The Chemical Building Blocks of Life Chapter 3

The Chemical Building Blocks of Life Chapter 3

Outline • Biological Molecules – Macromolecules § Carbohydrates v Transport and Storage § Lipids v Fats and Phospholipids § Proteins v Structure and Denaturation § Nucleic Acids v DNA and RNA 2

Biological Molecules • The framework of biological molecules consists of carbon bonded to other carbon molecules, or other types of atoms. – Hydrocarbons consist of carbon and hydrogen. § Covalent bonds store considerable energy. 3

Biological Molecules • • • Functional groups – specific groups of atoms attached to carbon backbones § retain definite chemical properties Macromolecules. Other than water, biomolecules fall into 4 classes – proteins – nucleic acids – lipids – carbohydrates 4
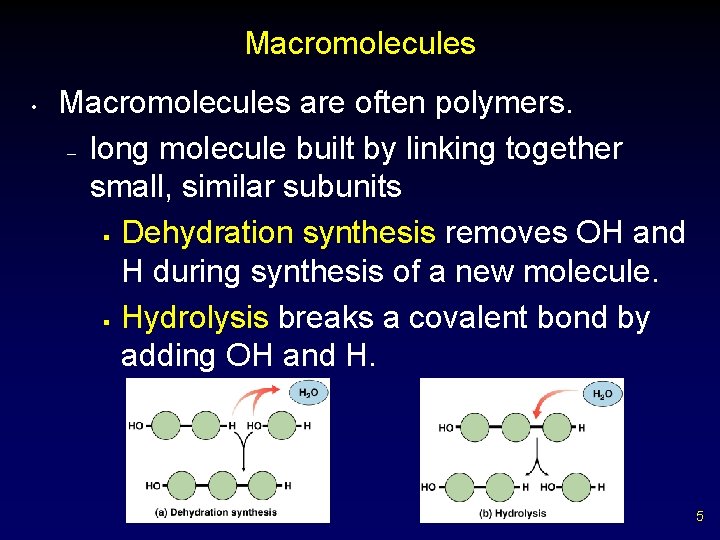
Macromolecules • Macromolecules are often polymers. – long molecule built by linking together small, similar subunits § Dehydration synthesis removes OH and H during synthesis of a new molecule. § Hydrolysis breaks a covalent bond by adding OH and H. 5

Table 3. 1 6
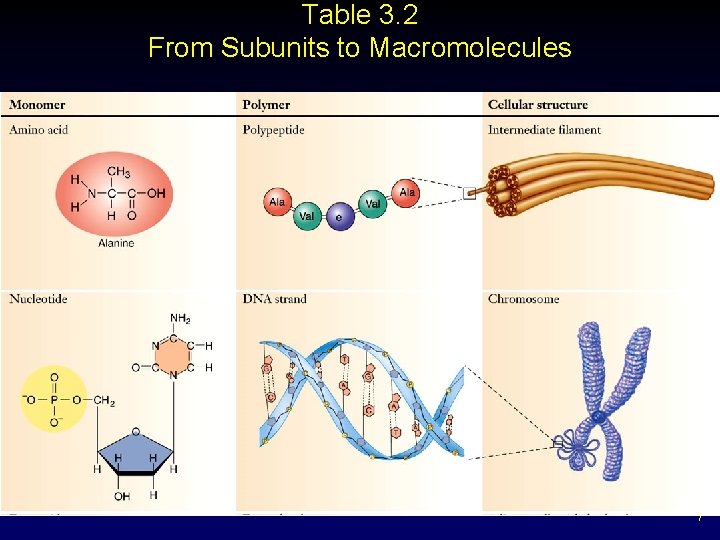
Table 3. 2 From Subunits to Macromolecules 7
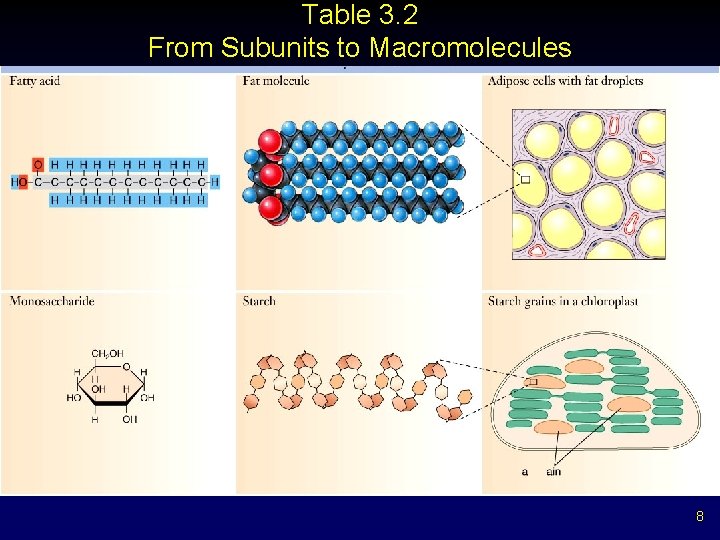
Table 3. 2 From Subunits to Macromolecules 8

Carbohydrates • • • Molecules made of C and Hydrates (H and O) Energy packed molecules used by most life Can also be structural molecules in life empirical formula is Cx(H 2 O)y many named with ending “ose” ( ex: sucrose) 3 basic types: – – – monosaccharides disaccharides polysaccharides 9

Carbohydrates • Carbohydrates are loosely defined as molecules that contain carbon, hydrogen, and oxygen in a 1: 2: 1 ratio. – monosaccharides - simple sugars – disaccharides - two monosaccharides joined by a covalent bond – polysaccharides - macromolecules made of monosaccharide subunits § isomers - alternative forms of the same substance 10

Monosaccharides • • • Are the “simple sugars” – composed of (3 -7) linked carbons – chains of carbons with a hydroxyl most common = 6 -----> hexoses (C 6 H 12 O 6) – examples of hexoses are glucose and fructose many are Isomers: – same empirical formula but different structural formula – gives a different “sweetness” and solubility 11
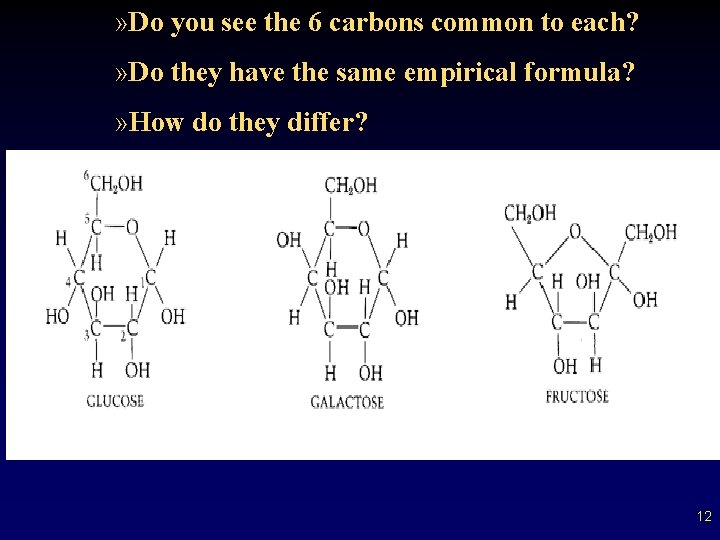
» Do you see the 6 carbons common to each? » Do they have the same empirical formula? » How do they differ? 12

Disaccharides • • More complex sugars (6 -14) carbons in a chain or ring (C 12 H 22 O 11) includes table sugar (sucrose) and lactose (milk) created by uniting two monosaccharides through dehydration synthesis (Gee, where have you seen those words ? !? !? ) 13
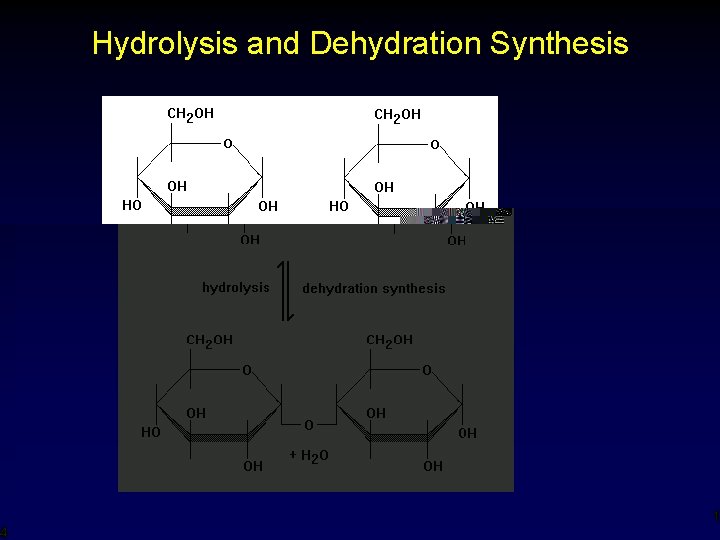
Hydrolysis and Dehydration Synthesis 14

Polysaccharides • • • Huge!!!! 100’s of mono’s linked Animal form = glycogen – storage of energy contained in glucose – insulin promotes conversion to glycogen for liver – unique structural polysaccharide is called chitin; hydroxyl group is replaced by an amino group Plant forms = starch and cellulose – starch is for energy storage 15 – cellulose is a structural form - not soluble

Carbohydrate Transport and Storage • • Transport disaccharides – Humans transport glucose as a simple monosaccharide. – Plants transform glucose into a disaccharide transport form. Storage polysaccharides – plant polysaccharides formed from glucose - starches § most is amylopectin 16

Structural Carbohydrates • • Cellulose - plants – alpha form or beta form of ring Chitin - arthropods and fungi – modified form of cellulose 17

Lipids • • • Lipids are loosely defined as groups of molecules that are insoluble in water. – fats and oils C, H and O primarily (not exclusively) non-polar therefore insoluble in water 18

More Lipids Phospholipids form the core of all biological membranes. – composed of three subunits § glycerol § fatty acid § phosphate group 19

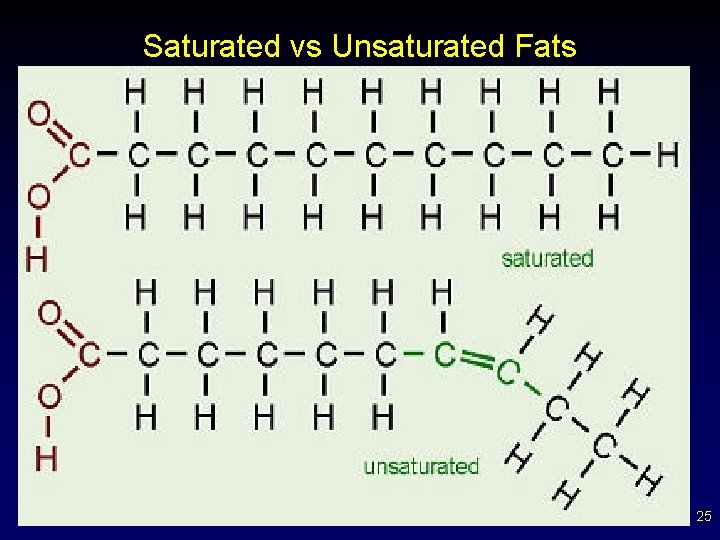
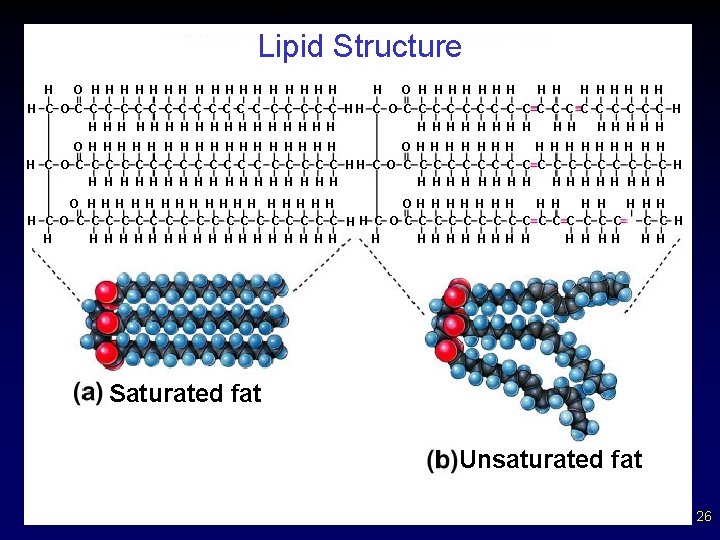
• • Fats and Other Lipids Fats consist a of glycerol molecule with three attached fatty acids (triglyceride / triglycerol). Saturated fats - all internal carbon atoms are bonded to at least two hydrogen atoms – carbons are single bonded - solids at room temp – contains maximum number of H atoms ( saturated with “H” ) – straight chains – not preferred in diet medically (beef, pork) 20

Fats and Other Lipids Unsaturated fats - at least one double bond between successive carbon atoms – carbons in double or triple bonds, less H - liquids – tend to be “kinked” chains – if many double or triple bonds -----> polyunsaturated – diet preferred (fish and vegetables) • Polyunsaturated - contains more than one double bond § usually liquid at room temperature Why would unsaturated fats be more ideal in your diet then saturated fats? Why are trans fats bad? (structurally) • 21
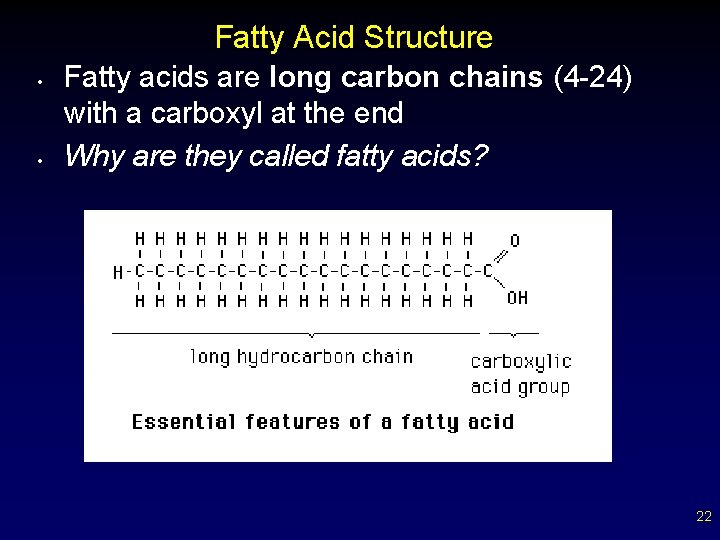
Fatty Acid Structure • • Fatty acids are long carbon chains (4 -24) with a carboxyl at the end Why are they called fatty acids? 22
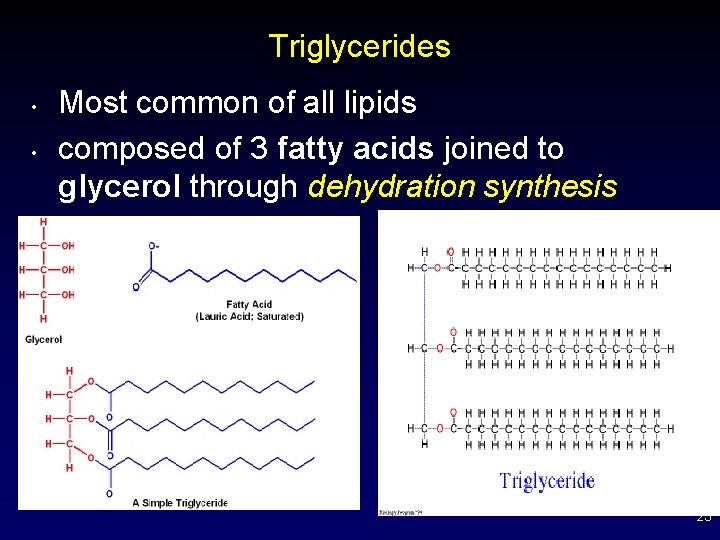
Triglycerides • • Most common of all lipids composed of 3 fatty acids joined to glycerol through dehydration synthesis 23
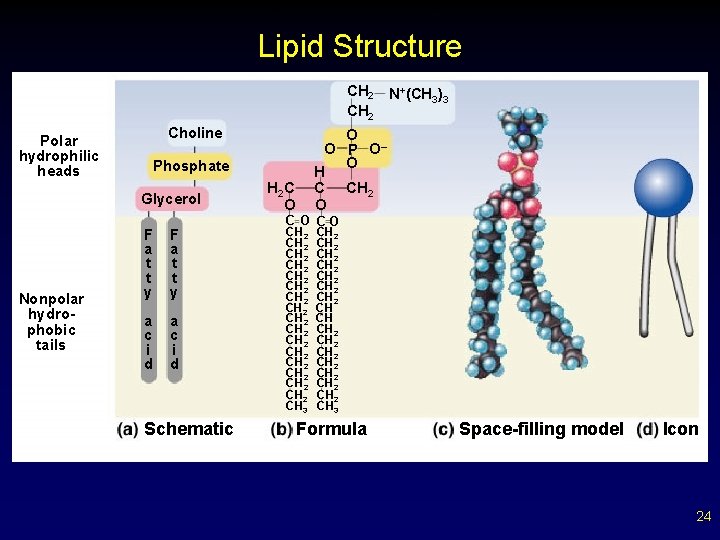
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Lipid Structure CH 2 Polar hydrophilic heads Choline Nonpolar hydrophobic tails O O P O– O H C CH 2 O Phosphate Glycerol F a t t y a c i d Schematic N+(CH 3)3 H 2 C O CO CH 2 CH 2 CH 2 CH 2 CH 3 CH 2 CH 2 CH CH CH 2 CH 2 CH 3 Formula Space-filling model Icon 24
Saturated vs Unsaturated Fats 25
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Fig. 3. 22(TE Art) Lipid Structure H O H H H H H H H H C O C C C C C HH C O C C C C C H H H H H H H H H O H H H H H H H H H C O C C C C C C C C C H H H H H H H H H O H H H H H O H H H H C O C C C C C C C C C H H H H H H H H HH H H Saturated fat Unsaturated fat 26

Fats as Energy Storage Molecules • Fats, on average, yield about 9 kcal per gram versus 4 kcal per gram for carbohydrates. – Animal fats are saturated while most plant fats are unsaturated. § Consumption of excess carbohydrates leads to conversion into starch, glycogen, or fats for future use. 27

Fats and Lipids in the Body • • • Formed from excess glucose consumed – floats in blood to be made into adipose tissue Provide reserves e supply More energy than carbs/proteins: § § § • • • 1 g carb -----> 4. 3 kcal 1 g prot-------> 4. 6 kcal 1 g lipid -----> 9. 0 kcal Used after glycogen reserves gone (12 hours) other functions: cushions, protects, insulates tremendous diversity in types 28
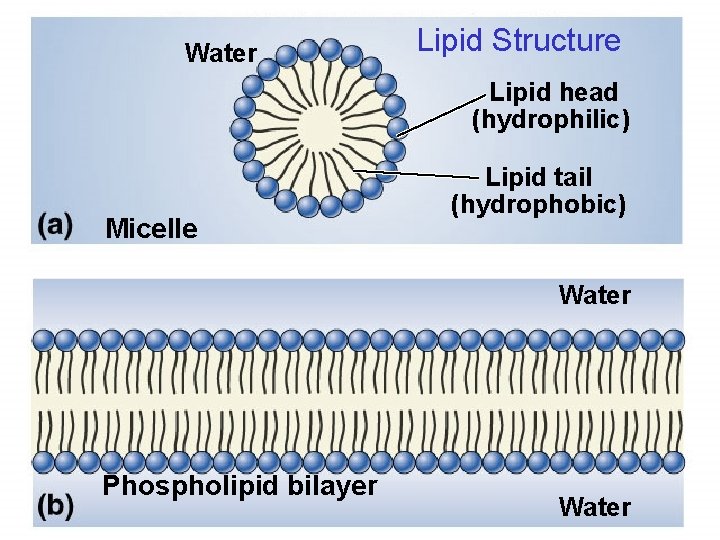
Fig. Water 3. 20(TELipid Art) Structure Lipid head (hydrophilic) Micelle Lipid tail (hydrophobic) Water Phospholipid bilayer Water 29

• • • Hydrogenation of Polyunsaturates Corn oil is a liquid polyunsaturated fat Many double carbon bonds Want to solidify it? – Heat oil – add pressurized hydrogen gas and catalyst – carbons replaced by hydrogen – produce a solid called “partially hydrogenated vegetable oil” or PHVO – called “trans fats” - once thought to be good but now known to increase cholesterol levels - 30

Additional Lipid Types • • • Phosolipids – 2 fatty acids plus a phosphate – part of cell membranes – transports other lipids in blood Steroids – 4 connecting carbon rings + functional group – cell membranes, hormones – cholesterol (carried by other fats - too much indicates excess fats in blood) Waxes – extremely hydrophobic 31
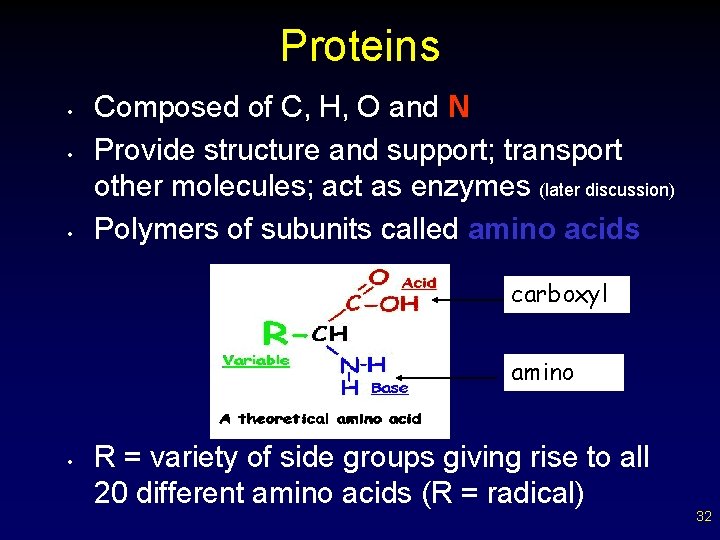
Proteins • • • Composed of C, H, O and N Provide structure and support; transport other molecules; act as enzymes (later discussion) Polymers of subunits called amino acids carboxyl amino • R = variety of side groups giving rise to all 20 different amino acids (R = radical) 32
Proteins • Protein functions: – enzyme catalysis – defense – transport – support – motion – regulation – storage 33
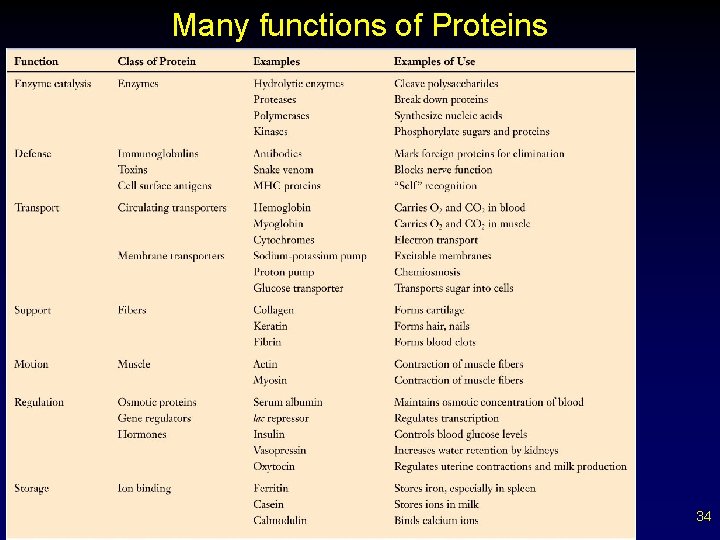
Many functions of Proteins 34

Amino Acids • contain an amino group (-NH 2), a carboxyl group (-COOH) and a hydrogen atom, all bonded to a central carbon atom – twenty common amino acids grouped into five classes based on side groups § nonpolar amino acids § polar uncharged amino acids § aromatic amino acids § special-function amino acids 35
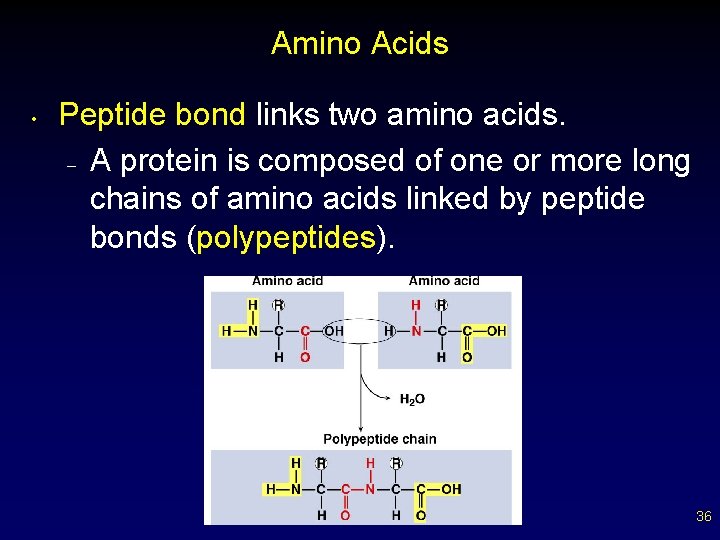
Amino Acids • Peptide bond links two amino acids. – A protein is composed of one or more long chains of amino acids linked by peptide bonds (polypeptides). 36

Protein Structure • • Solubility/charge of proteins related to nature of side groups (R groups) Combining amino acids – OH removed from one carboxyl – H removed from amino group of 2 nd a. a. – this is a dehydration synthesis reaction 37

Protein Structure – – – this is a dehydration synthesis reaction results in bond between C of one and N of second amino acid called a peptide bond compound called a dipeptide many amino acids = polypeptide (polymer) tremendous number of polypeptides 38
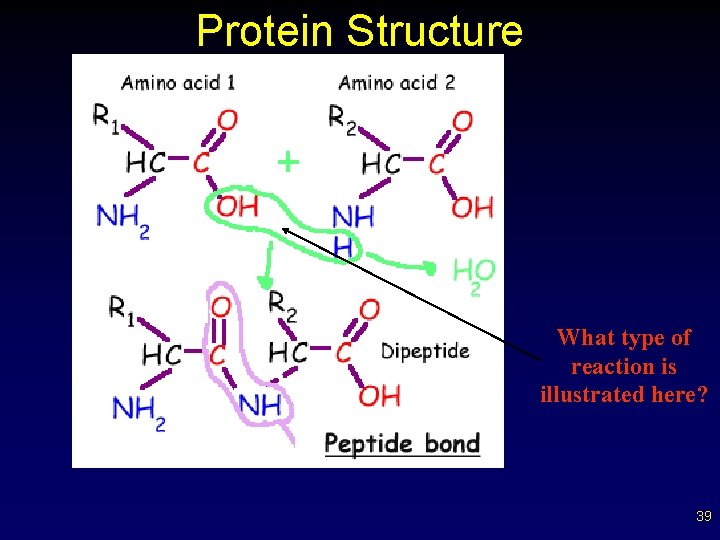
Protein Structure What type of reaction is illustrated here? 39

Protein Structure • Protein function is determined by its shape. – Protein structure § primary - specific amino acid sequence § secondary - folding of amino acid chains § motifs - folds or creases v supersecondary structure 40

Protein Structure • Linear order of amino acids ----> primary structure – • • • (order determined by information in DNA) bonds cause the protein to fold, kink or pleat and results in ----> secondary structure protein undergoes more complex twisting and turning producing ----> tertiary structure some proteins composed of two or more separable polypeptide chains to produce ----> quaternary structure – many metabolic enzymes are 4 to 6 intertwined proteins or protein complex 41

Protein Structure – – – tertiary - final folded shape of globular protein domains - functional units § Large portions (sequence) of or on a protein quaternary - forms when two or more polypeptide chains associate to form a functional protein 42
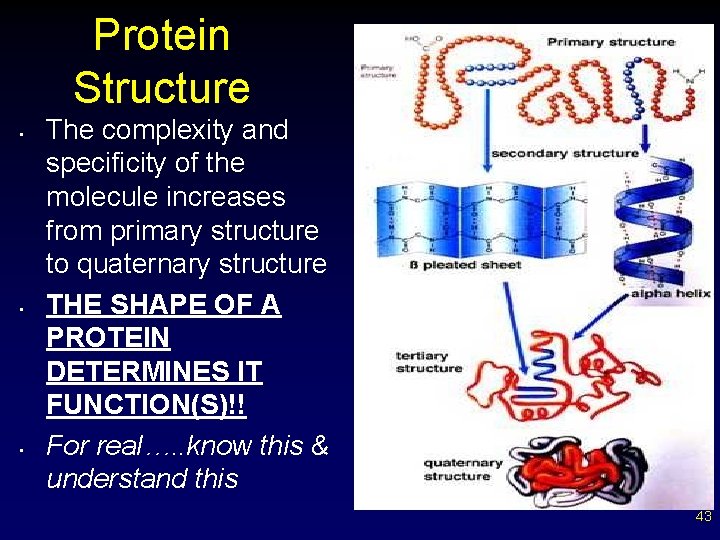
Protein Structure • • • The complexity and specificity of the molecule increases from primary structure to quaternary structure THE SHAPE OF A PROTEIN DETERMINES IT FUNCTION(S)!! For real…. . know this & understand this 43

Chaperonins • Special chemicals involved with helping proteins assume final shape – Assist proteins as they go from linear chains to some 3 -dimensional entity – inhibit misfolded states – refold as a kind of proofreading function – maintain unfolded state for protein import/export 44
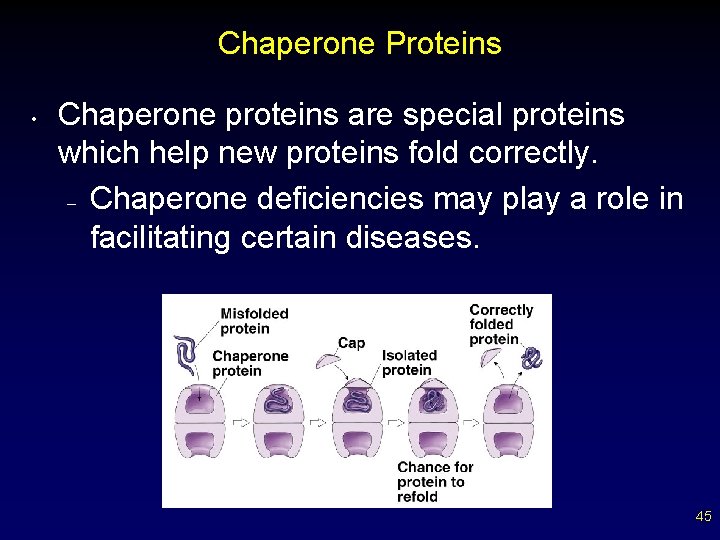
Chaperone Proteins • Chaperone proteins are special proteins which help new proteins fold correctly. – Chaperone deficiencies may play a role in facilitating certain diseases. 45

Protein Structure • Denaturation refers to the process of changing a protein’s shape. distortion of protein structure due to heat, detergents, p. H – molecule loses ability to do its ordinary job For proteins, structure determines function – • • usually rendered biologically inactive § salt-curing and pickling used to preserve food Why do you cook meat before you eat it? What evolutionary advantage would cooking meat have to a species? 46
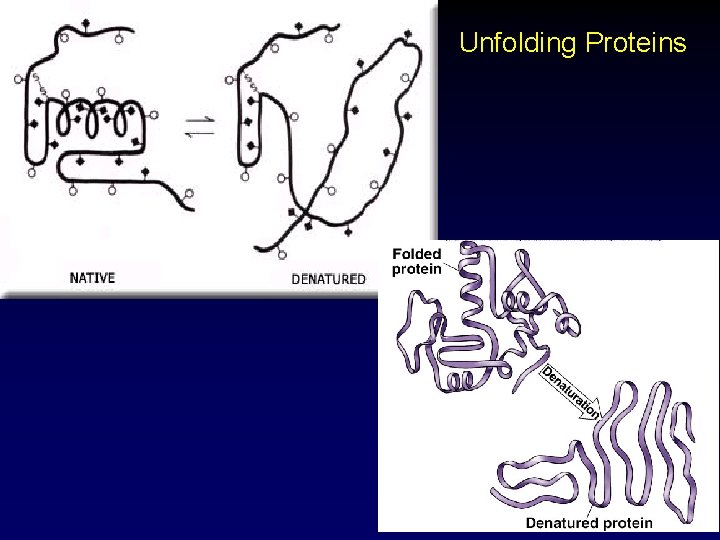
Unfolding Proteins 47
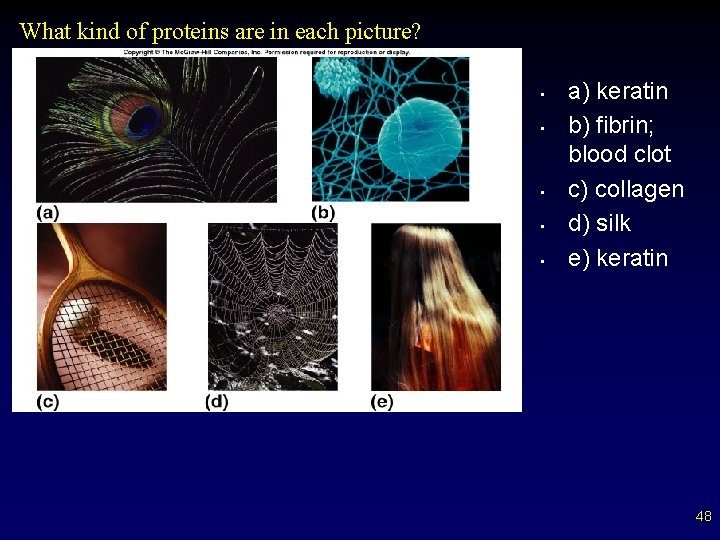
What kind of proteins are in each picture? • • • a) keratin b) fibrin; blood clot c) collagen d) silk e) keratin 48

Information Molecules • The biochemical activity of a cell depends on the production of a large number of very specifically sequenced proteins (You are what you are because of your proteins!) – Nucleic Acids direct this production! § § § Information storage devices (how to make proteins) Templates to produce exact copies of themselves Two kinds found in living organisms DNA (deoxyribonucleic acid) v RNA (ribonucleic acid) v 49

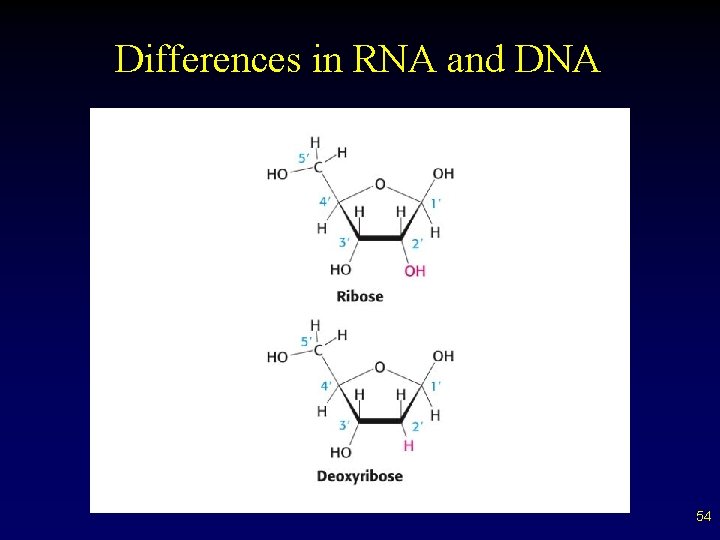
Basic Nucleic Acid Structure • • Composed of units called nucleotides Each nucleic acid made of a: – – – Five carbon sugar (look at picture for subtle difference in RNA and DNA on next slide) Phosphate Nitrogen containing base (two types) § Purines (Adenine and Guanine – often “A” and “G”) § Pyrimidines (Cytosine, Thymine and Uracil – “C”, “T” and “U”) 50
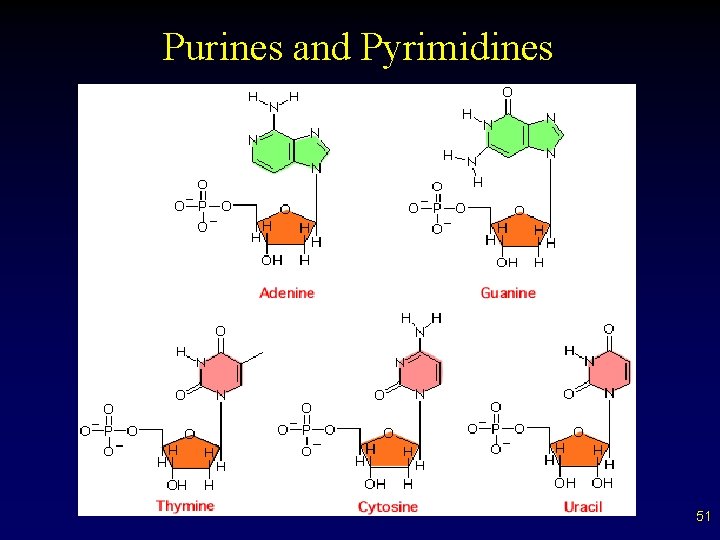
Purines and Pyrimidines 51
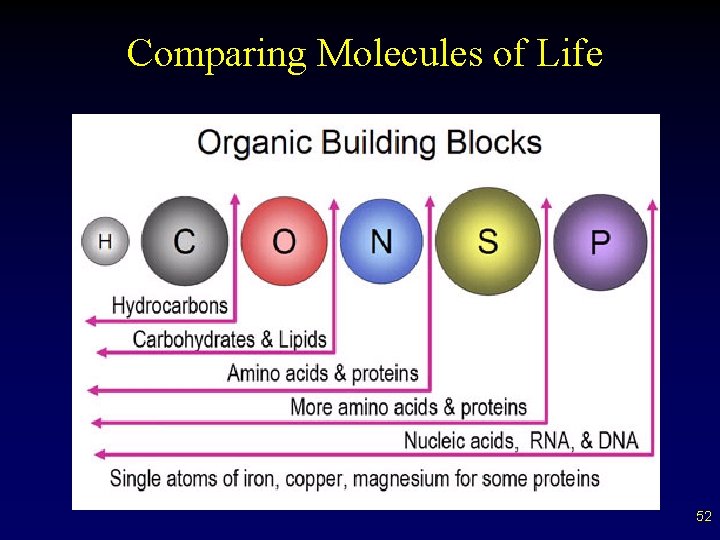
Comparing Molecules of Life 52

Nucleic Acids • • Deoxyribonucleic Acid (DNA) – Encodes information used to assemble proteins. Ribonucleic Acid (RNA) – Reads DNA-encoded information to direct protein synthesis. 53
Differences in RNA and DNA 54

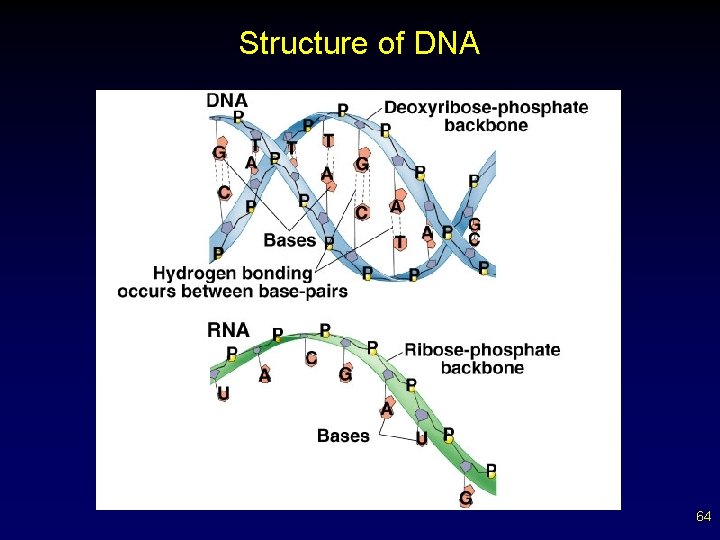
DNA • • Found in ALL living things “Blueprints” for an organism’s development and growth (hereditary information) Exists as a twisted staircase, or double helix The sides of the ladder are alternating sugars and phosphates The steps are composed of base pairs Bases are complementary paired (A-T, G-C) Bases held together by hydrogen bonds from attracted base pairs (weak bonds) 55
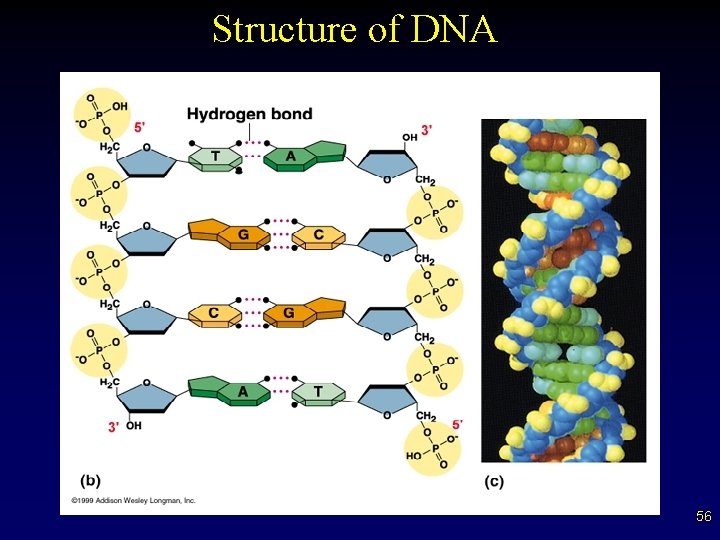
Structure of DNA 56
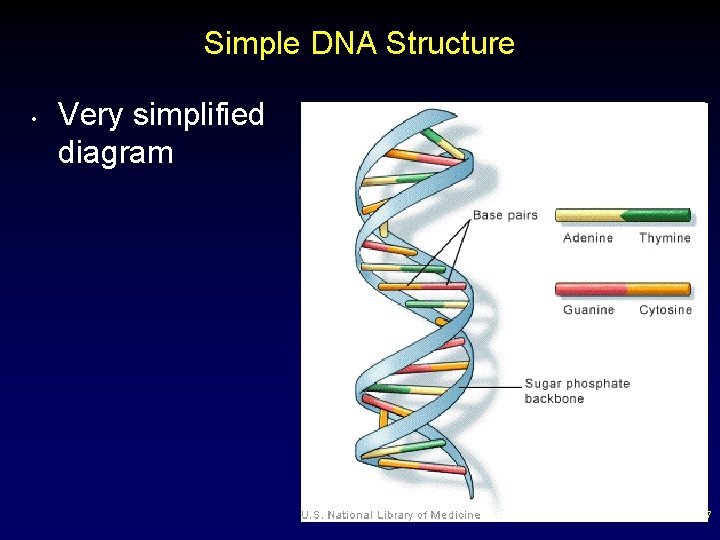
Simple DNA Structure • Very simplified diagram 57
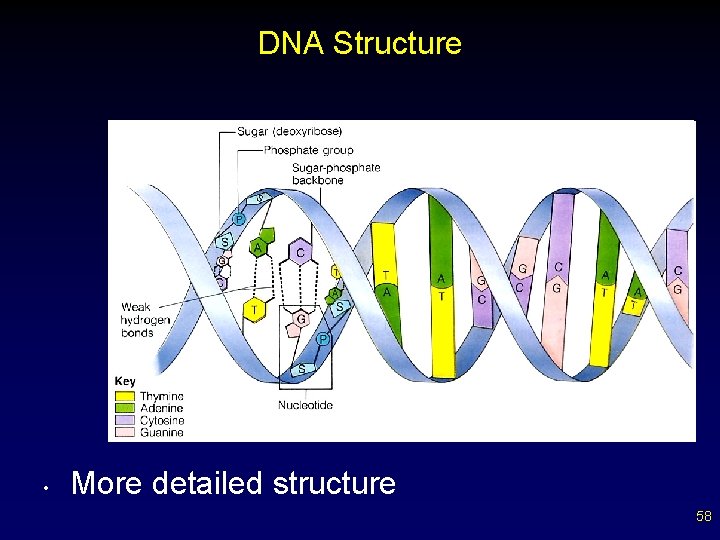
DNA Structure • More detailed structure 58
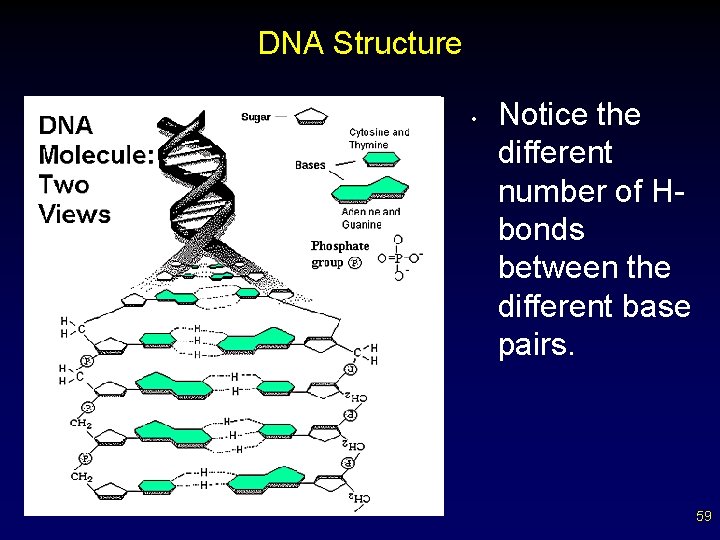
DNA Structure • Notice the different number of Hbonds between the different base pairs. 59
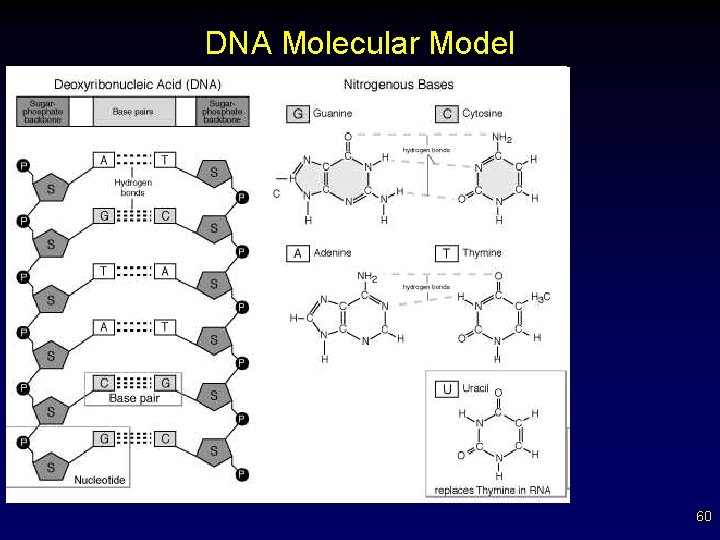
DNA Molecular Model 60
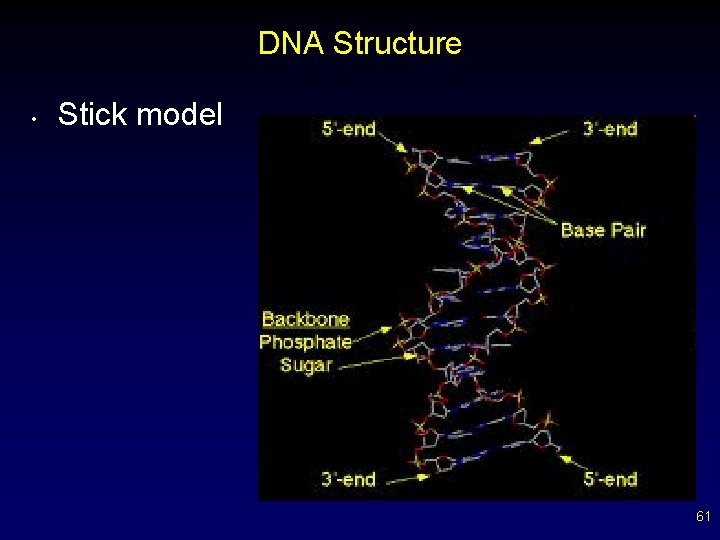
DNA Structure • Stick model 61

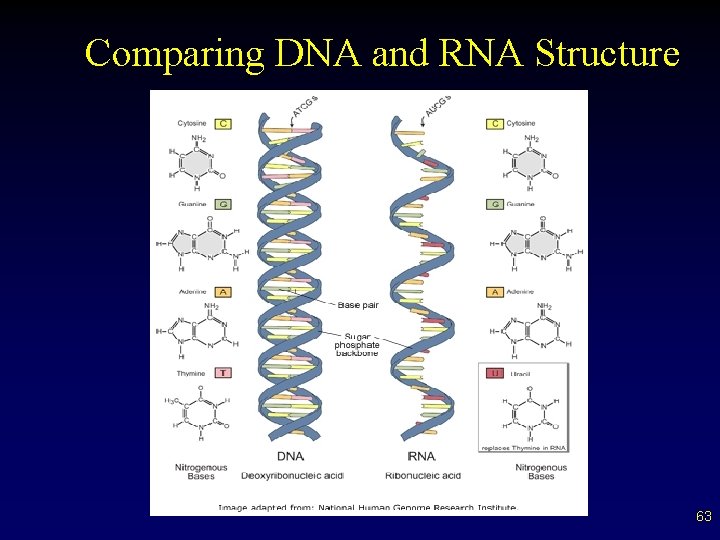
RNA • • • Carries message of DNA from nucleus to cytoplasm of cell on how to put together a protein (construction foreman) Several ‘types’ exist Different from DNA in a few ways – Sugar is ribose, not deoxyribose – No thymine present, uracil replaces it – Is half a ladder, a single stranded molecule 62
Comparing DNA and RNA Structure 63
Structure of DNA 64

Nucleic Acid Structure • Nucleic acids are composed of long polymers of repeating subunits, nucleotides. – five-carbon sugar – phosphate – nitrogenous base § purines v adenine and guanine § pyrimidines v cytosine, thymine, and uracil 65

Nucleic Acid Structure • • DNA exists as double-stranded molecules. – double helix – complementary base pairing § hydrogen bonding RNA exists as a single stand. – contains ribose instead of deoxyribose – contains uracil in place of thymine 66
- Slides: 66