The Concept of Equilibrium Chemical equilibrium occurs when
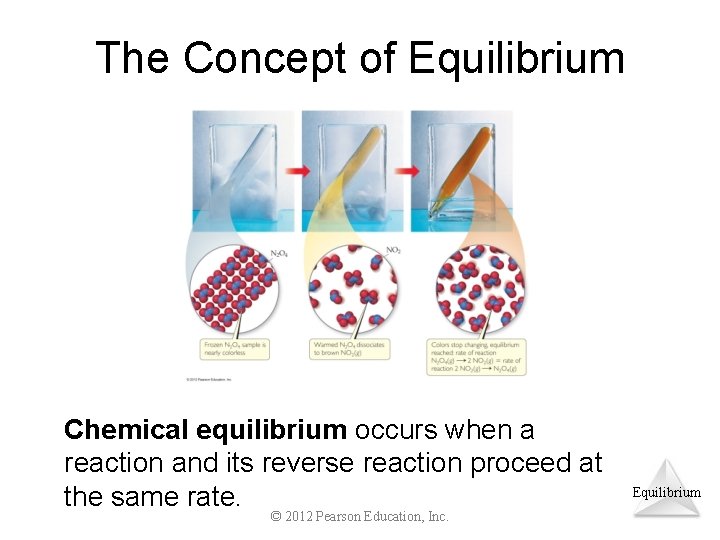
The Concept of Equilibrium Chemical equilibrium occurs when a reaction and its reverse reaction proceed at the same rate. © 2012 Pearson Education, Inc. Equilibrium
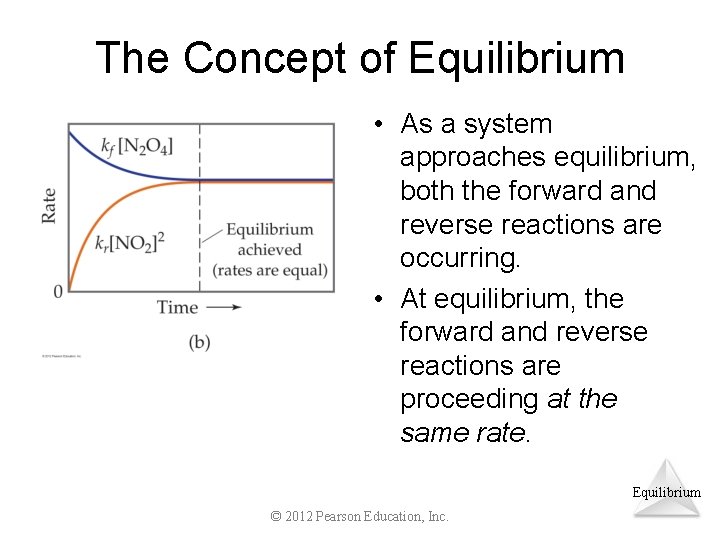
The Concept of Equilibrium • As a system approaches equilibrium, both the forward and reverse reactions are occurring. • At equilibrium, the forward and reverse reactions are proceeding at the same rate. Equilibrium © 2012 Pearson Education, Inc.
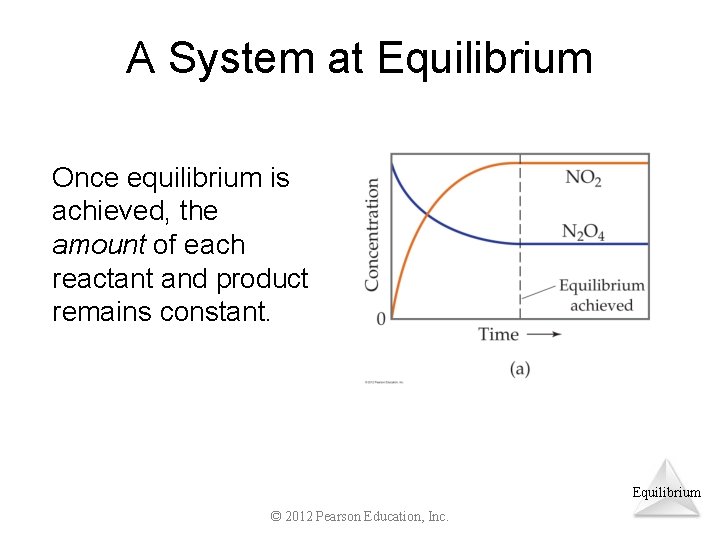
A System at Equilibrium Once equilibrium is achieved, the amount of each reactant and product remains constant. Equilibrium © 2012 Pearson Education, Inc.

Depicting Equilibrium Since, in a system at equilibrium, both the forward and reverse reactions are being carried out, we write its equation with a double arrow: N 2 O 4(g) 2 NO 2(g) Equilibrium © 2012 Pearson Education, Inc.

The Equilibrium Constant Equilibrium © 2012 Pearson Education, Inc.

The Equilibrium Constant • Forward reaction: N 2 O 4(g) 2 NO 2(g) • Rate law: Rate = kf[N 2 O 4] Equilibrium © 2012 Pearson Education, Inc.

The Equilibrium Constant • Reverse reaction: 2 NO 2(g) N 2 O 4(g) • Rate law: Rate = kr[NO 2]2 Equilibrium © 2012 Pearson Education, Inc.
![The Equilibrium Constant • Therefore, at equilibrium Ratef = Rater kf[N 2 O 4] The Equilibrium Constant • Therefore, at equilibrium Ratef = Rater kf[N 2 O 4]](http://slidetodoc.com/presentation_image_h2/fafe9bedf21c2c8ec52da70c7ff15e9c/image-8.jpg)
The Equilibrium Constant • Therefore, at equilibrium Ratef = Rater kf[N 2 O 4] = kr[NO 2]2 • Rewriting this, it becomes kf [NO 2]2 = kr [N 2 O 4] Equilibrium © 2012 Pearson Education, Inc.

The Equilibrium Constant The ratio of the rate constants is a constant at that temperature, and the expression becomes kf [NO 2]2 Keq = = kr [N 2 O 4] Equilibrium © 2012 Pearson Education, Inc.

The Equilibrium Constant • Consider the generalized reaction a. A + b. B c. C + d. D • The equilibrium expression for this reaction would be [C]c[D]d Kc = [A]a[B]b © 2012 Pearson Education, Inc. Equilibrium

The Equilibrium Constant Since pressure is proportional to concentration for gases in a closed system, the equilibrium expression can also be written (PCc) (PDd) Kp = (PAa) (PBb) Equilibrium © 2012 Pearson Education, Inc.

Equilibrium Can Be Reached from Either Direction As you can see, the ratio of [NO 2]2 to [N 2 O 4] remains constant at this temperature no matter what the initial concentrations of NO 2 and N 2 O 4 are. Equilibrium © 2012 Pearson Education, Inc.
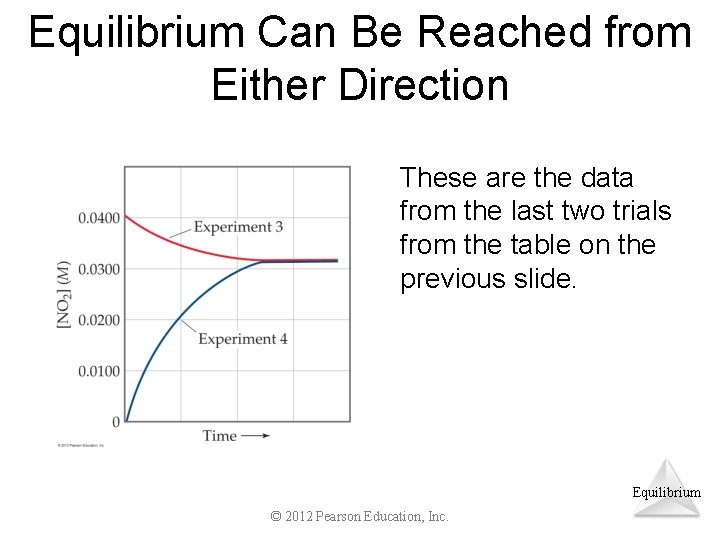
Equilibrium Can Be Reached from Either Direction These are the data from the last two trials from the table on the previous slide. Equilibrium © 2012 Pearson Education, Inc.
What Does the Value of K Mean? • If K>>1, the reaction is product-favored; product predominates at equilibrium. • If K<<1, the reaction is reactant-favored; reactant predominates at equilibrium. Equilibrium © 2012 Pearson Education, Inc.

The Reaction Quotient, Q • Consider the generalized reaction a. A + b. B c. C + d. D • The reaction quotient at any time t for this reaction would be Q= [C]c[D]d [A]a[B]b © 2012 Pearson Education, Inc. Equilibrium

Le Châtelier’s Principle Equilibrium © 2012 Pearson Education, Inc.

Le Châtelier’s Principle “If a system at equilibrium is disturbed by a change in temperature, pressure, or the concentration of one of the components, the system will shift its equilibrium position so as to counteract the effect of the disturbance. ” Equilibrium © 2012 Pearson Education, Inc.

Le Châtelier’s Principle • Changes in pressure and concentrations do not effect K (K is specific to a given temperature; changing T changes K) Equilibrium

Le Châtelier’s Principle • If increase concentration or P of a species, the reaction will shift so as to reestablish equilibrium by consuming part of the added reagent • If decrease concentration or P of a species, the reaction will shift so as to reestablish equilibrium by generating more of the removed reagent Equilibrium

Le Châtelier’s Principle • For H 2(g) + I 2(g) 2 HI(g) assume at equilibrium, [H 2] = 0. 065 M [I 2] = 1. 065 M [HI] = 1. 87 M What happens if increase [H 2] to 1 M? Equilibrium
![Le Châtelier’s Principle • Q = [HI]2/[H 2][I 2] becomes < K as [H Le Châtelier’s Principle • Q = [HI]2/[H 2][I 2] becomes < K as [H](http://slidetodoc.com/presentation_image_h2/fafe9bedf21c2c8ec52da70c7ff15e9c/image-21.jpg)
Le Châtelier’s Principle • Q = [HI]2/[H 2][I 2] becomes < K as [H 2] is now larger than equilibrium value • Need to make Q smaller – increase concentrations of products and reduce concentration of reactant • Reaction net proceeds to right (toward products) until Q = K Equilibrium
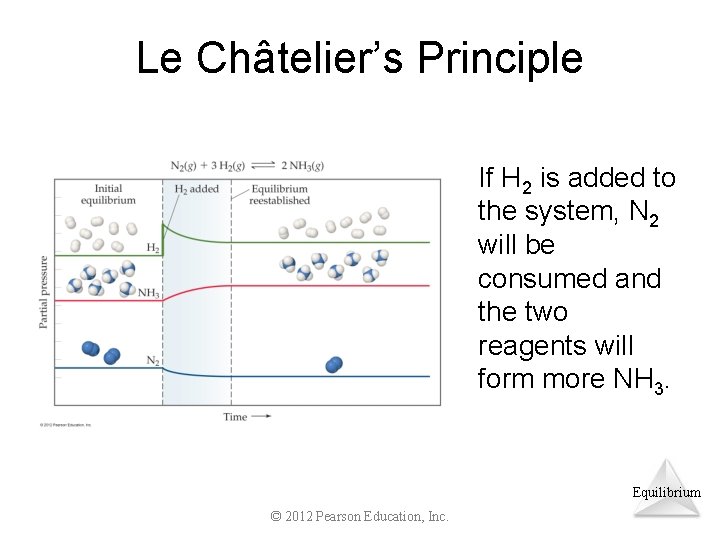
Le Châtelier’s Principle If H 2 is added to the system, N 2 will be consumed and the two reagents will form more NH 3. Equilibrium © 2012 Pearson Education, Inc.
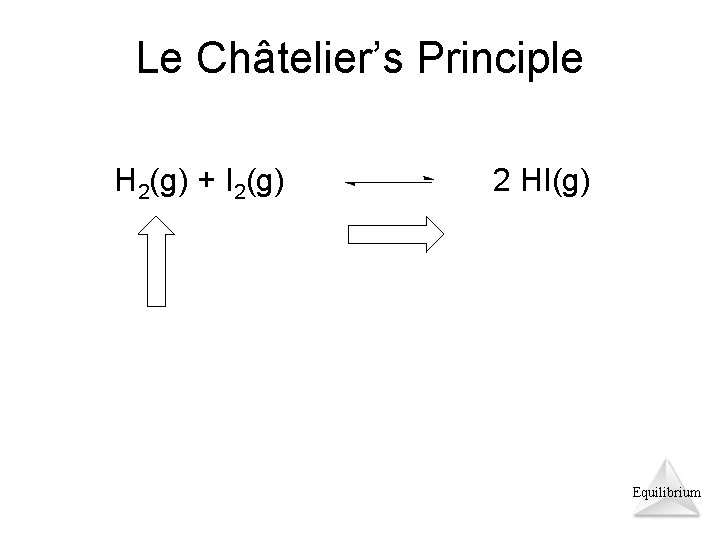
Le Châtelier’s Principle H 2(g) + I 2(g) 2 HI(g) Equilibrium
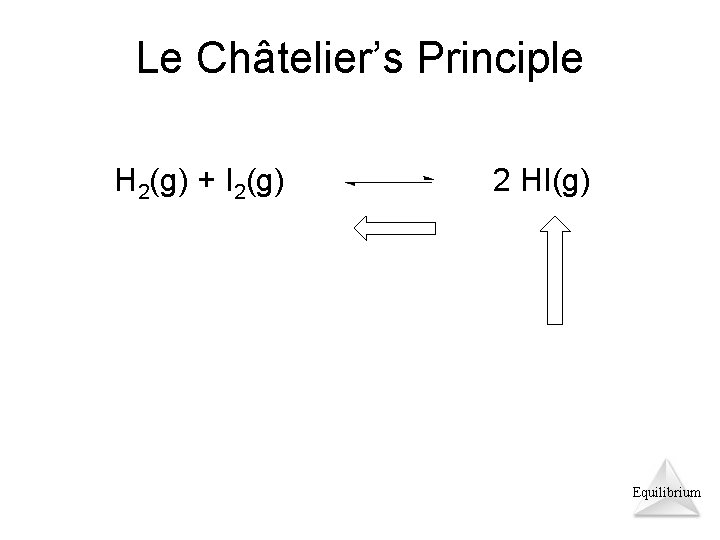
Le Châtelier’s Principle H 2(g) + I 2(g) 2 HI(g) Equilibrium

Changing Pressure and Volume • If a system is at equilibrium and the total pressure is increased by application of an external pressure, the system will shift in the direction that reduces the pressure Equilibrium

Changing Pressure and Volume N 2(g) + 3 H 2(g) 4 moles gas 2 NH 3(g) 2 moles gas • By applying external P, decrease volume of gases • Equilibrium shifts in direction that reduces total number of moles of gas Equilibrium

Changing Pressure and Volume N 2(g) + 3 H 2(g) 4 moles gas 2 NH 3(g) 2 moles gas • By reducing external P, increase volume of gases • Equilibrium shifts in direction that increases total number of moles of gas Equilibrium

Changing Pressure and Volume H 2(g) + I 2(g) 2 mol 2 HI(g) 2 mol What about this reaction? - Changing P will not influence position of equilibrium Equilibrium

Effect of Temperature • Generally K changes as T changes • Changes are small for small changes in T • Consider heat as a reactant or product Equilibrium

Effect of Temperature A+B Heat +A + B C + D + heat exothermic, DH<0 C+D endothermic, DH<0 When heat is added to system, the system shifts in direction that absorbs heat Equilibrium
![Changes in Temperature [Co(H 2 O)6]2+(aq) + 4 Cl(aq) Co. Cl 4(aq) + 6 Changes in Temperature [Co(H 2 O)6]2+(aq) + 4 Cl(aq) Co. Cl 4(aq) + 6](http://slidetodoc.com/presentation_image_h2/fafe9bedf21c2c8ec52da70c7ff15e9c/image-31.jpg)
Changes in Temperature [Co(H 2 O)6]2+(aq) + 4 Cl(aq) Co. Cl 4(aq) + 6 H 2 O(l) Equilibrium © 2012 Pearson Education, Inc.
- Slides: 31