The Development of Bioabsorbable DrugEluting Stents for Peripheral

The Development of Bioabsorbable Drug-Eluting Stents for Peripheral Vascular Interventions Ron Waksman, MD, FACC, FSCAI Professor of Medicine, Georgetown University, Associate Chief of Cardiology, Washington Hospital Center, Washington DC

Ron Waksman, MD §Consulting Fee §Abbott Laboratories §Biotronik, Inc. §Boston Scientific Corporation §Medtronic, Inc. §Merck and Company, Inc.

§ Honoraria § Abbott Laboratories § Boston Scientific Corporation § Medtronic, Inc. Consulting Fee § Merck and Company, Inc.

Why absorbable scaffolds? Why permanent stents? Vessel scaffolding is necessary only for a certain, limited time, than the permanent implant has no known advantage • Restoration of natural physiologic vasomotor function • Enable vascular remodeling and tissue adaptation • Elimination of chronic sources of vessel irritation and sources for chronic inflammation • Avoid current challenges with leaving a metal implant behind • Potentially reduce the need for prolonged DAPT • No permanent implant to complicate future interventions and reinterventions, particularly in younger patients • Non-invasive imaging with MSCT or MRA without ‘blooming artifact’
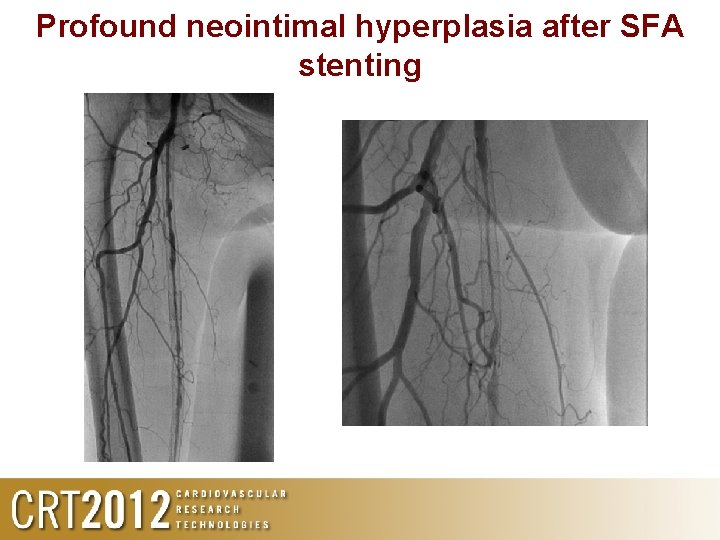
Profound neointimal hyperplasia after SFA stenting

Amaranth Medical PLLA biodegradable Scaffold • Capabilities: − 3. 0 -9. 0 mm diameters − 10 -200 mm lengths • Stent designs suitable for multiple vascular applications − Coronary − Peripheral − Neuro • First product - superficial femoral artery (SFA) stent
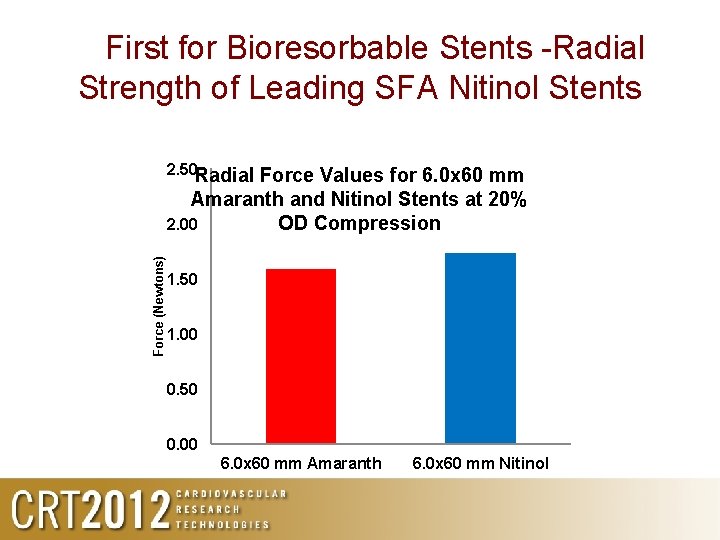
A First for Bioresorbable Stents -Radial Strength of Leading SFA Nitinol Stents Amaranth stents show radial strength similar to Nitinol stent 2. 50 Radial Force (Newtons) Force Values for 6. 0 x 60 mm Amaranth and Nitinol Stents at 20% 2. 00 OD Compression 1. 50 1. 00 0. 50 0. 00 6. 0 x 60 mm Amaranth 6. 0 x 60 mm Nitinol
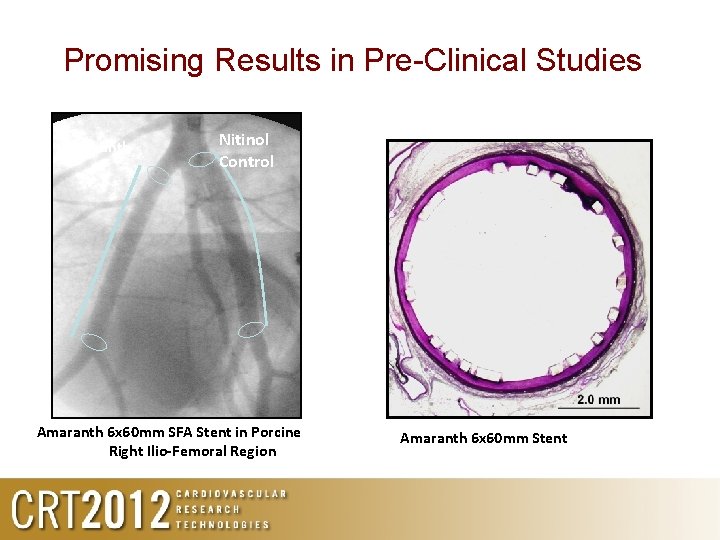
Promising Results in Pre-Clinical Studies Amaranth Nitinol Control Amaranth 6 x 60 mm SFA Stent in Porcine Right Ilio-Femoral Region Amaranth 6 x 60 mm Stent
Motivation for Bioabsorbable Stent
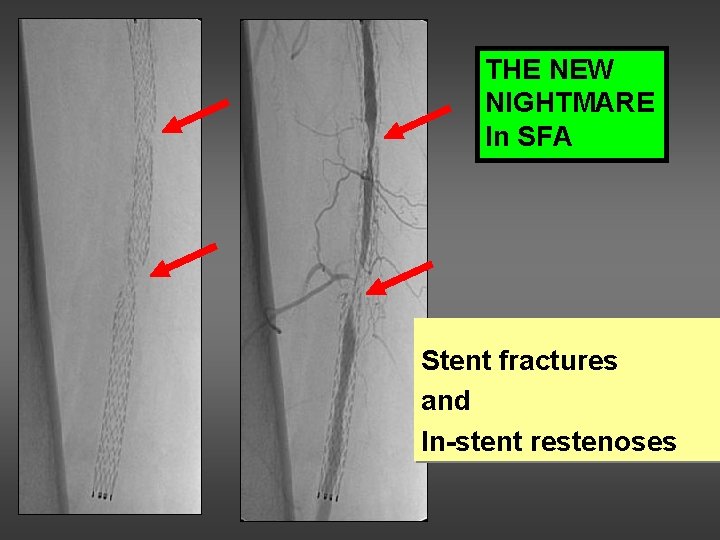
THE NEW NIGHTMARE In SFA Stent fractures and In-stent restenoses
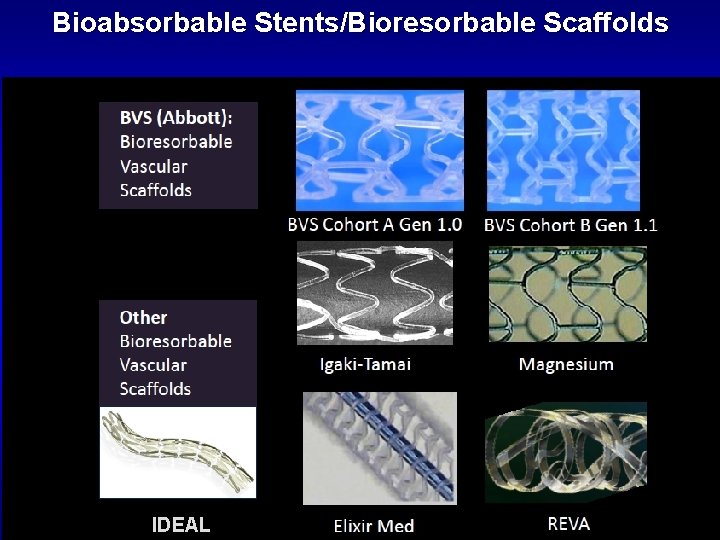
Bioabsorbable Stents/Bioresorbable Scaffolds IDEAL

Ikagi Tamai Stent CE Mark 2006 For SFA Use
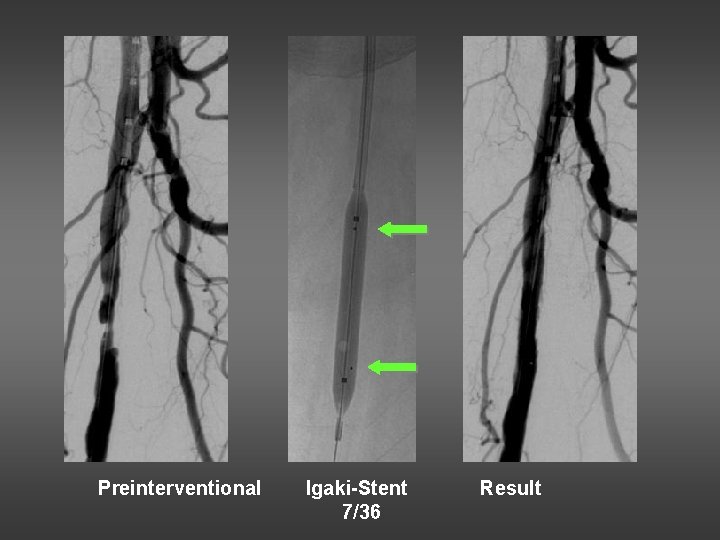
Preinterventional Igaki-Stent 7/36 Result
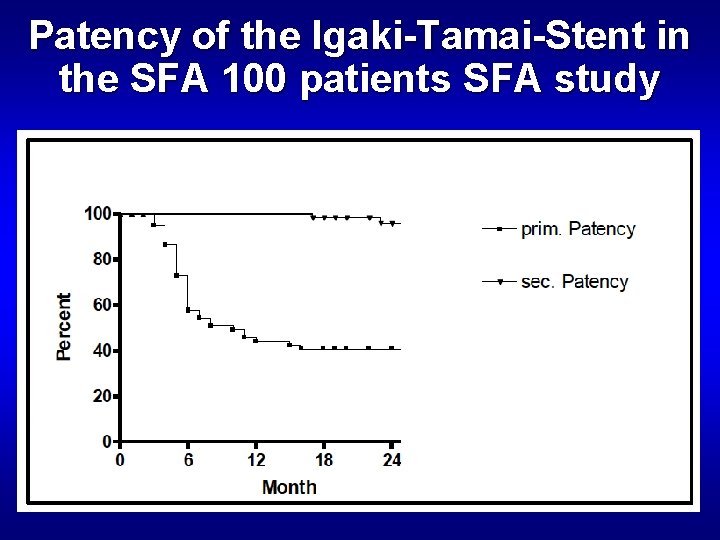
Patency of the Igaki-Tamai-Stent in the SFA 100 patients SFA study
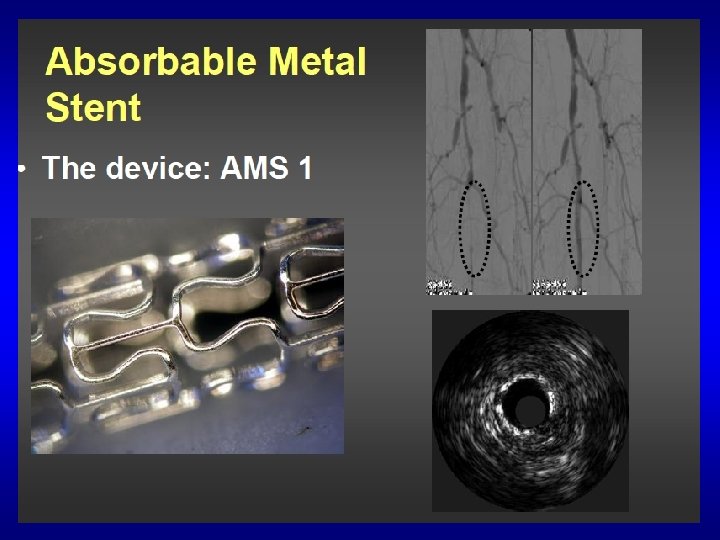

ABSORB neither approved nor available for. INSIGHT. sale in the CIRSE U. S. Bosiersis. M, Deloose K, Peeters P. AMS 2008.
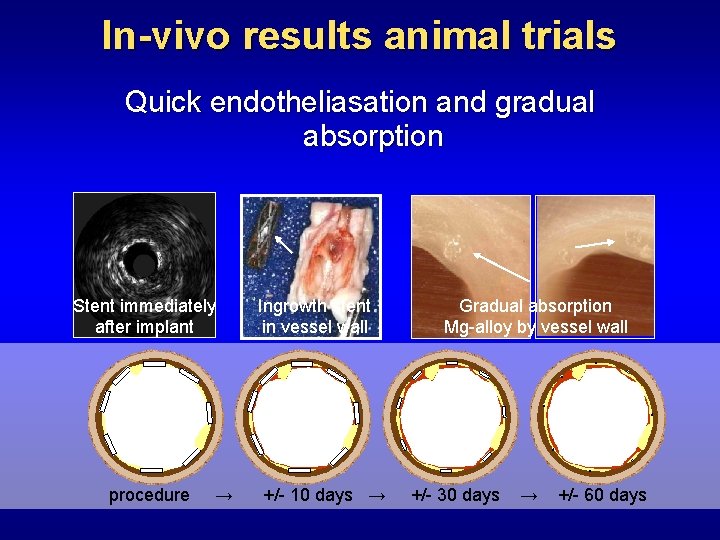
In-vivo results animal trials Quick endotheliasation and gradual absorption Stent immediately after implant procedure → Ingrowth stent in vessel wall +/- 10 days → Gradual absorption Mg-alloy by vessel wall +/- 30 days → +/- 60 days
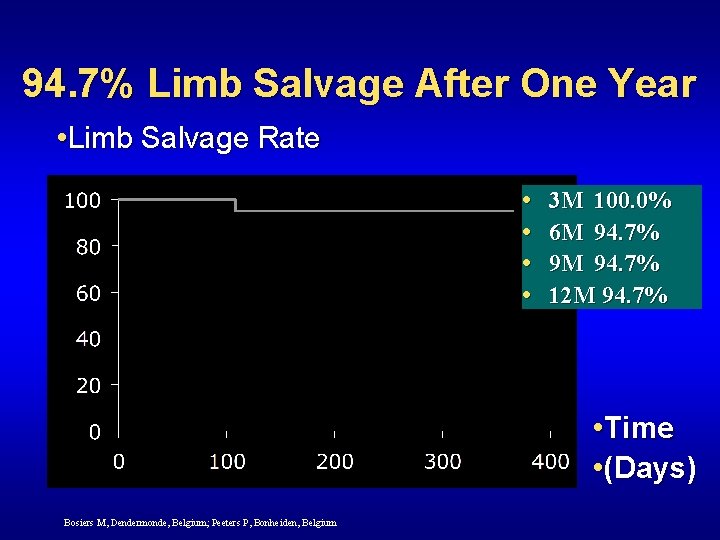
94. 7% Limb Salvage After One Year • Limb Salvage Rate • • 3 M 100. 0% 6 M 94. 7% 9 M 94. 7% 12 M 94. 7% • Time • (Days) Bosiers M, Dendermonde, Belgium; Peeters P, Bonheiden, Belgium
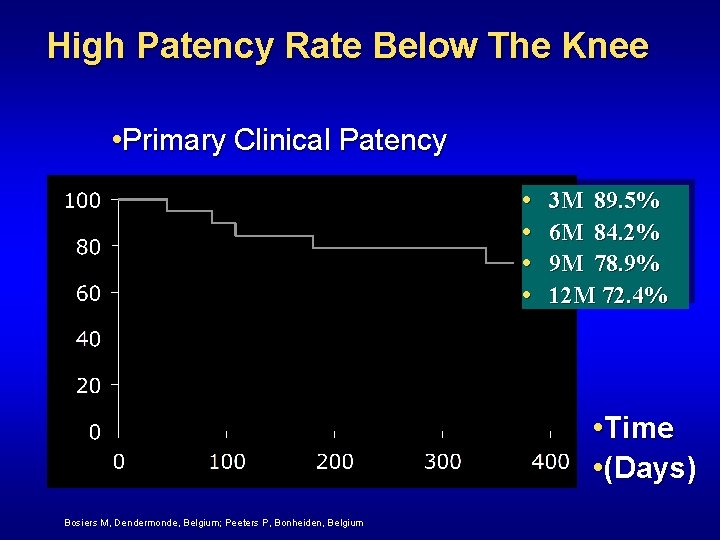
High Patency Rate Below The Knee • Primary Clinical Patency • • 3 M 89. 5% 6 M 84. 2% 9 M 78. 9% 12 M 72. 4% • Time • (Days) Bosiers M, Dendermonde, Belgium; Peeters P, Bonheiden, Belgium
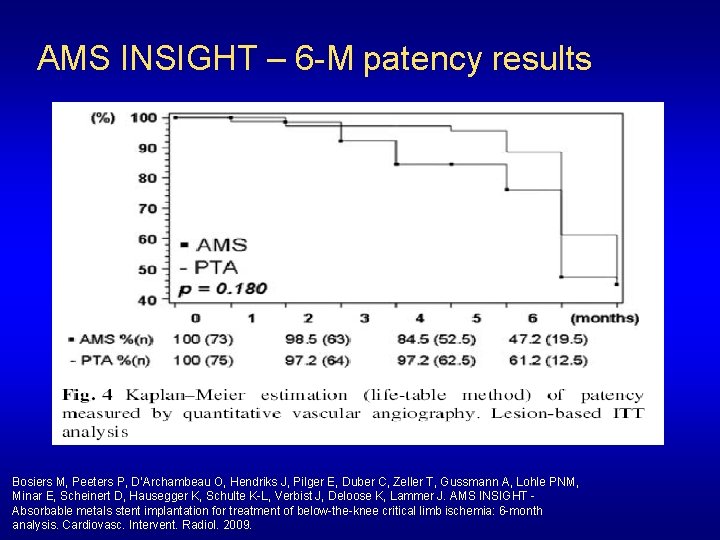
AMS INSIGHT – 6 -M patency results Bosiers M, Peeters P, D’Archambeau O, Hendriks J, Pilger E, Duber C, Zeller T, Gussmann A, Lohle PNM, Minar E, Scheinert D, Hausegger K, Schulte K-L, Verbist J, Deloose K, Lammer J. AMS INSIGHT Absorbable metals stent implantation for treatment of below-the-knee critical limb ischemia: 6 -month analysis. Cardiovasc. Intervent. Radiol. 2009.

DREAMS, the DRug Eluting Absorbable Metal Scaffold Is Fully Degradable Drug carrier • Fast degrading polymer Backbone • Customized Mg-alloy • Good biocompatibility Delivery system • Modified PRO-Kinetic Drug • Antiproliferative paclitaxel Sizes used in FIM • 3. 25/3. 5 x 16 • 6 F compatibility
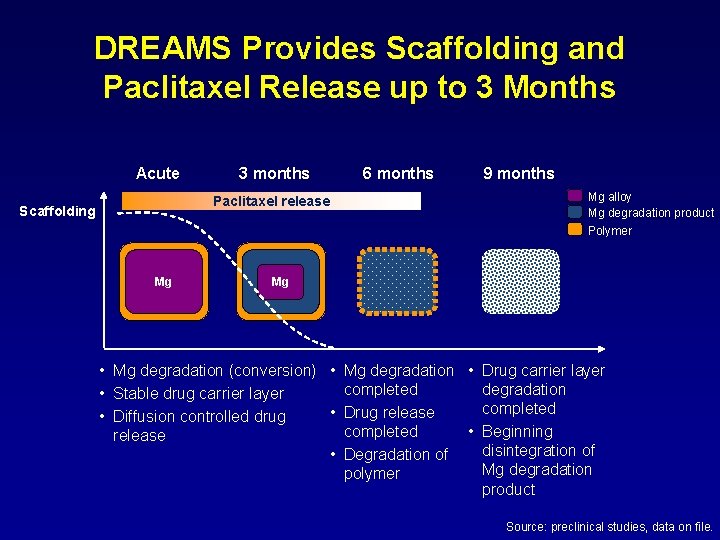
DREAMS Provides Scaffolding and Paclitaxel Release up to 3 Months Acute 3 months Paclitaxel release Scaffolding Mg 6 months 9 months Mg alloy Mg degradation product Polymer Mg • Mg degradation (conversion) • Mg degradation • Drug carrier layer completed degradation • Stable drug carrier layer completed • Drug release • Diffusion controlled drug completed • Beginning release disintegration of • Degradation of Mg degradation polymer product Source: preclinical studies, data on file.

Early Prototypes for “big AMS” 8 mm BE AMS 8 mm SE AMS

ABSORB Scaffold Coating Drug Delivery system Bioresorbable Everolimus XIENCE V® Poly(D, L-lactide) Poly(L-lactide) (PDLLA) coating (PLLA) Naturally resorbed, fully metabolized ABSORB is neither approved nor available for sale in the U. S. Similar dose density and release rate to XIENCE V® World-class deliverability
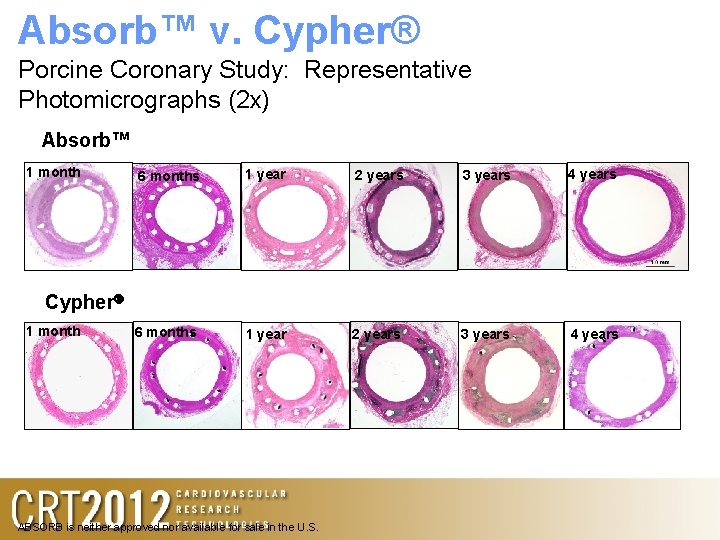
Absorb™ v. Cypher® Porcine Coronary Study: Representative Photomicrographs (2 x) Absorb™ 1 month 6 months 1 year 2 years 3 years 4 years Cypher 1 month ABSORB is neither approved nor available for sale in the U. S.
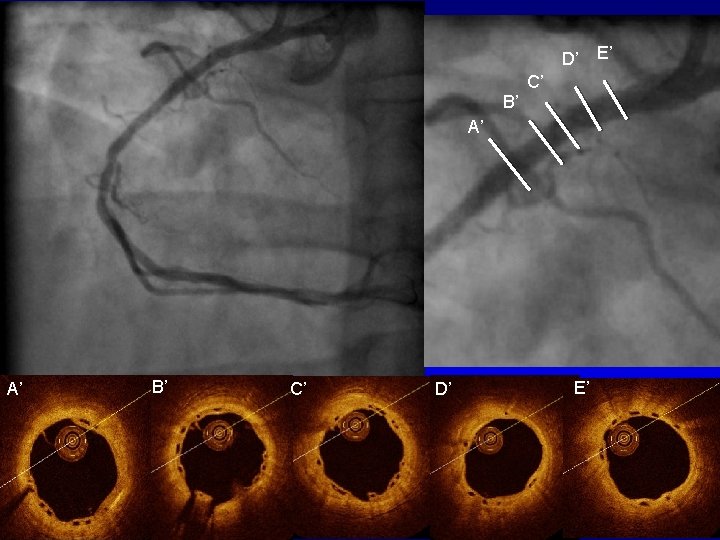
D’ C’ B’ A’ A’ B’ C’ ABSORB is neither approved nor available for sale in the U. S. D’ E’ E’
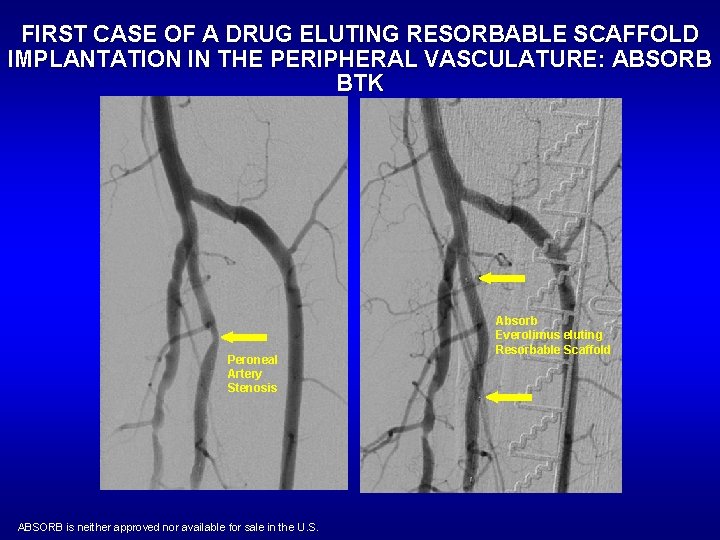
FIRST CASE OF A DRUG ELUTING RESORBABLE SCAFFOLD IMPLANTATION IN THE PERIPHERAL VASCULATURE: ABSORB BTK Peroneal Artery Stenosis ABSORB is neither approved nor available for sale in the U. S. Absorb Everolimus eluting Resorbable Scaffold
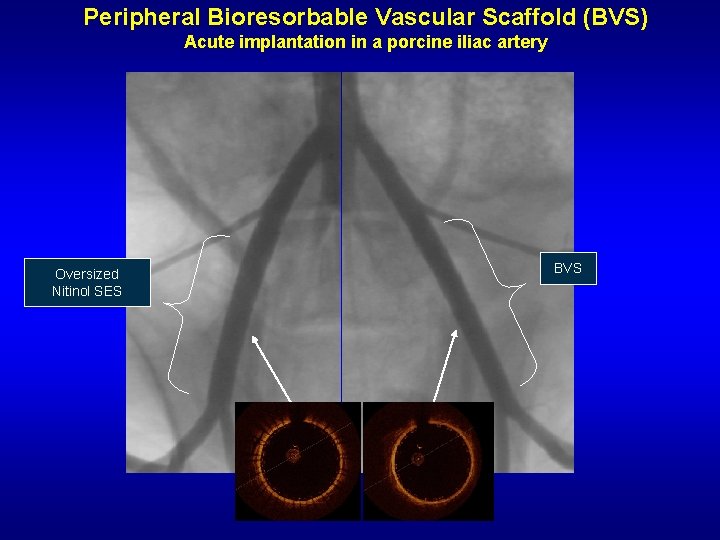
Peripheral Bioresorbable Vascular Scaffold (BVS) Acute implantation in a porcine iliac artery Oversized Nitinol SES BVS
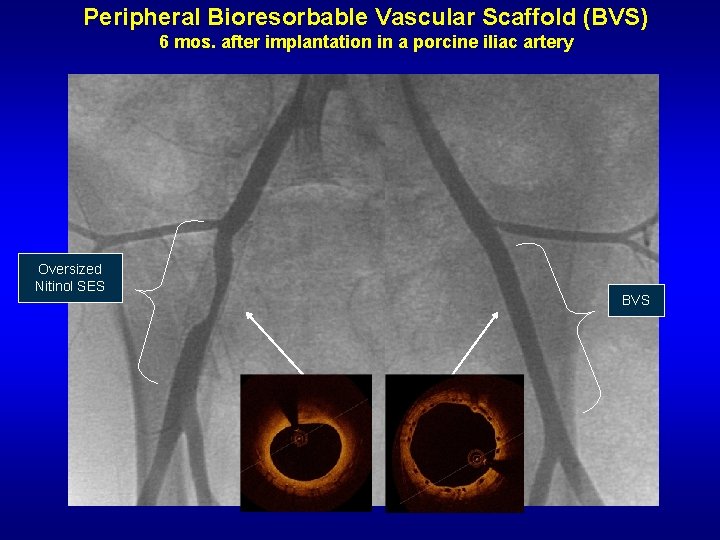
Peripheral Bioresorbable Vascular Scaffold (BVS) 6 mos. after implantation in a porcine iliac artery Oversized Nitinol SES BVS

ESPRIT I: First Clinical Trial to Study Drug Eluting Bioresorbable Therapy for Treatment of Peripheral Artery Disease §Esprit is a drug eluting bioresorbable vascular scaffold that uses PLLA the same polymer and drug as the Absorb BVS §The ESPRIT I trial is a single-arm, multi-center trial designed to evaluate the safety and performance of the Esprit therapy in approximately 30 patients with claudication resulting from PAD of the SFA or iliac arteries. § Up to 10 clinical trial sites in Europe will participate in the trial. Patients will undergo clinical hemodynamic functional and quality-of-life follow-up at one, six and 12 months and at two and three years following the procedure. December 2011

Bioabsorbable Scaffold for PAD • Treating blockages in the SFA is a clinical challenge, as there are unique biomechanical forces exerted on the SFA during normal leg movement, which can lead to restenosis or re-narrowing of the vessel • The drug eluting bioabsorbable scaffold technology is now expanding for the treatment of SFA and BTK lesions. • A bioresorbable drug eluting device, designed to act as a temporary scaffold to support the vessel and then fully dissolve, may change the way we treat peripheral artery disease.
- Slides: 32