THE COMPTON EFFECT Energy and momentum are conserved









- Slides: 10

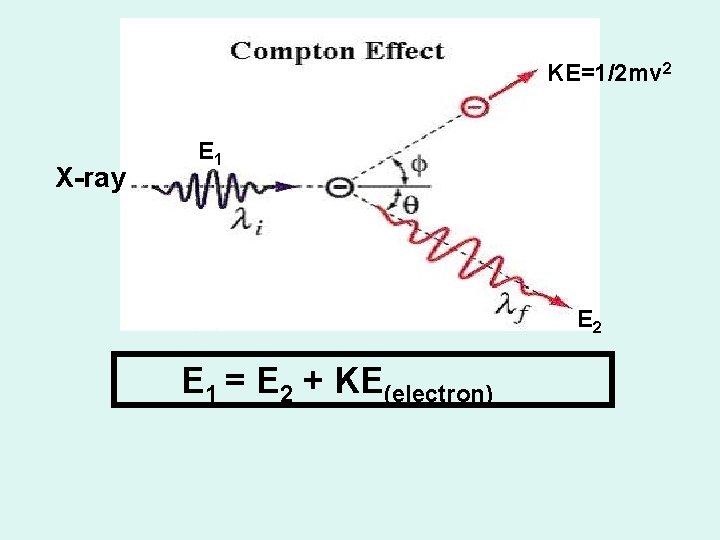
THE COMPTON EFFECT Energy and momentum are conserved when a photon collides with an electron.

KE=1/2 mv 2 X-ray E 1 E 2 E 1 = E 2 + KE(electron)

EXAMPLE A) A Photon having an energy of 3. 8 e. V collides with an electron at rest. The electron has a gain in kinetic energy of 1. 2 e. V, what will be the energy of the emitted photon in e. V and joules? E 1 = E 2 + KE(electron) 3. 8 e. V = E 2 + 1. 2 e. V E 2= 2. 6 e. V E 2 = 4. 16 x 10 -19 J *(1 e. V = 1. 6 x 10 -19 J)*

B) What is the frequency of the scattered light wave and what color is it? E 2 =hf 4. 16 x 10 -19 J= 6. 63 x 10 -34 Js (f) f = 6. 23 x 1014 Hz color = blue

c) What is the velocity of the electron? KE= 1/2 mv 2 1. 2 e. V(1. 6 x 10 -19 J/e. V)= ½ (9. 11 x 10 -31 kg)v 2 1. 92 x 10 -19 J = 4. 56 x 10 -31(v 2) 4. 2 x 1011 = v 2 v= 6. 49 x 105 m/s

De. Broglie Wavelength If light has particle properties then particles must have wave properties λ= h mv ρ λ- wavelength h- Planks constant m- mass v-velocity ρ- momentum

EXAMPLE 1 • What is the De. Broglie wavelength of a 0. 25 kg ball thrown at 20 m/s? λ= h mv = 6. 63 x 10 -34 Js (0. 25 kg)(20 m/s) λ =1. 33 x 10 -34 m *This is too small to observe. *

EXAMPLE 2 What is the De. Broglie wavelength of an electron traveling at 1 x 106 m/s? λ= h mv = 6. 63 x 10 -34 Js (9. 11 x 10 -31 kg)(1 x 106 m/s) λ= 7. 29 x 10 -10 m * Suitable for interference and diffraction and can be observed*

EXAMPLE 3 • What is the momentum of a proton having a De. Broglie wavelength of 2. 81 x 10 -11 m? λ=h/ρ 2. 81 x 10 -11 m = 6. 63 x 10 -34 Js ρ ρ = 2. 36 x 10 -23 kgm/s

Heisenberg Uncertainty Principle • “To study the nature and motion of a moving electrons by bombarding them with photons would change the motion and position of the electron and lead to uncertainty” • You can’t accurately measure both position and momentum of a moving electron at any given time.