The Chemical Name and Formula of an Ionic











- Slides: 12

The Chemical Name and Formula of an Ionic Compound • The name of an ionic compound = positive ion + negative ion-ide. w For example, an ionic compound forms between magnesium and oxygen. § +ve ion is 1 st § -ve ion is 2 nd § Add -ide to the end of the name to form magnesium oxide. Magnesium oxide is used as a drying agent. • Ionic formulas are based on the ions of the atoms involved. w For example, what is the name of Ca 3 N 2? § Ca, the cation, is calcium. § N, the anion, is nitrogen. § Drop the end of the anion & add -ide. (c) Mc. Graw Hill Ryerson 2007

The Chemical Name and Formula of an Ionic Compound (continued) • Writing formulas for ionic compounds: w + charges balance out - charges. w The ratio of +ve: -ve charges gives the proper formula. § The ratio is always written in reduced form. w For ex. , what is the formula for magnesium phosphide? § magnesium is Mg 2+ & phosphorous is P 3 Calcium oxide, also known as “quicklime” was once produced by cooking limestone in ancient kilns. – § LCM of 2 and 3 is 6 § 3 Mg 2+ ions and 2 P 3– ions (6 +ve & 6 –ve charges) § = Mg 3 P 2 (c) Mc. Graw Hill Ryerson 2007

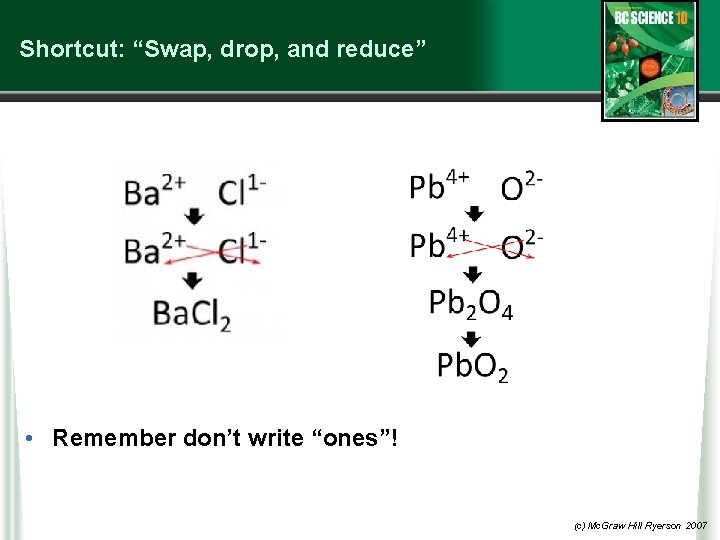
Shortcut: “Swap, drop, and reduce” • Remember don’t write “ones”! (c) Mc. Graw Hill Ryerson 2007

You try! w What is the formula for calcium oxide? § calcium is Ca 2+ & oxygen is O 2– § 1 Ca 2+ ion and 1 O 2– ions § Ca 2 O 2 § which is reduced and written as Ca. O

Formula of an Ionic Compound with a Multivalent Metal • Some transitional metals are multivalent, (= have more than one ion form) w the most common form of the ion is listed on top w When naming, Roman numerals are used following the +ve ion to indicate which ion was used. w Ex) What is the formula manganese (III) sulphide? § This manganese is Mn 3+ and sulfur is S 2– § LCM of 3 & 2 is 6 § 2 Mn 3+ ions and 3 S 2– ions § = Mn 2 S 3 (c) Mc. Graw Hill Ryerson 2007

You try! What is the name for Ti. F 4? § titanium can be Ti 4+ or Ti 3+ & fluorine is F– § Look at the formula & you can work out that it is Ti 4+ § 1 Ti 4+ ion and 4 F– ions § Titanium (IV) fluoride

Polyatomic Ions • Polyatomic ions, are made up of several atoms joined together with covalent bonds. w The whole group has a + or – charge, not the individual atoms. (c) Mc. Graw Hill Ryerson 2007

• What is the formula of sodium sulphate? Na+ and SO 42– Na 2 SO 4 • What is the formula for Calcium nitrate? Ca 2+ and NO 3 Ca(NO 3)2 * Note the brackets around NO 3 show there are two of the ions present • What is the name of the compound KCl. O? K+ = potassium. Cl. O– = hypochlorite potassium hypochlorite (c) Mc. Graw Hill Ryerson 2007

Names and Formulas of Covalent Compounds • a. k. a. molecules w made up of two or more non-metals. w Names may reveal the components, but often don’t. w Subscripts mean something different in covalent compounds § Ionic compounds subscripts show the smallest whole-number ratio between the ions in the compound. § Covalent molecules have subscripts that show the actual number of atoms in the molecule. w Ex) ethanol? § C 2 H 6 O, a name that must be memorized or looked up w Ex) when C O needed. H ? 12 22 11 § Sucrose, also called table sugar. (c) Mc. Graw Hill Ryerson 2007

Naming Binary Covalent Compounds • Binary covalent compounds (two nonmetal atoms) use prefixes. w may have many or few atoms sharing electrons. § Ex) CH 4 = methane and C 25 H 52 = candle wax w Prefixes are often used before the atom name to indicate the number of atoms in the molecule. § CO = carbon monoxide, CO 2 = carbon dioxide (c) Mc. Graw Hill Ryerson 2007

Examples: • What is the name of the molecule Si 3 P 6? w Trisilicon hexaphosphide • What is the chemical formula for the molecule trinitrogen tetrachloride? w N 3 Cl 4

Comparing Ionic and Covalent Compounds • To determine whether a compound is ionic or covalent: 1. Examine the formula. • Ionic compounds start with a metal or the ammonium ion. • Covalent compounds start with a non-metal. 2. If the compound is covalent: • If it is binary & doesn’t start with H, use the prefix system • Note: If there are >2 different elements, or it starts with H, there is probably a different, simpler name 3. If the compound is ionic: • Check the metal to see if it is multivalent (add a Roman numeral if it is) • If it ends with a single non-metal, naming will just end in -ide. • If it ends in a polyatomic ion, look up the name/formula. (c) Mc. Graw Hill Ryerson 2007