin and e. GFR
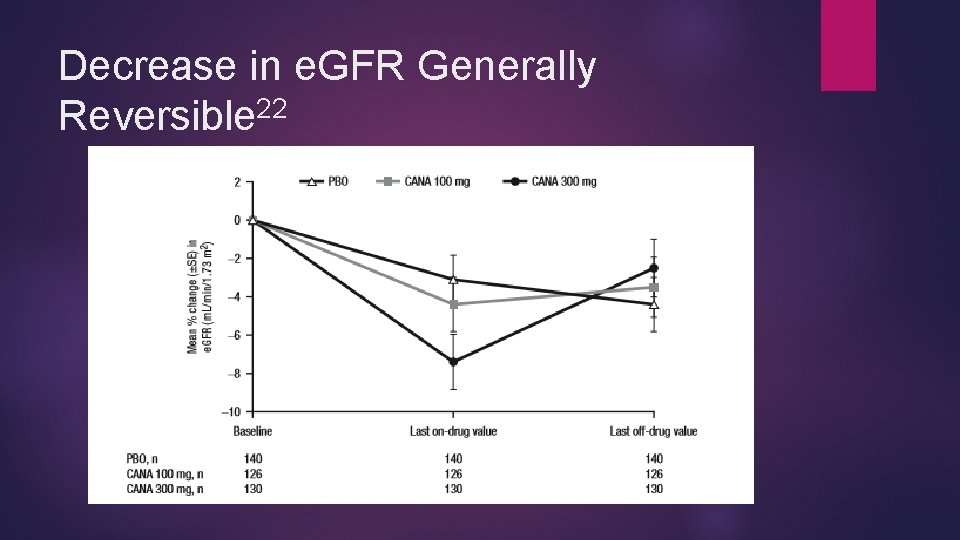
Decrease in e. GFR Generally Reversible 22

Intern tells you he learned in class SGLT 2 s may be renal protective… BAD? OR GOOD? http: //www. ebay. com/itm/DARTH-VADER-STAR-WARS-LIFESIZE-CARDBOARD-STANDUP-STANDEE-CUTOUT-POSTER-FIGURE/271239202903 https: //en. wikipedia. org/wiki/Yoda Accessed 12/3/2016
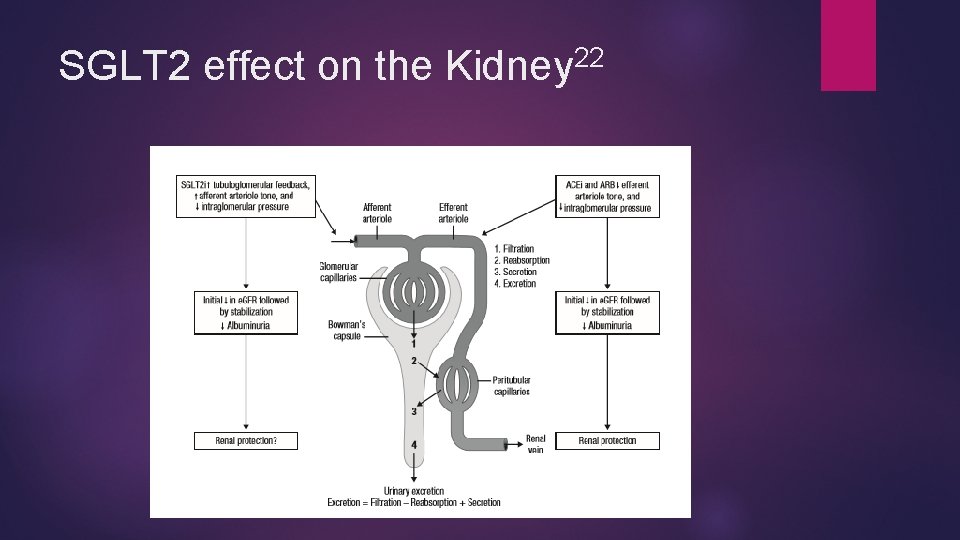
22 SGLT 2 effect on the Kidney

Empagliflozin and Renal Protection EMPA-REG OUTCOME trial 24 Empagliflozin showed renal protection Less incident nephropathy (12. 7 % vs 18. 8%, P < 0. 001) Most independent components significantly less in empagliflozin group as well (shown as empagliflozin vs placebo) Progression to macroalbuminuria (11. 2% vs 16. 2%), RRR 38% Doubling of serum creatinine (1. 5 vs 2. 6%) RRR 44% Progression to renal replacement therapy (0. 3% vs 0. 6%) rrr 55% Benefits seen across pre-specified subgroups and both doses Less acute renal failure in empagliflozin than placebo
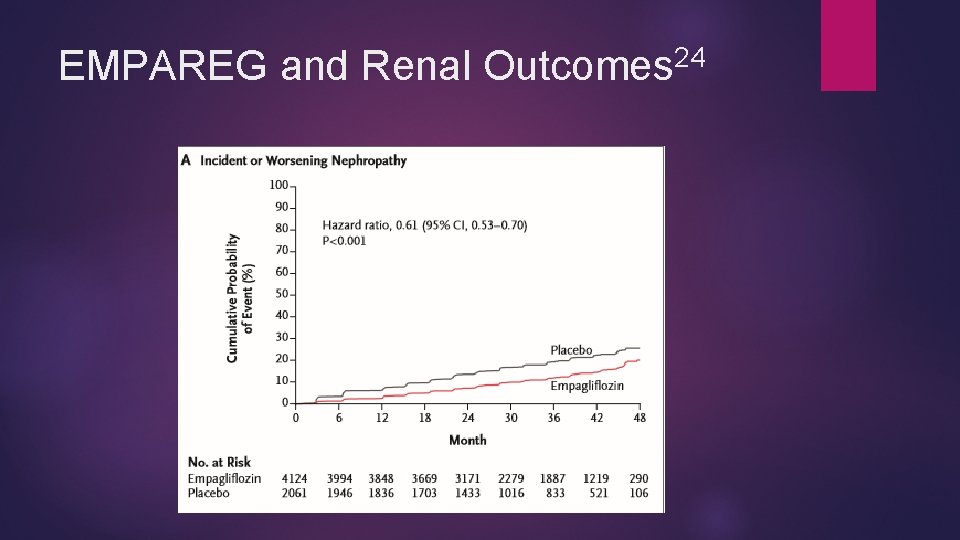
24 EMPAREG and Renal Outcomes
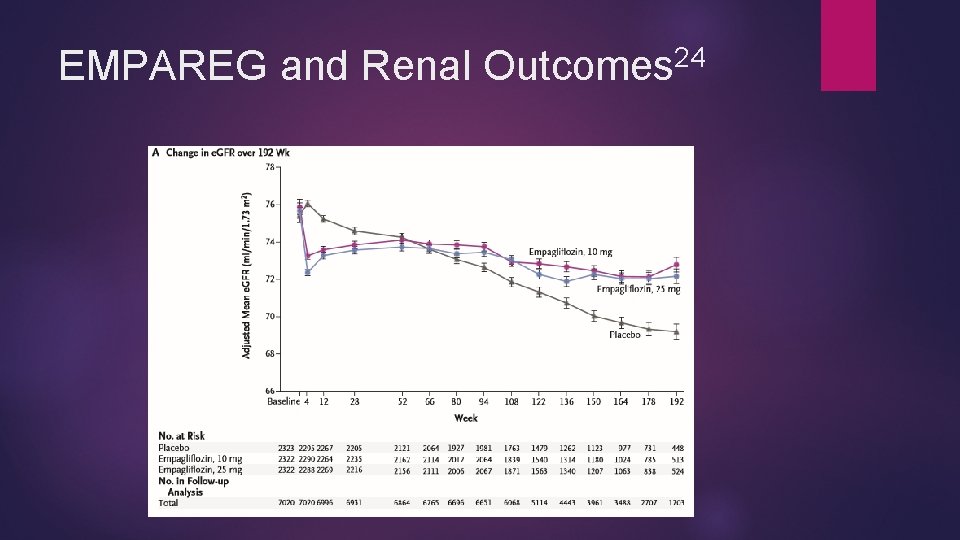
24 EMPAREG and Renal Outcomes
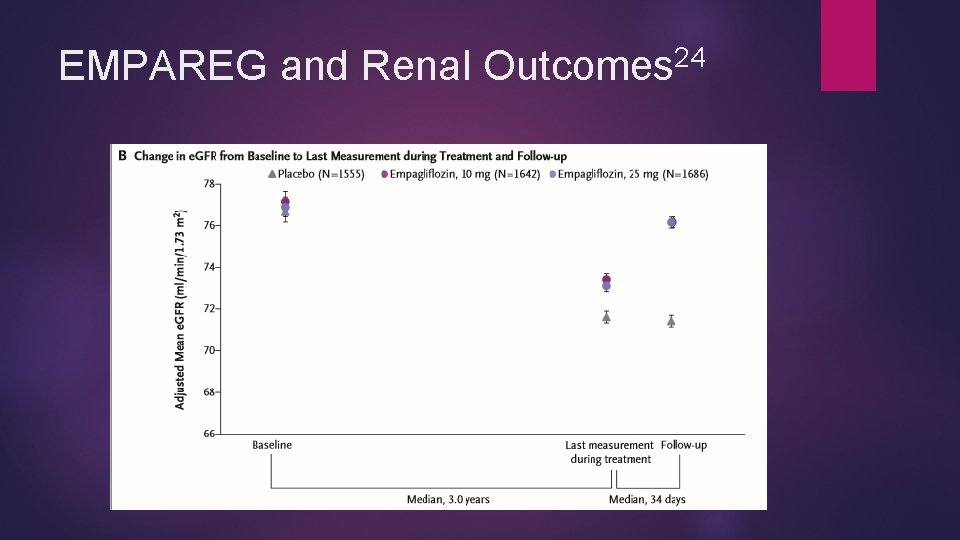
24 EMPAREG and Renal Outcomes

Good or Bad? lif loz Ca g na in? Da pa glif loz in? Empagliflozin http: //www. ebay. com/itm/DARTH-VADER-STAR-WARS-LIFESIZE-CARDBOARD-STANDUP-STANDEE-CUTOUT-POSTER-FIGURE/271239202903 https: //en. wikipedia. org/wiki/Yoda Accessed 12/3/2016

Other SGLT 2 s and Albuminuria Some studies have shown reductions in albumin to creatinine ratios with canagliflozin 25 -28 Most favored improvement in albuminuria but not statistically significant Kohan et al 23 showed dapagliflozin more likely to regress to a lower albumin excretion category

SGLT 2 and Renal Impairment Concern for risk of acute renal impairment Canagliflozin Dapagliflozin Important time for monitoring is in the first 4 -6 weeks Use caution in: HF Poor hydration status Fluid loss Acute GI illness ACE, ARB, NSAIDs, and diuretics Other meds which can potentially cause renal impairment Normotensive patients

SGLT 2 and Renal Impairment Renal impairment is often reversible Empagliflozin provides long term renal protection May be some new data when CANVAS R study is completed SGLT 2 not recommended in patients with impaired renal function Dapagliflozin: Do not initiate therapy with e. GFR less than 60 m. L/min/1. 73 m 2 Not recommended if e. GFR is persistently between 30 and 60 m. L/min/1. 73 m 2 Empagliflozin Do not initiate therapy with e. GFR less than 45 m. L/min/1. 73 m 2 Discontinue if e. GFR is persists below 45 m. L/min/1. 73 m 2 Canagliflozin Do not exceed 100 mg orally once daily if e. GFR is 45 to 60 m. L/min/1. 73 m 2 Do not initiate therapy with e. GFR less than 45 m. L/min/1. 73 m 2 Discontinue if e. GFR is persists below 45 m. L/min/1. 73 m 2

SGLT 2 Counseling Preventative foot care monitoring for cuts/scrapes Seek attention right away if any open wounds, discoloration, or new onset pain or tenderness Importance of good hydration Seek attention if s/sx of water or salt loss Use caution w/diuretics, HF Signs of renal impairment Decreased urine production Swelling in legs or feet Importance of lab follow up

SGLT 2 Counseling Risk of genital infection/UTI, corresponding symptoms Risk of hypotension Seek attention if develops dizziness Recommend blood pressure monitoring while on tx DKA Counsel on symptoms and risk Highly importance of seeking IMMEDIATE attention if has symptoms Highlight for empagliflozin only CV benefit in patients with CV disease Renal protection

Metformin and Renal Dosing Previous labeling: Contraindicated Men with SCr >1. 5 mg/d. L Women with SCr >1. 4 mg/d. L Intended to prevent use in patients with any type of renal impairment for safety Main concern was lactic acidosis risk

Metformin and Renal Dosing 4/8/2016 FDA determined after evaluating the current evidence 29 e. GFR should be used for determination of renal function Safe use of metformin in mild renal impairment Labeling modified New guidance has been used prior to FDA label change 30

29 FDA Changes to Metformin Labeling Before starting, check creatinine/determine e. GFR Check e. GFR At least annually More often in high risk populations Iodinated contrast imaging Stop prior in below patients e. GFR 30 -60 m. L/minute/1. 73 m 2 Liver disease Alcoholism Heart failure Re-check e. GFR 48 hours after Resume when renal function stable

29 FDA Changes to Metformin Labeling e. GFR level Recommendation Less than 30 ml/min per 1. 73 m 2 -Contraindicated. Do NOT use. -Discontinue if currently taking <45 and > 30 ml/min per 1. 73 m 2 -Assess the benefits and risks of continuing treatment -Do not start new patients on metformin

Metformin and Renal Dosing: Clinical Application Possible implications Decrease in medical costs Delayed need for insulin May decrease weight in some patients from net decrease in insulin requirements Other factors beyond renal impairment must be considered prior to metformin initiation/continuation

TZDs and Bladder Cancer 12/12/2016 “Updated FDA review concludes that use of type 2 diabetes medicine pioglitazone may be linked to an increased risk of bladder cancer” 31 Risk already included in current labeling FDA approved label updates to include new data that has been reviewed Animal models have shown bladder tumors with pioglitazone exposure 32 Higher reporting of bladder cancer to FDA Adverse Event Reporting System for pioglitazone than other diabetes treatment 33 Human studies produced conflicting evidence

Is Pioglitazone Associated with a Higher Risk of Bladder cancer? Studies with significant risk Lewis et al 5 year interim analysis 34 Only in patients on pio >2 years Hazard ratio 1. 4 (CI 1. 03 -2. 0) Lewis et al 10 Year follow up study 37** Tuccori et al 35 No significant difference overall, based on duration OR dose PROactive trial 38** Higher risk overall (HR 1. 63, 1. 22 -2. 19) Higher risk >2 years use (HR 1. 78, 1. 21 -2. 64) 14/2605 in pio vs 6/2633 in placebo P = 0. 069 Dose response relation (p =0. 01 for trend) P = 0. 309 when cases in first year were removed Studies with no significant risk Ferwana meta-analysis 36 Overall risk (HR 1. 23, CI 1. 09 -1. 39) PROactive 10 year follow up 39** 21 (1. 2%) in pio vs 14 (0. 8%) in placebo, (RR 0. 65, CI 0. 33 -1. 28) **denotes no significant risk but trend suggested higher risk in pio group

What Do We Make of All This? Incidence of bladder cancer in pio and placebo is low Causality cannot be confirmed RCT study suggests no significant risk 38 Risk in observational studies may be in part related to unequal distribution of risk factors in pioglitazone vs placebo Bladder cancer risk cannot be excluded Until more definitive conclusions, must use caution and take heed of this possible risk Not recommended in patients with bladder cancer or a history of bladder cancer Recommend caution in family history of bladder cancer or other risk factors for bladder cancer Risk/benefit must be weighed on a patient specific basis

Counseling Advise patients to seek attention if: Blood in urine New or worsening urge to urinate Pain when urinating Other counseling points Edema SOB Weight gain

Reminders from 2016 FDA Alerts With new medication benefits come new risks Diabetes treatment selection is highly patient specific Education is very important for patient safety

American Diabetes Association 2017 Updates 40 Recommend periodic B 12 monitoring in patients on metformin Figure 8. 1 updated to acknowledge high insulin costs
American Diabetes Association 2017 Updates 40 Update to Figure 8. 2
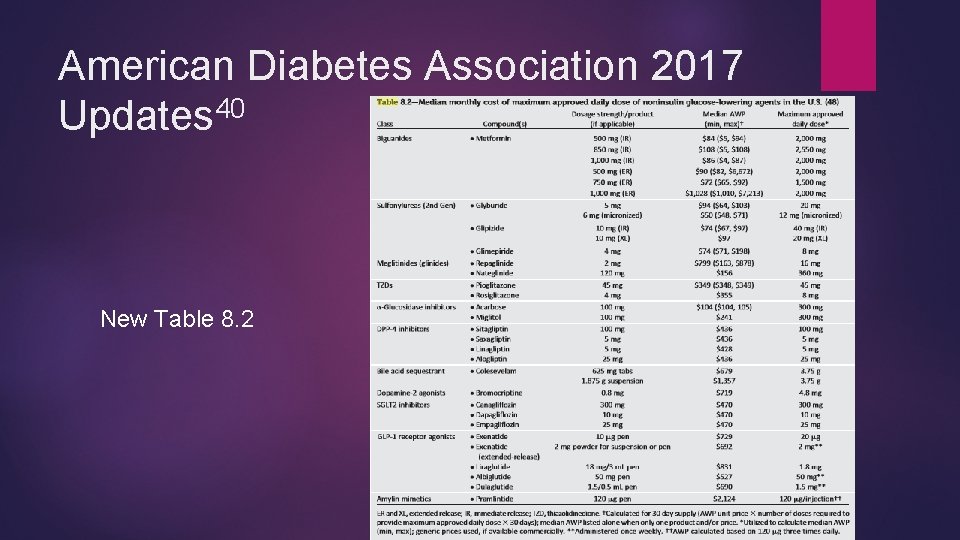
American Diabetes Association 2017 Updates 40 New Table 8. 2
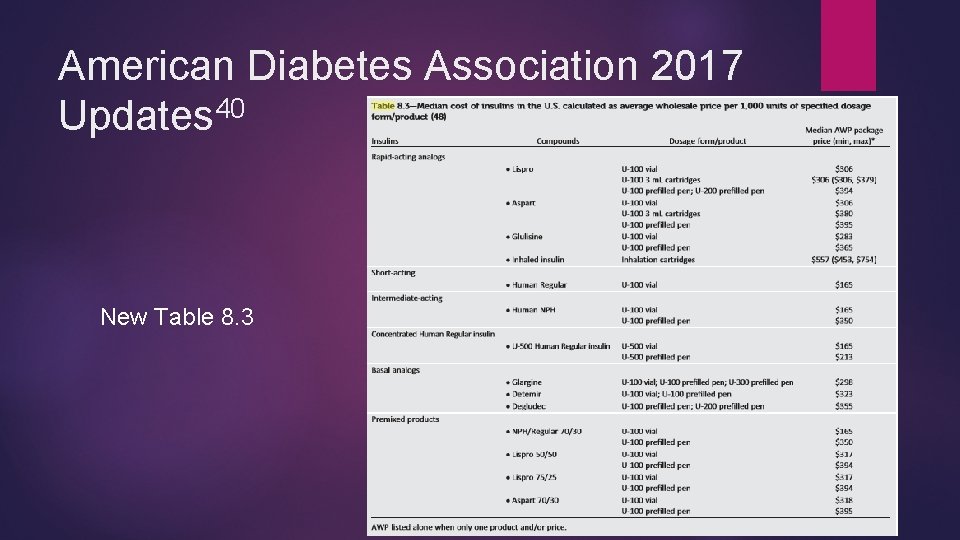
American Diabetes Association 2017 Updates 40 New Table 8. 3

American Diabetes Association 2017 Updates 40 Recommendation to consider empagliflozin or liraglitide in patients with history of cardiovascular disease to reduce risk of mortality Section on new biosimilar insulins Any of following recommended for hypertension treatment if no albuminuria ACEI/ARB Thiazide diuretics Dihydropyridine calcium channel blockers

American Diabetes Association 2017 Updates 40 Specific recommendations added for treatment of neuropathic pain Pregabalin Duloxetine Inpatient diabetes management Basal + correctional scale ok in non-critically ill patients but not correctional scale alone

American Diabetes Association 2017 Updates 40

American Diabetes Association 2017 Updates 40 Non pharmacologic updates (included but not limited to) Advise interruption of sitting every 30 minutes Possibly discuss unifying DM classification scheme based on beta cell function Blood pressure targets for pregnant women changed: 120 -160/80 -105 optimize maternal health without inducing fetal harm Recommendations added surrounding incorporating social considerations into treatment decisions

Pharmacists’ Roles in Diabetes ate c o dv A Inves tigato r Teac h er COACH er d a rle e e h C https: //timemanagementninja. com/2014/03/how-many-hats-are-you-trying-to-wear/

Potential Areas of Pharmacist Impact in Diabetes Medication-related Proper Timing Administration Storage Hypoglycemia Lifestyle/Other Risks of poor DM control Eating behaviors Exercise Lab monitoring Signs and symptoms Blood sugar testing Treatment Glucose/A 1 c targets Lancets Social support Optimizing drug treatment Safety Efficacy Cost

“It’s the little details that are vital. Little things make big things happen. ” ~John Wooden

Questions?

References 1. U. S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research. Guidance for Industry: Diabetes Mellitus—Evaluating Cardiovascular risk in new antidiabetic therapies to treat type 2 diabetes. December 2008. Pages 1 -5 2. Smith RJ, Goldfine AB, Hiatt WR. Evaluating the cardiovascular safety of new medications for Type 2 diabetes: time to reassess? Diabetes Care 2016; 39: 738 -42. 3. FDA Drug Safety Communication. FDA adds warning about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin. http: //www. fda. gov/Drugs/Drug. Safety/ucm 486096. htm Issued 4/5/16, Accessed 12/2/16. 4. Scirica BM, Bhatt DL, Braunwald E, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. NEJM 2013; 369: 1317 -26. 5. Zannad F, Cannon CP, Cushman WC, et al. Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: a multicenter, randomized, double blind trial. Lancet 2015; 285: 2067 -76. White WB, Cannon CP, Heller SR, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. NEJM 2013; 369: 1327 -35. 7. Green JB, Bethel MA, Armstorng PW, et al. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. NEJM 2015; 373: 232 -42. (TECOS) 8. Toh S, Hamp C, Reichman ME, et al. Risk for hospitalized heart failure among new users of saxagliptin, sitagliptin, and other antihyperglycemic drugs: a retrospective cohort study. Ann Intern Med 2016; 164: 705 -14. 9. Weir DL, Mc. Alister FA, Senthilselvan, et al. Sitagliptin use in patients with diabetes and heart failure: a population based retrospective cohort study. JACC Heart Fail 2014; 2: 573 -82. 10. Wang KL, Liu CJ, Chao TF, et al. Sitagliptin and the risk of hospitalization for heart failure: a population-based study. Int J Cardiol 2014; 177: 86 -70.

References 11. Chen DY, Wang SH, Mao CT, et al. Sitagliptin and cardiovascular outcomes in diabetic patients with chronic kidney disease and acute myocardial infarction; a nationwide cohort study. Int J Cardiol 2015; 181: 200 -6. 12. Raschi E, Poluzzi E, Koci A, et al. Dipeptidyl peptidase-4 inhibitors and heart failure: Analysis of spontaneous reports submitted to the FDA Adverse Event Reporting System. Nutrition, Metabolism & Cardiovascular Diseases 2016; 26: 380 -6. 13. Lehrke M, Leiter LA, Hehnke U, et al. Safety and efficacy of linagliptin in patients with type 2 diabetes mellitus and coronary artery disease: analysis of pooled events from 19 clinical trials. Journal of Diabetes and Its Complications 2016; 30: 1378 -84. 14. FDA approves Jardiance to reduce cardiovascular death in adults with type 2 diabetes. FDA News Release http: //www. fda. gov/News. Events/Newsroom/Press. Announcements/ucm 531517. htm Published 12/2/16, Accessed 12/5/16. 15. Zinman B, Wanner C, Lachin JM, et al. Empagliflozing, cardiovascular outcomes, and mortality in type 2 diabetes. NEJM 2015; 373: 2117 -28. 16. PL Detail-Document, Empagliflozin and Cardiovascular Disease. Pharmacist’s Letter/Prescriber’s Letter. November 2015 17. FDA. Canagliflozin (Invokana®, Invokanamet®): Drug safety communication – clinical trial results find increased risk of leg and foot amputations. http: //www. fda. gov/Drugs/Drug. Safety/ucm 500965. htm Issued 5/18/16, Accessed 12/2/16. 18. Canagliflozin and the risk of leg and foot amputations. http: //www. diabetesincontrol. com/canaglfilozin-amputation-risk/ Article published 6/4/16, Accessed 12/15/16. 19. Drug Safety Update vol 9 issue 11, June 2016: 1. (accessed https: //www. gov. uk/drug-safety-update/canagliflozin-invokana-vokanamet -signal-of-increased-risk-of-lower-extremity-amputations-observed-in-trial-in-high-cardiovascular-risk-patients) 20. Inagaki N, Harashima S, Maruyama N, et al. Efficacy and safety of canagliflozin in combination with insulin: a double-blind, randomized, placebo-controlled study in Japanese patients with type 2 diabetes mellitus. Cardiovasc Diabetol 2016; 15: 89.

References 21. FDA. Drug Safety Communication: FDA strengthens kidney warnings for diabetes medicines canagliflozin (Invokana®, Invokanamet®) and dapagliflozin (Farxiga®, Xigduo XR®). http: //www. fda. gov/Drugs/Drug. Safety/ucm 505860. htm Issued 6/14/16, Accessed 12/2/16. 22. Perkovic V, Jardine M, Vijapurkar U, et al. Renal effects of canagliflozin in type 2 diabetes mellitus. Current Medical Research and Opinion 2015; 31: 2219 -31. 23. Kohan DE, Fioretto P, Tang W, et al. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney International 2014; 85: 962 -71. 24. Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and progression of kidney disease in type 2 diabetes. NEJM 2016; 375: 323 -34. 25. Leiter LA, Yoon KH, Arias P, et al. Canagliflozin Provides Durable Glycemic Improvements and Body Weight. Reduction. Over 104 Weeks Versus Glimepiride in Patients With Type 2 Diabetes on Metformin: A Randomized, Double-Blind, Phase 3 Study. Diabetes Care 2015; 38: 355 -64. 26. Yale JF, Bakris G, Cariou B, et al. Efficacy and safety of canagliflozin in subjects with type 2 diabetes and chronic kidney disease. Diabetes, Obesity, and Metabolism 2013; 15: 463 -73. 27. Yale JF, Bakris G, Cariou B, et al. Efficacy and safety of canagliflozin over 52 weeks in patients with type 2 diabetes mellitus and chronic kidney disease. Diabetes, Obesity, and Metabolism 2014; 16: 1016 -27. 28. Cefalu WT, Leiter LA, Yoon KH, et al. E�cacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet 2013; 382: 941 -50. 29. FDA. Drug Safety Communication: FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function. http: //www. fda. gov/Drugs/Drug. Safety/ucm 493244. htm Issued 4/8/16, Accessed 12/2/16. 30. Insucchi SE, Lipska KJ, Mayo H, et al. Metformin in patients with type 2 diabetes and kidney disease: a systematic review. JAMA 2014; 312: 2668 -75.

References 31. FDA. Drug Safety Communication: Updated FDA review concludes that use of type 2 diabetes medicine pioglitazone may be linked to an increased risk of bladder cancer. http: //www. fda. gov/Drugs/Drug. Safety/ucm 519616. htm Issued 12/12/16, Accessed 12/20/16. 32. Suzuki S, Arnold LL, Pennington KL, et al. Effects of pioglitazone, a peroxisome proliferator-activated receptor gamma agonist, on the urine and urothelium of the rat. Toxicol Sci 2010; 113: 349 -57. 33. Piccini C, Motola D, Marchesni G, et al. Assessing the association of pioglitazone use and bladder cancer through adverse drug event reporting. Diabetes Care 2011; 34: 1369 -71. 34. Lewis JD, Ferrara A, Peng T, et al. Risk of bladder cancer among diabetic patients treated with pioglitazone: interim report of a longitudinal cohort study. Diabetes Care 2011; 34: 916 -22. 35. Tuccori M, Filion KB, Yin H, et al. Pioglitazone use and risk of bladder cancer: population based cohort study. BMJ 2016; 352: i 1541. 36. Ferwana M, Firwana B, Hasan R, et al. Pioglitazone and risk of bladder cancer: a meta-analysis of controlled studies. Diabet Med 2013; 30: 1026 -32. 37. Lewis JD, Habel LA, Quesenberry CP, et al. Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 2015; 314; 265 -77. 38. Dormandy JA, Charbonnel B, Eckland DJA, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitazone clinical trial in macrovascular events): a randomized controlled trial. Lancet 2005; 366: 1279 -89. 39. Erdmann E, Harding S, Lam H, et al. Ten-year observational follow-up of PROactive: a randomized cardiovascular outcomes trial evaluating pioglitazone in type 2 diabetes. Diabetes, Obesity and Metabolism 2016; 18: 266 -73. 40. American Diabetes Association. Promoting health and reducing disparities in populations. Sec. 1. In Standards of Medical Care in Diabetesd 2017. Diabetes Care 2017; 40(Suppl. 1): S 1–S 129

References Photo/cartoon references 1. Three minion photo. http: //www. steamworksstudio. com/bparty. html Accessed 1/1/2017 2. Minion wallpaper. http: //weheartit. com/entry/73189313/in-set/11689299 -minions? context_user=nelita_fer Accessed 1/1/2017 3. Blood pressure and cancer image. http: //abcnews. go. com/Health/Hypertension. News/study-links-popular-blood-pressure-drugs-cancer/story? id=10909261 Accessed 1/1/2017 4. Heartburn and cancer image. http: //www. cnn. com/2016/11/15/health/heartburn-medication-stroke-risk/ Accessed 1/1/2017 5. Statins and memory loss image. http: //www. cbsnews. com/news/statins-and-memory-loss-should-you-worry/ Accessed 1/1/2017 6. “Please have a seat” photo. https: //www. pinterest. com/kascott 29/rx/ Accessed 1/1/2017 7. New Kids on the Block Photo. http: //images 4. fanpop. com/image/photos/22100000/nkotb-new-kids-on-the-block-22146585 -1152 -1156. jpg Accessed 12/2/2016. 8. Monopoly guy photo https: //koshersamurai. files. wordpress. com/2012/12/monopoly-man-rich-guy. jpg accessed 1/1/2017. 9. Jardiance 10 mg photo http: //www. multivu. com/players/English/7617351 -bi-lilly-empa-reg-cv-results/ 10. SGLT Inhibitor Photos. http: //www. mims. co. uk/sglt 2 -inhibitors-risk-diabetic-ketoacidosis/diabetes/article/1353980 Copyright Haymark Media Group. Accessed 12/3/2016 11. Darth Vader photo. http: //www. ebay. com/itm/DARTH-VADER-STAR-WARS-LIFESIZE-CARDBOARD-STANDUP-STANDEE-CUTOUT-POSTER-FIGURE/271239202903 Accessed 12/1/2016 12. Yoda photo https: //en. wikipedia. org/wiki/Yoda Accessed 12/3/2016 13. Kylo Ren photo. https: //twitter. com/kylor 3 n 14. Balancing hats photo. https: //timemanagementninja. com/2014/03/how-many-hats-are-you-trying-to-wear/ Accessed 1/1/2017
- Slides: 76