THE CHEMISTRY OF LIFE Chapter 2 A Matter














![B. The Importance of Water 1. Properties ] Cohesion - the attraction of water B. The Importance of Water 1. Properties ] Cohesion - the attraction of water](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-19.jpg)
![] High heat of vaporization - a lot of heat is required to evaporate ] High heat of vaporization - a lot of heat is required to evaporate](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-20.jpg)
![3. Acids & Bases ] Acids - substances that add H+ to a solution. 3. Acids & Bases ] Acids - substances that add H+ to a solution.](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-21.jpg)

![2. Lipids F contain C, H, O [ C >> O] F do not 2. Lipids F contain C, H, O [ C >> O] F do not](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-26.jpg)
![Proteins have a 3 -dimensional shape (conformation): ] primary (1 o) structure - amino Proteins have a 3 -dimensional shape (conformation): ] primary (1 o) structure - amino](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-31.jpg)


- Slides: 35

THE CHEMISTRY OF LIFE Chapter 2

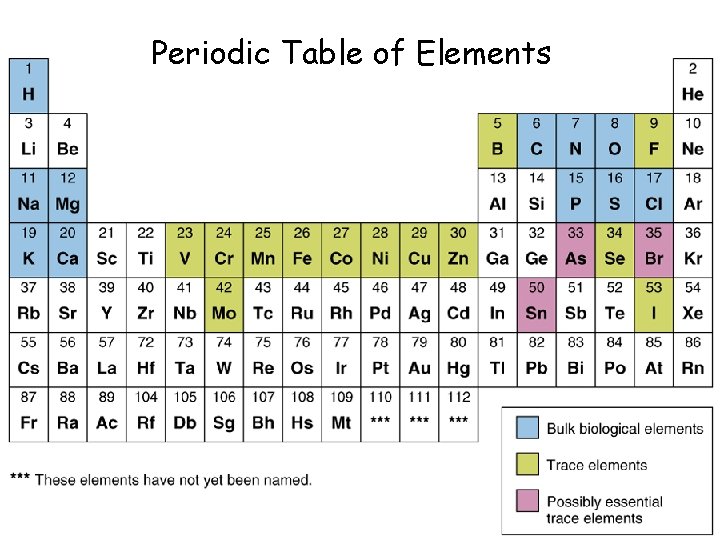
A. Matter Material that takes up space. 1. Elements Pure chemical substances composed of atoms. F Examples? F How many elements exist? F How many of these elements are essential to life?

Periodic Table of Elements

2. Atom The smallest “piece” of an element that retains the characteristics of that element. Composed of 3 subatomic particles: F Protons F Neutrons F Electrons

Characteristics of Subatomic Particles


Atomic number # protons in nucleus of an atom (establishes identity of the atom) Since most atoms are electrically neutral, atomic number indicates # of electrons as well. Atomic mass # protons plus # neutrons in nucleus of an atom

How can we determine the number of neutrons in an atom? # neutrons = atomic mass - atomic # ] Determine # neutrons in a carbon atom (atomic mass = 12; atomic # = 6). # neutrons = 12 - 6 = 6 Do all carbon atoms have the same number of protons? Do all carbon atoms have the same number of neutrons?

Isotopes Atoms having the same number of protons, but differing numbers of neutrons. Ex. Carbon isotopes F carbon 12 (12 C) 6 neutrons F carbon 13 (13 C) 7 neutrons F carbon 14 (14 C) 8 neutrons

Periodic table information on carbon: Atomic mass given in table is average mass of all the element’s isotopes.

3. Compound A pure substance formed when atoms of different elements bond. The number of atoms of each element is written as a subscript. Examples: F CO 2 carbon dioxide F H 2 O water F CH 4 methane F C 6 H 12 O 6 glucose

4. Molecule Smallest piece of a compound that retains characteristics of that compound. The number of molecules is written as a coefficient. Examples: F 4 CO 2 4 molecules of carbon dioxide F 2 C 6 H 12 O 6 2 molecules of glucose F 6 O 2 6 molecules of oxygen

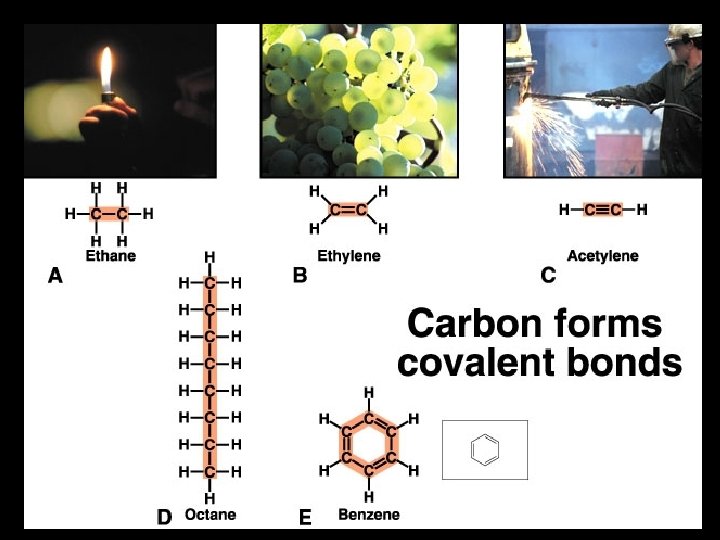
5. Chemical Bonds Type of bond formed is determined by the number of valence electrons in the interacting atoms [octet rule]. a) Covalent bonds - form when atoms share electron pairs. F strongest type of bond F tend to form when atoms have 3, 4 or 5 valence electrons F can be nonpolar or polar


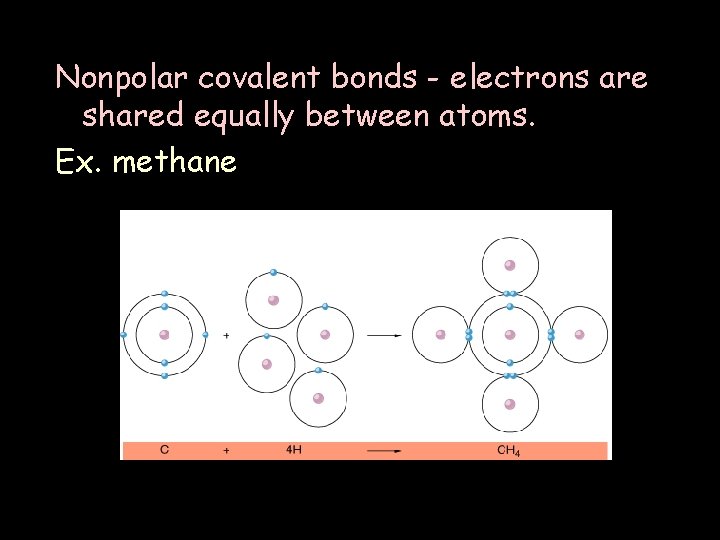
Nonpolar covalent bonds - electrons are shared equally between atoms. Ex. methane

Polar covalent bonds - electrons are drawn more strongly to 1 atom’s nucleus than the other. Form when less electronegative atoms bond with more highly electronegative atoms. Ex. water

b) Ionic bonds - form when oppositely charged ions are attracted to each other. F weaker than covalent bonds F atoms with 1, 2 or 3 valence electrons give up electrons to atoms with 7, 6 or 5 valence electrons F form salts Ex. Na. Cl

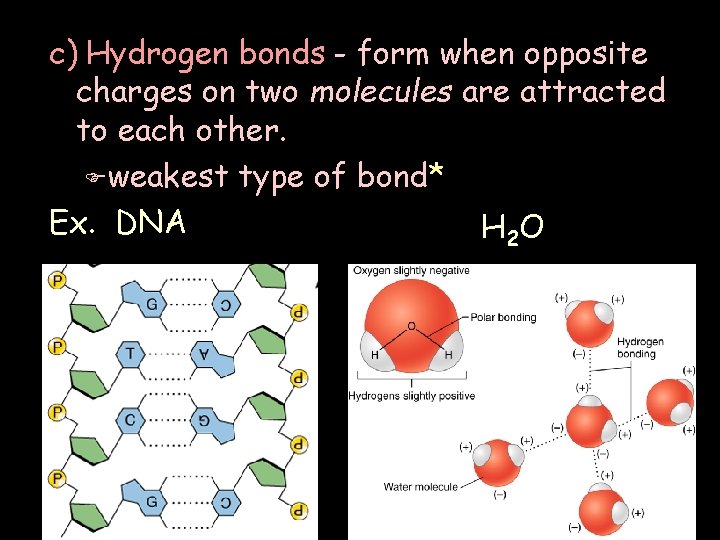
c) Hydrogen bonds - form when opposite charges on two molecules are attracted to each other. F weakest type of bond* Ex. DNA H 2 O
![B The Importance of Water 1 Properties Cohesion the attraction of water B. The Importance of Water 1. Properties ] Cohesion - the attraction of water](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-19.jpg)
B. The Importance of Water 1. Properties ] Cohesion - the attraction of water molecules for each other. ] Adhesion - the attraction of water molecules for other compounds. ] High heat capacity – takes a great deal of heat to raise the temperature of water.
![High heat of vaporization a lot of heat is required to evaporate ] High heat of vaporization - a lot of heat is required to evaporate](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-20.jpg)
] High heat of vaporization - a lot of heat is required to evaporate water. ] Exists as solid, liquid or gas - solid (ice) is less dense than liquid. 2. Solutions A solution is a mixture of one or more solutes dissolved in a solvent. If solvent is water, then it is an aqueous solution. Water is a strong solvent because it separates charged atoms or molecules.
![3 Acids Bases Acids substances that add H to a solution 3. Acids & Bases ] Acids - substances that add H+ to a solution.](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-21.jpg)
3. Acids & Bases ] Acids - substances that add H+ to a solution. ] Bases - substances that remove H+ from solution. p. H scale is measure of acidity/alkalinity based on H+ concentration.

C. Major Organic Molecules that contain carbon in combination with hydrogen. 1. Carbohydrates F contain C, H & O [ C O] F function to store energy & provide support F building blocks (monomers) are monosaccharides

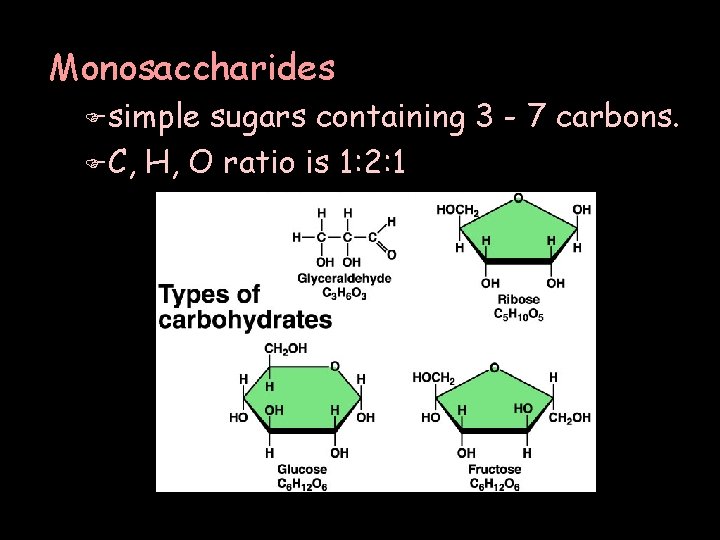
Monosaccharides F simple sugars containing 3 - 7 carbons. F C, H, O ratio is 1: 2: 1

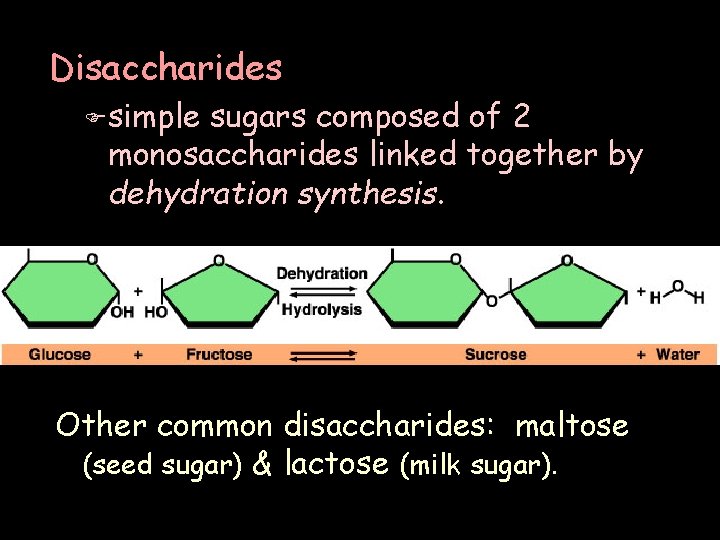
Disaccharides F simple sugars composed of 2 monosaccharides linked together by dehydration synthesis. Other common disaccharides: maltose (seed sugar) & lactose (milk sugar).

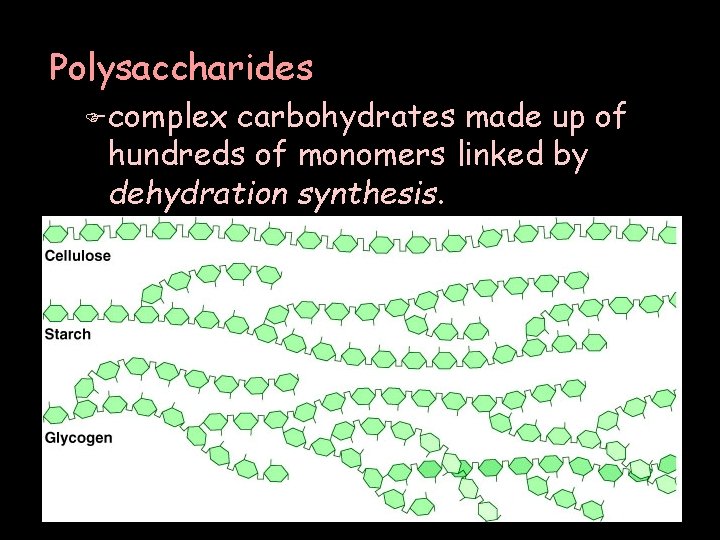
Polysaccharides F complex carbohydrates made up of hundreds of monomers linked by dehydration synthesis.
![2 Lipids F contain C H O C O F do not 2. Lipids F contain C, H, O [ C >> O] F do not](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-26.jpg)
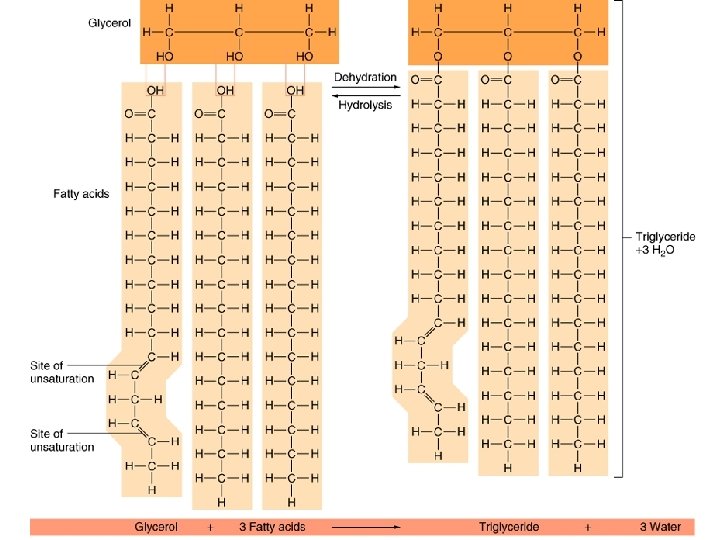
2. Lipids F contain C, H, O [ C >> O] F do not dissolve in water Triglycerides (fats) F composed of glycerol linked to 3 fatty acid chains by dehydration synthesis. F function to cushion organs, as insulation & in long-term energy storage (adipose tissue).


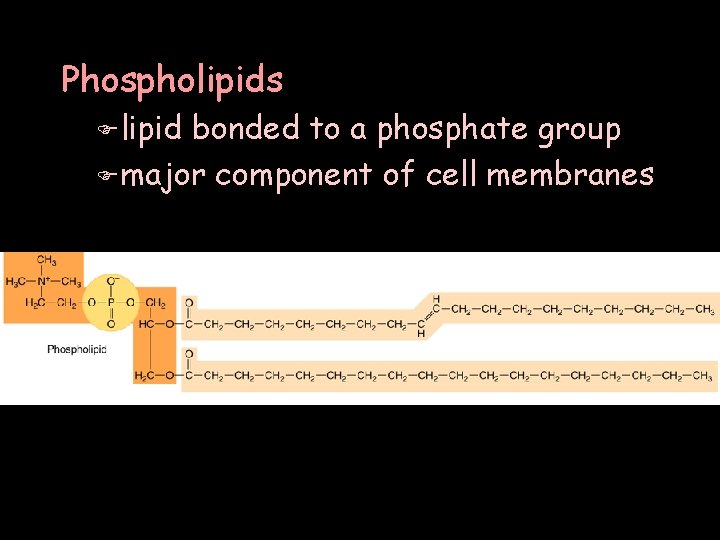
Phospholipids F lipid bonded to a phosphate group F major component of cell membranes

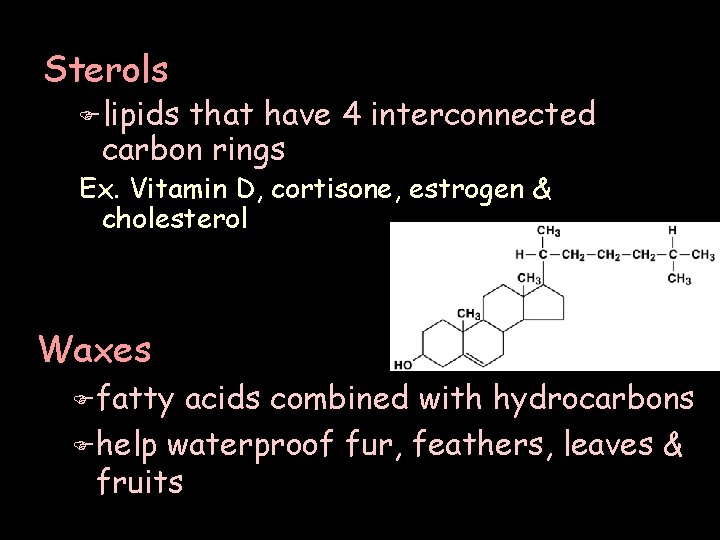
Sterols F lipids that have 4 interconnected carbon rings Ex. Vitamin D, cortisone, estrogen & cholesterol Waxes F fatty acids combined with hydrocarbons F help waterproof fur, feathers, leaves & fruits

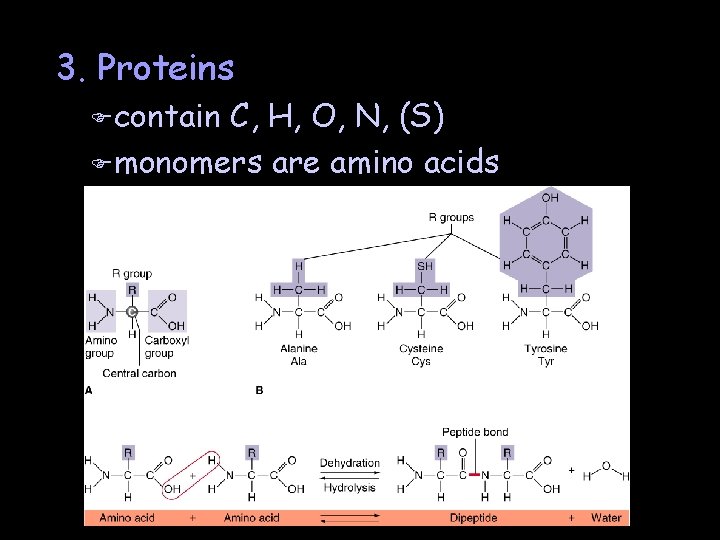
3. Proteins F contain C, H, O, N, (S) F monomers are amino acids
![Proteins have a 3 dimensional shape conformation primary 1 o structure amino Proteins have a 3 -dimensional shape (conformation): ] primary (1 o) structure - amino](https://slidetodoc.com/presentation_image_h2/b621c9687d768fb1b993545e1985ba66/image-31.jpg)
Proteins have a 3 -dimensional shape (conformation): ] primary (1 o) structure - amino acid sequence of polypeptide chain ] secondary (2 o) structure - coiling & folding produced by hydrogen bonds ] tertiary (3 o) structure - shape created by interactions between R groups ] quarternary (4 o) structure - shape created by interactions between two or more polypeptides

Examples: F antibodies F hemoglobin F insulin & glucagon F keratin F fibrin & thrombin F spider silk (strongest natural fiber known) F enzymes (maltase, pepsin, lipase)

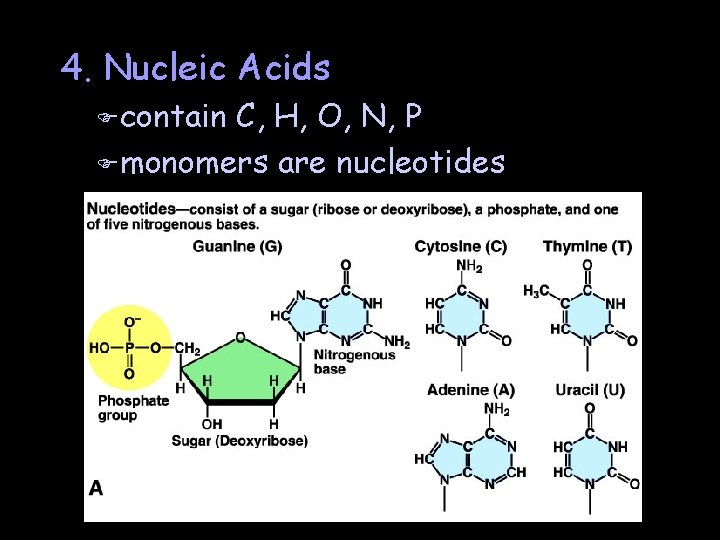
4. Nucleic Acids F contain C, H, O, N, P F monomers are nucleotides

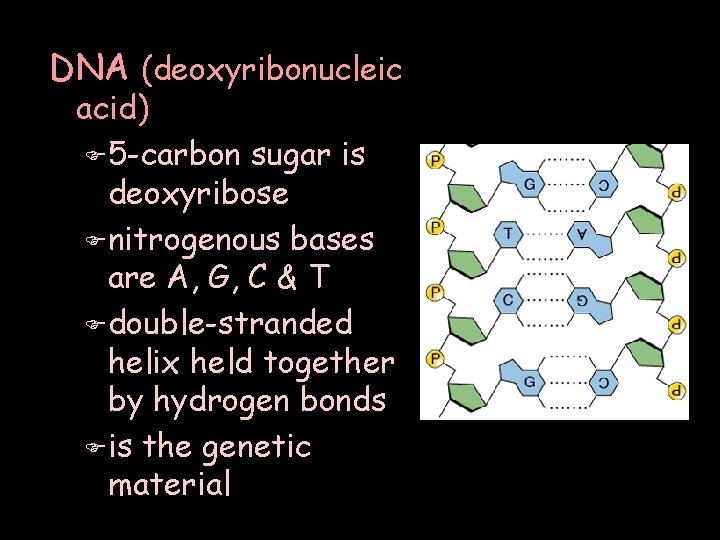
DNA (deoxyribonucleic acid) F 5 -carbon sugar is deoxyribose F nitrogenous bases are A, G, C & T F double-stranded helix held together by hydrogen bonds F is the genetic material

RNA (ribonucleic acid) F 5 -carbon sugar is ribose F nitrogenous bases are A, G, C & U F single-stranded F enables information in DNA to be expressed