The Cell Membrane Structure Function RECALL PLASMA MEMBRANE
The Cell Membrane Structure & Function

RECALL: PLASMA MEMBRANE (IS) a “gate keeper“ AKA CELL MEMBRANE made of a double- Glycoproteins Embedded Proteins Phospholipid Bilayer of sheets of phospholipids between the inside and outside of the cell. (DOES) Regulates what enters and leaves the cell. It also aids in the protection and support of the cell.
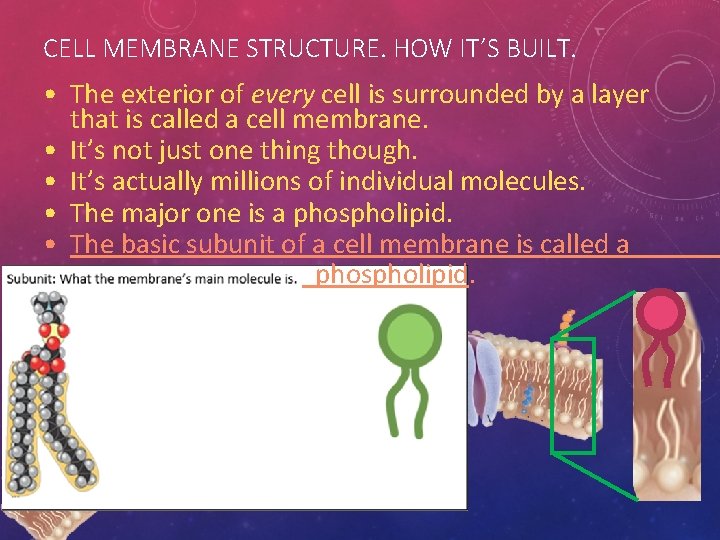
CELL MEMBRANE STRUCTURE. HOW IT’S BUILT. • The exterior of every cell is surrounded by a layer that is called a cell membrane. • It’s not just one thing though. • It’s actually millions of individual molecules. • The major one is a phospholipid. • The basic subunit of a cell membrane is called a phospholipid.
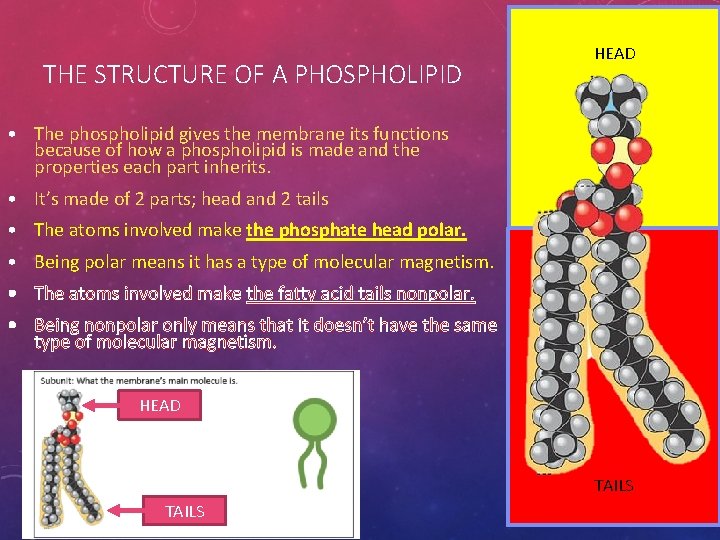
THE STRUCTURE OF A PHOSPHOLIPID HEAD • The phospholipid gives the membrane its functions because of how a phospholipid is made and the properties each part inherits. • It’s made of 2 parts; head and 2 tails • The atoms involved make the phosphate head polar. • Being polar means it has a type of molecular magnetism. • The atoms involved make the fatty acid tails nonpolar. • Being nonpolar only means that it doesn’t have the same type of molecular magnetism. HEAD TAILS
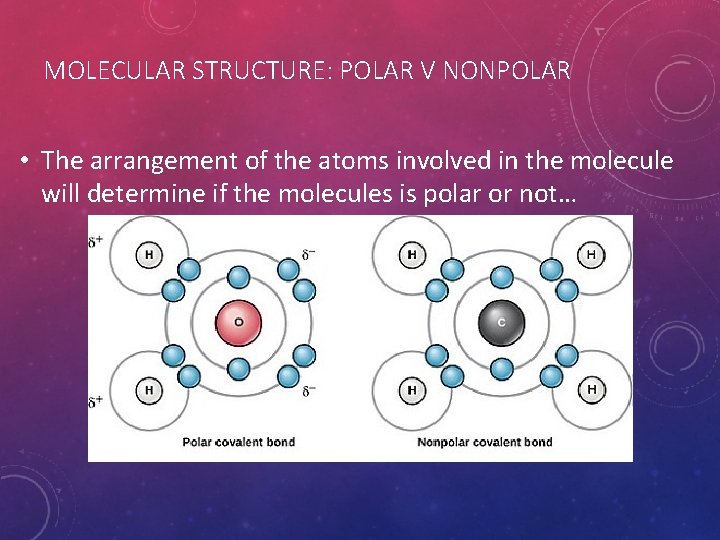
MOLECULAR STRUCTURE: POLAR V NONPOLAR • The arrangement of the atoms involved in the molecule will determine if the molecules is polar or not…

POLAR V NON-POLAR • Being a polar molecule is not a very complicated concept if you understand how magnets work. • If you try to put 2 magnets together they interact 1 of 2 ways… • Either they snap together or they push each other apart. • This has to do with which “poles” are facing each other. • Opposites attract and likes repel, so • if the (+) is facing the (-) they’ll stick… (like c and d) • If the (+) is facing the (+) or the (-) is facing the (-) they’ll repel… (like a & b) • This is how molecules work too. • Based on how a molecule is formed from the atoms involved, the resulting molecule can have either an unequal sharing of electrons (having a negative charge) or an equal sharing of electrons. • If the sharing is unequal, the resulting molecule will be more negative where there are more electrons most of the time. (like water) • By default the other side will be positive. • If the molecule has an equal sharing of electron the molecule will end up being nonpolar. (like methane and most other hydrocarbons, oils, and fats)
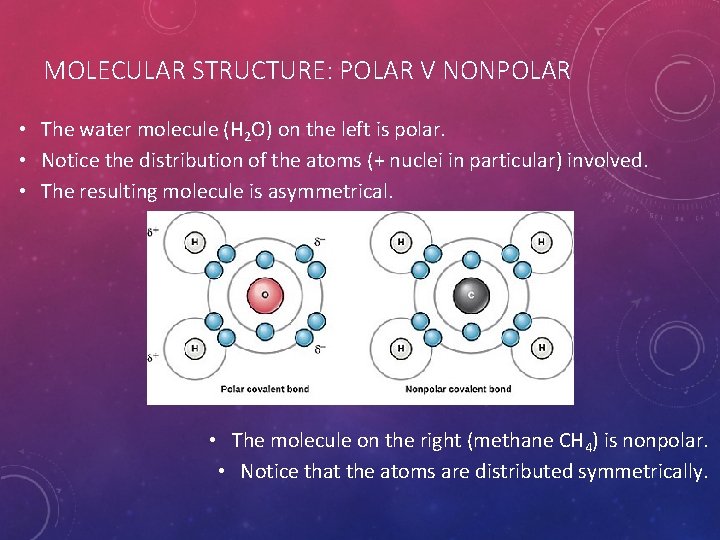
MOLECULAR STRUCTURE: POLAR V NONPOLAR • The water molecule (H 2 O) on the left is polar. • Notice the distribution of the atoms (+ nuclei in particular) involved. • The resulting molecule is asymmetrical. • The molecule on the right (methane CH 4) is nonpolar. • Notice that the atoms are distributed symmetrically.
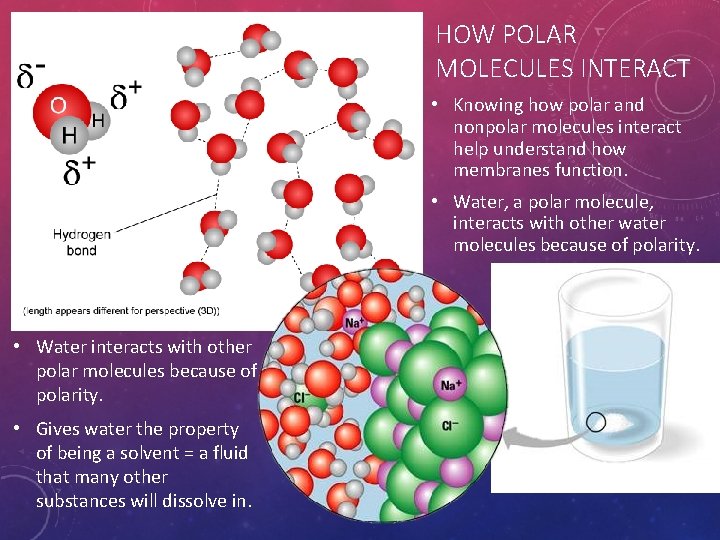
HOW POLAR MOLECULES INTERACT • Knowing how polar and nonpolar molecules interact help understand how membranes function. • Water, a polar molecule, interacts with other water molecules because of polarity. • Water interacts with other polar molecules because of polarity. • Gives water the property of being a solvent = a fluid that many other substances will dissolve in.

HOW NONPOLAR MOLECULES INTERACT • Lipids will dissolve in lipids just like polar substances will dissolve in polar substances. • This is the foundation of what’s known as being “lipid-soluble”. • Most cosmetic products utilize this phenomenon. • Lotions, spray-tan, etc. • Most food flavorings utilize this phenomenon as well. • Flavored oils, sautés, • Since the membrane is predominately lipids (fats) it can harness this feature as part of it’s function. • More on that in a moment…

HOW POLAR AND NON-POLAR SUBSTANCES INTERACT… • However, when mixed, polar and nonpolar substances don’t like to interact. • For example, when you mix water with oil, they separate… the lighter less dense oil floats on top of the heavier water. NONPOLAR LAYER
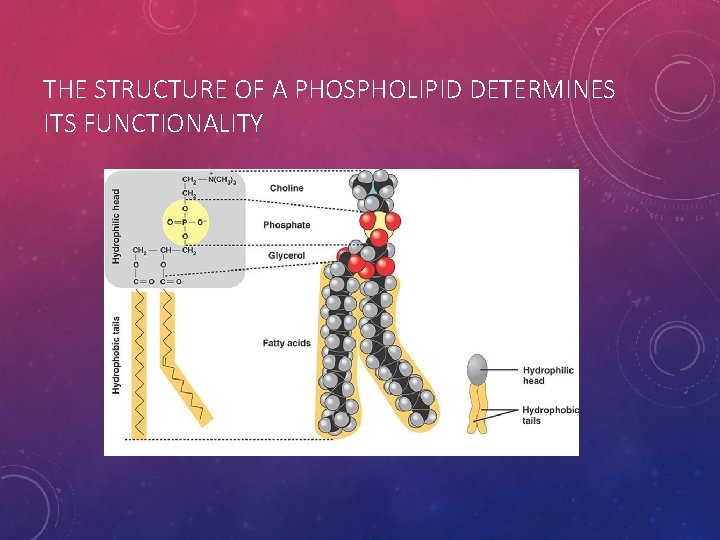
THE STRUCTURE OF A PHOSPHOLIPID DETERMINES ITS FUNCTIONALITY
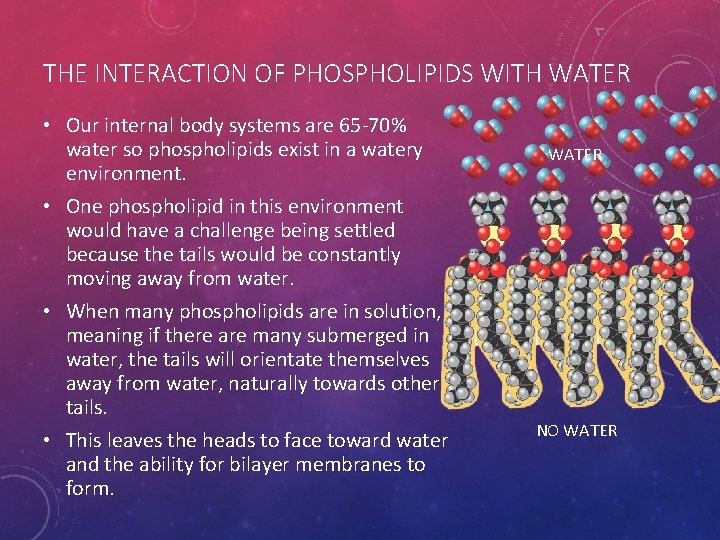
THE INTERACTION OF PHOSPHOLIPIDS WITH WATER • Our internal body systems are 65 -70% water so phospholipids exist in a watery environment. WATER • One phospholipid in this environment would have a challenge being settled because the tails would be constantly moving away from water. • When many phospholipids are in solution, meaning if there are many submerged in water, the tails will orientate themselves away from water, naturally towards other tails. • This leaves the heads to face toward water and the ability for bilayer membranes to form. NO WATER
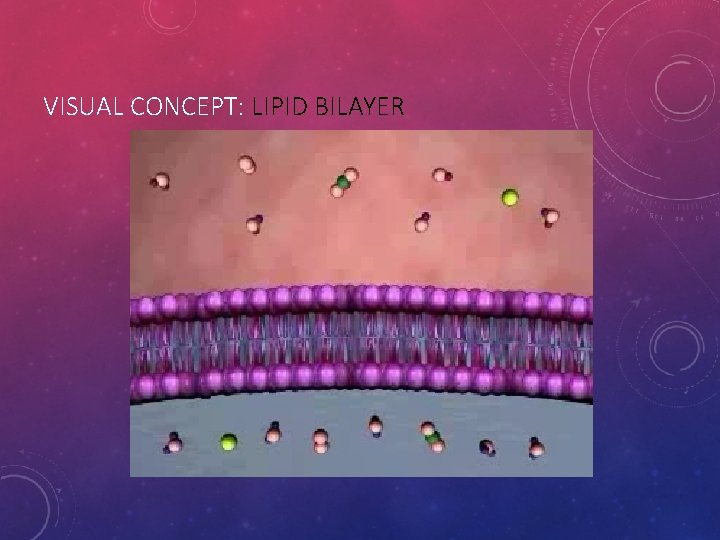
VISUAL CONCEPT: LIPID BILAYER

CELL MEMBRANES, A. K. A. LIPID BILAYER Membrane Structure = LIPID BILAYER • The basic structure of a biological membrane is composed of two layers of phospholipids • Because there is water inside and outside the cell, the phospholipids will naturally form a double layer called the lipid bilayer. • The nonpolar tails, repelled by water, make up the interior of the lipid bilayer. • The polar heads are attracted to the water, so they point toward the surfaces of the lipid bilayer. • One layer of polar heads faces inside the cell, the cytoplasm • The other layer is in contact with the cell’s immediate surroundings outside. • The outside of the cell is known as the “Extra-cellular matrix”
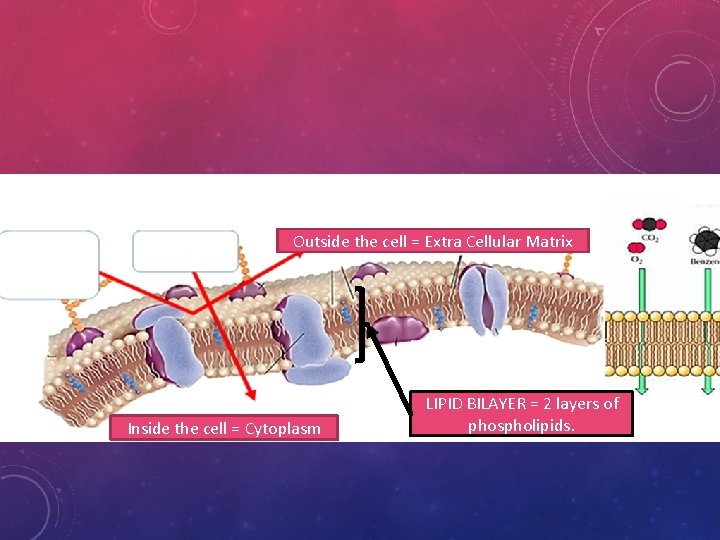
Outside the cell = Extra Cellular Matrix Inside the cell = Cytoplasm LIPID BILAYER = 2 layers of phospholipids.

WATER REGION PHOSPHOLIPID • Polar Heads are called… HYDROPHILIC HEADS • They love water Hydrophilic Heads • Made of polar phosphates • Tails are called… HYDROPHOBIC TAILS • They hate water. • Made of non-polar fatty acids Hydrophobic Tails NON- WATER REGION

LIPID BILAYER: SELECTIVELY PERMEABLE • Selectively = actively chooses among variables • Permeable = can allow things to pass through • Because of the properties of the phospholipids, • The cell membrane will allow only select molecules directly through. • Only SMALL POLAR AND NONPOLAR substances can pass. • LARGE AND CHARGED MOLECULES CANNOT PASS FREELY. • Which molecules qualify.
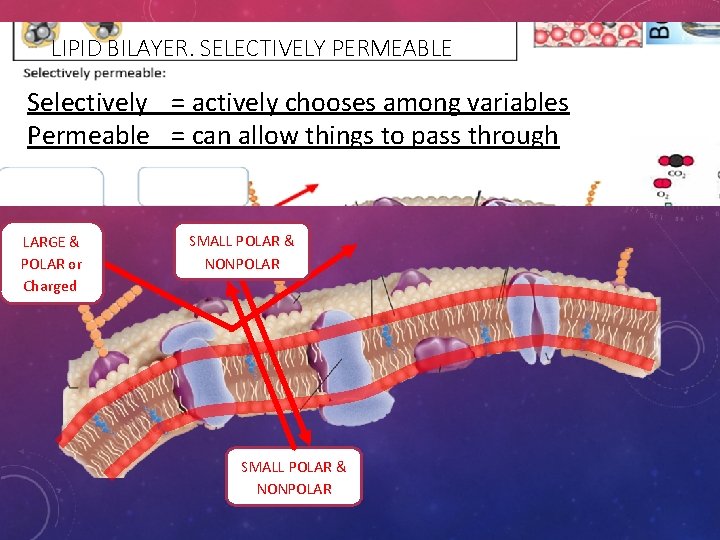
LIPID BILAYER. SELECTIVELY PERMEABLE Selectively = actively chooses among variables Permeable = can allow things to pass through LARGE & POLAR or Charged SMALL POLAR & NONPOLAR
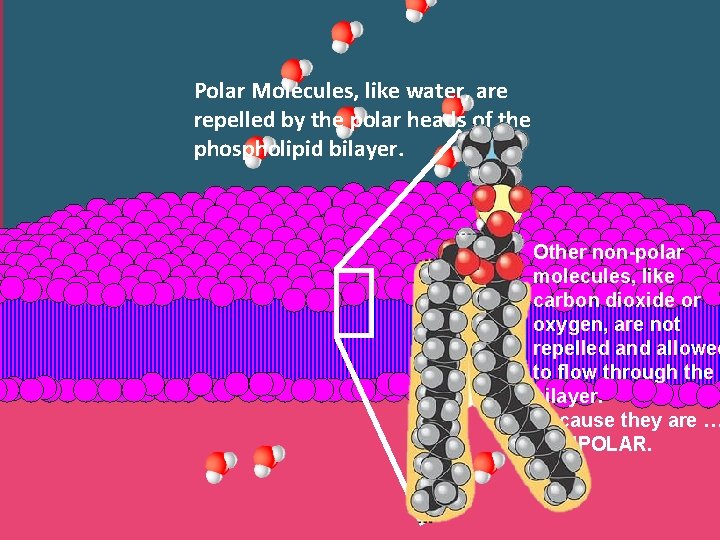
Polar Molecules, like water, are repelled by the polar heads of the phospholipid bilayer. Other non-polar molecules, like carbon dioxide or oxygen, are not repelled and allowed to flow through the bilayer. Because they are … NONPOLAR.
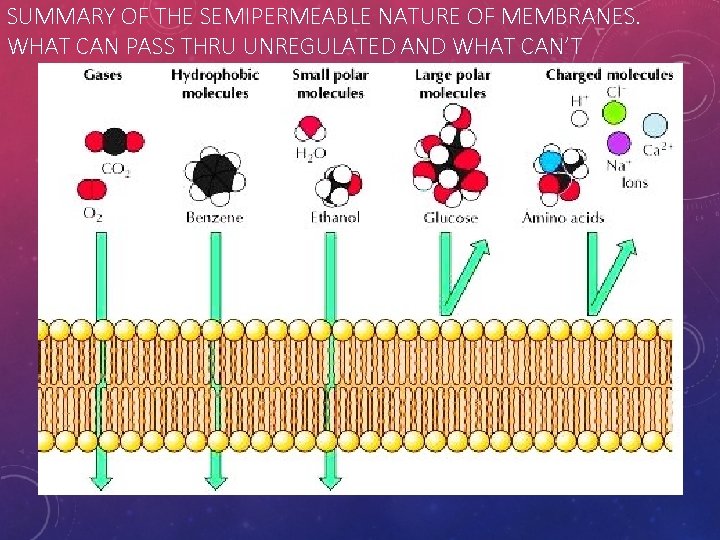
SUMMARY OF THE SEMIPERMEABLE NATURE OF MEMBRANES. WHAT CAN PASS THRU UNREGULATED AND WHAT CAN’T

MEMBRANE AND HOMEOSTASIS What are the four functions that the cell membrane performs to maintain homeostasis? 1. Regulates what goes in and out of the cell. • Acting as a gate keeper 2. To provide structural support. • Provides pressure against the internal environment 3. Recognizes foreign material. • Uses ‘feelers’ called receptors to identify good and bad stuff in the immediate area 4. Communicates and organizes with other cells. • Uses the ‘feelers’ and glycoproteins to organize with other cells into tissues forming the Extra Cellular Matrix (ECM)

MEMBRANE PROTEINS • Since getting things in and out of the cell helps maintain homeostasis & not everything can freely pass through the membrane, there has to be a way for polar & large molecules to get into the cell. • Homeostasis is also helped out by various proteins that can be found in the cell membrane. • Some proteins face inside the cell, and some face outside. Other proteins may stretch across the lipid bilayer and face both inside and outside. • Proteins are made of amino acids. Some amino acids are polar, and others are nonpolar. • The attraction and repulsion of polar and non-polar parts of the protein to water help hold the protein in the membrane.

MEMBRANE PROTEINS What are the 3 major types of membrane proteins? 1. Peripheral Proteins: these are buoyed to the surface of the membrane. 1. Can be associated with integral-type proteins. 2. Integral Proteins: these penetrate into the hydrophobic regions of the membrane 1. Cell surface markers: glycoproteins are proteins with attached sugar chains. These chains of sugars (remember polysaccharides and carbohydrates) act as markers to help identify themselves to other cells 2. Receptor proteins: these are the ‘feelers’ that identify good and bad substances in the environment 3. Enzymes: catalyze reactions that happen on the inside of the cell 3. Transmembrane Proteins: these span from outside to inside the cell. 1. Transport & Channel Proteins: allow large and/or polar substances to pass through the membrane
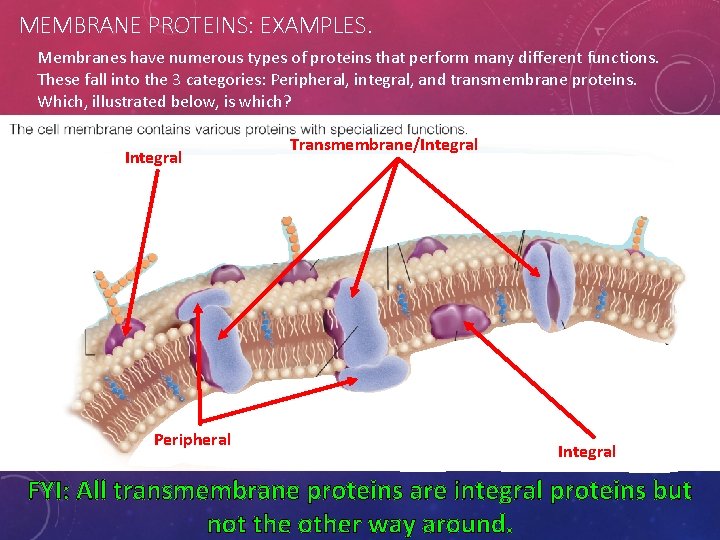
MEMBRANE PROTEINS: EXAMPLES. Membranes have numerous types of proteins that perform many different functions. These fall into the 3 categories: Peripheral, integral, and transmembrane proteins. Which, illustrated below, is which? Integral Peripheral Transmembrane/Integral FYI: All transmembrane proteins are integral proteins but not the other way around.
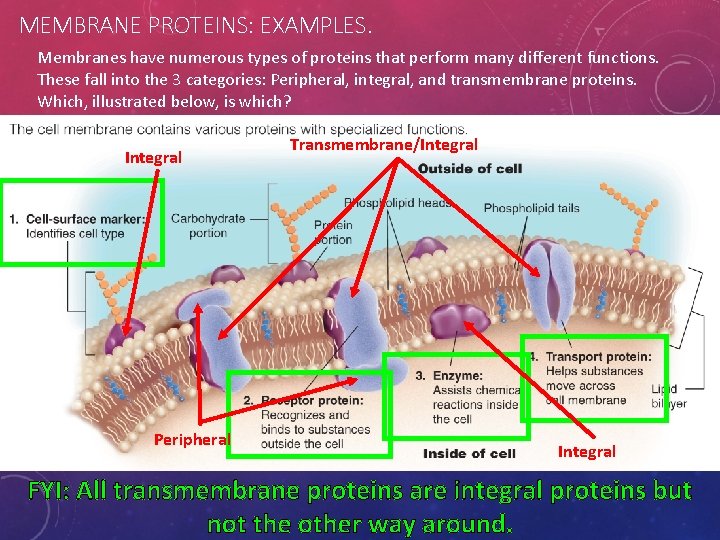
MEMBRANE PROTEINS: EXAMPLES. Membranes have numerous types of proteins that perform many different functions. These fall into the 3 categories: Peripheral, integral, and transmembrane proteins. Which, illustrated below, is which? Integral Peripheral Transmembrane/Integral FYI: All transmembrane proteins are integral proteins but not the other way around.
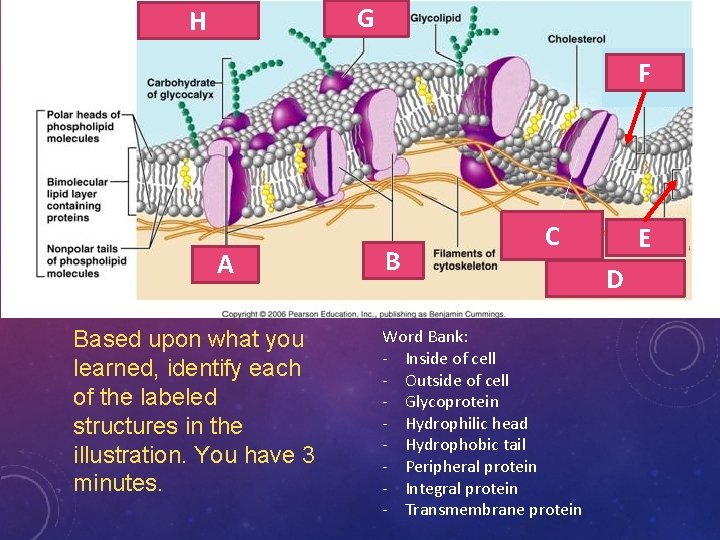
G H Hydrophili c Heads F A Based upon what you learned, identify each of the labeled structures in the illustration. You have 3 minutes. B C Transmembrane protein Word Bank: - Inside of cell - Outside of cell - Glycoprotein - Hydrophilic head - Hydrophobic tail - Peripheral protein - Integral protein - Transmembrane protein E Hydrophobi c Tails D
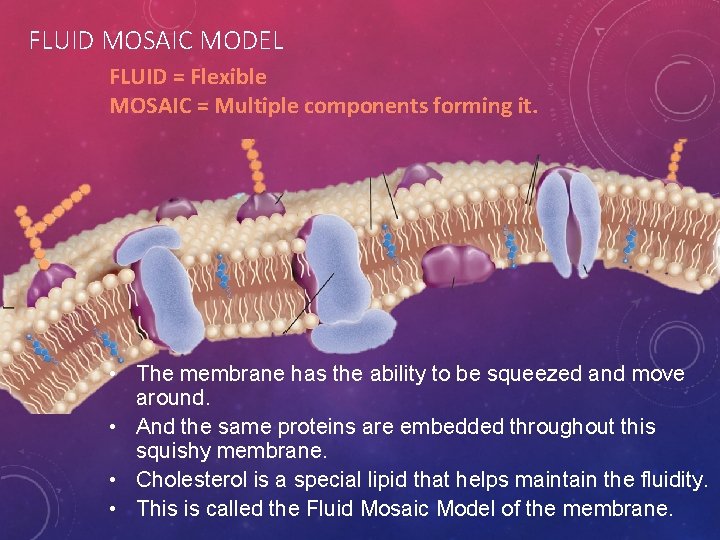
FLUID MOSAIC MODEL FLUID = Flexible MOSAIC = Multiple components forming it. • The membrane has the ability to be squeezed and move around. • And the same proteins are embedded throughout this squishy membrane. • Cholesterol is a special lipid that helps maintain the fluidity. • This is called the Fluid Mosaic Model of the membrane.

CLOSURE • REFLECT, I SAY! • What did you learn today? • (write this in your warm ups) • Do you have any questions? • Complete the 6 open-ended questions before tomorrow on a separate sheet of paper. • I’m expecting you to number and write the question. Then write your answers in complete sentences. • Use any extra time to get your notebooks squared away.
- Slides: 28