The Carbon Cycle Carbon Dioxide and Carbonate system

The Carbon Cycle

Carbon Dioxide and Carbonate system Why is it important? 1. Regulates temperature of the planet 2. Important for life in the ocean 3. Regulates the p. H of water
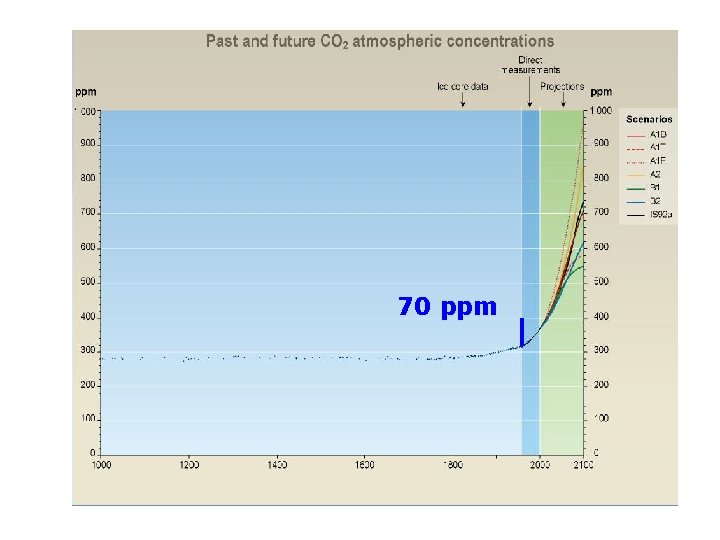
70 ppm
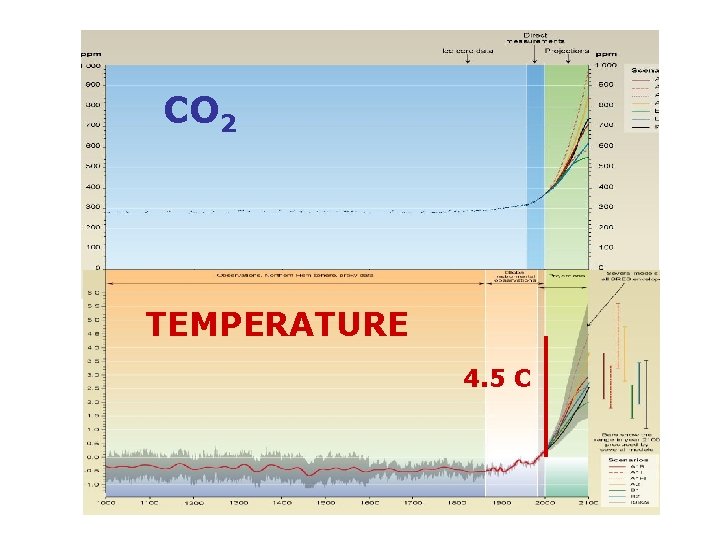
CO 2 TEMPERATURE 4. 5 C
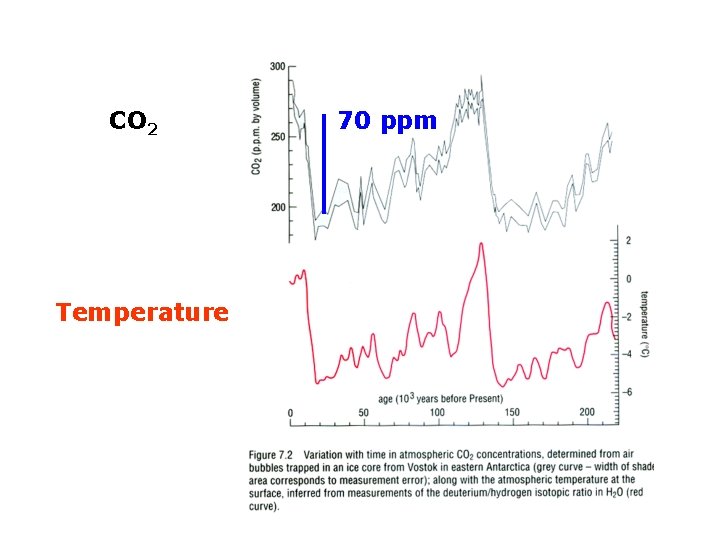
CO 2 Temperature 70 ppm
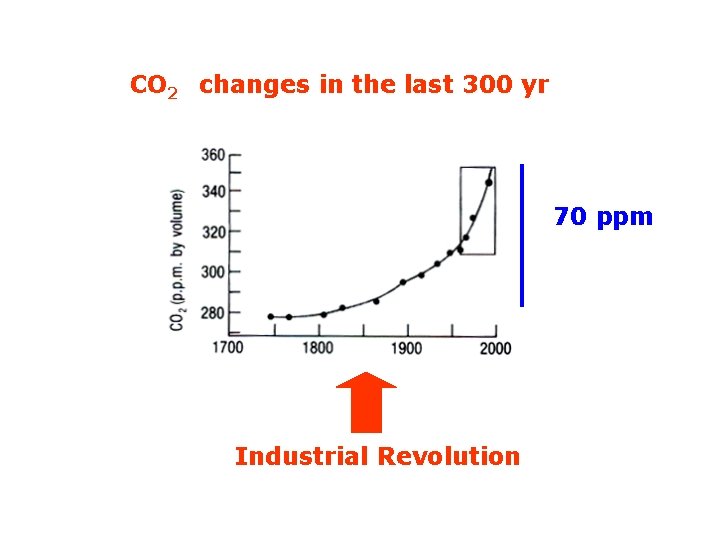
CO 2 changes in the last 300 yr 70 ppm Industrial Revolution
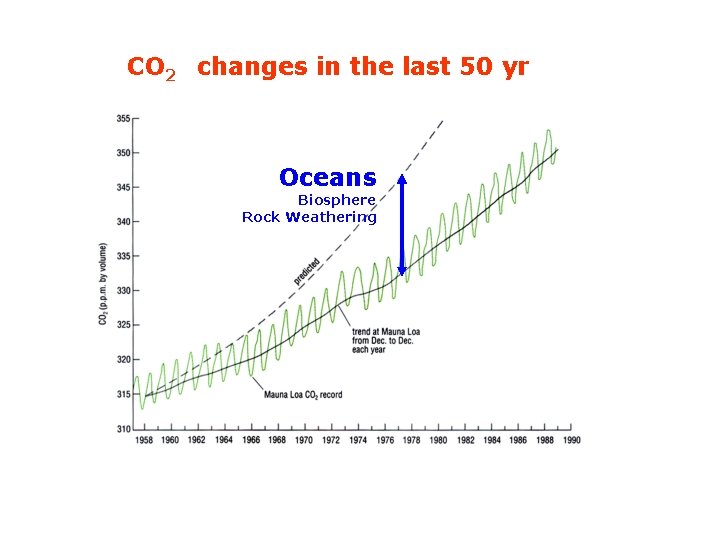
CO 2 changes in the last 50 yr Oceans Biosphere Rock Weathering

How much CO 2 can you sink in the ocean? Chemical Process that control CO 2 absorption in the ocean Biological Physical Carbon Cycle
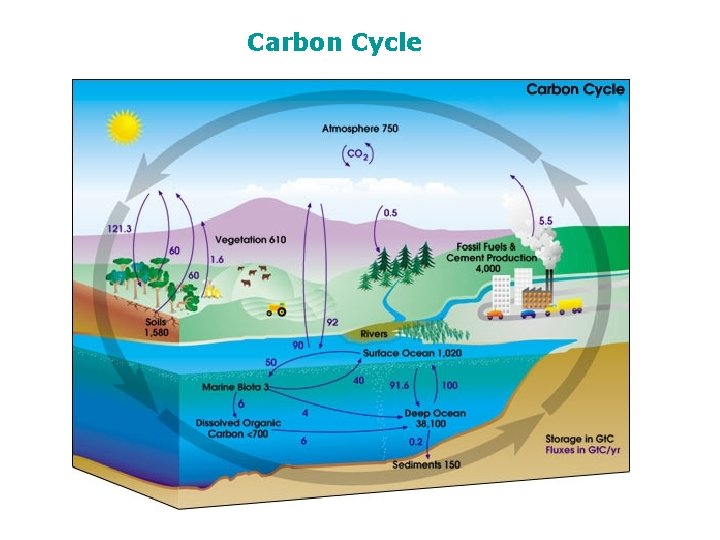
Carbon Cycle
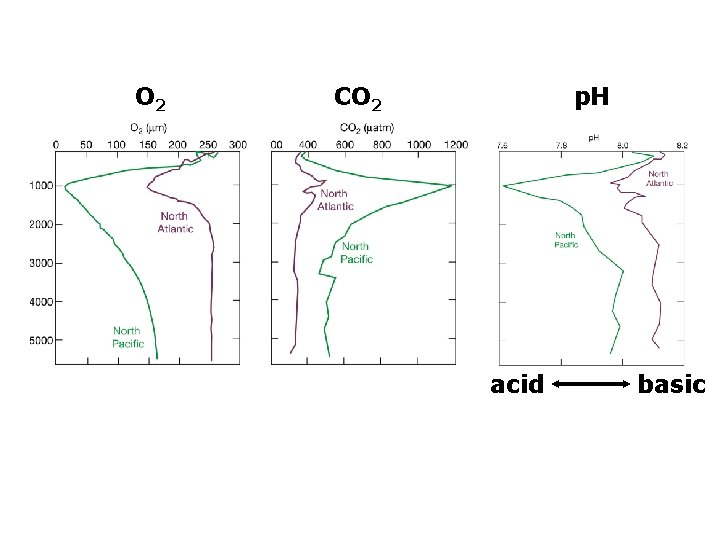
O 2 CO 2 p. H acid basic
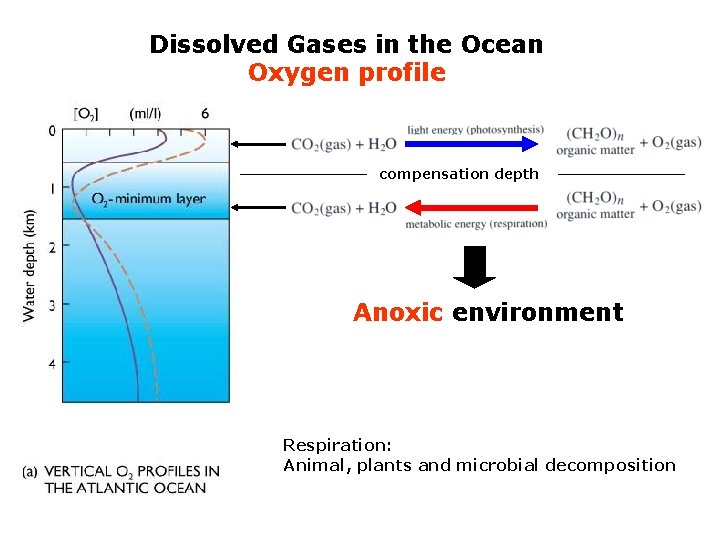
Dissolved Gases in the Ocean Oxygen profile compensation depth Anoxic environment Respiration: Animal, plants and microbial decomposition
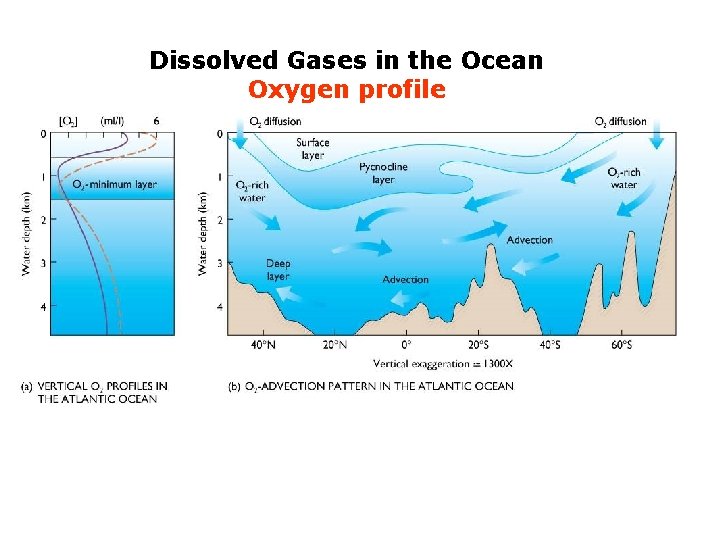
Dissolved Gases in the Ocean Oxygen profile
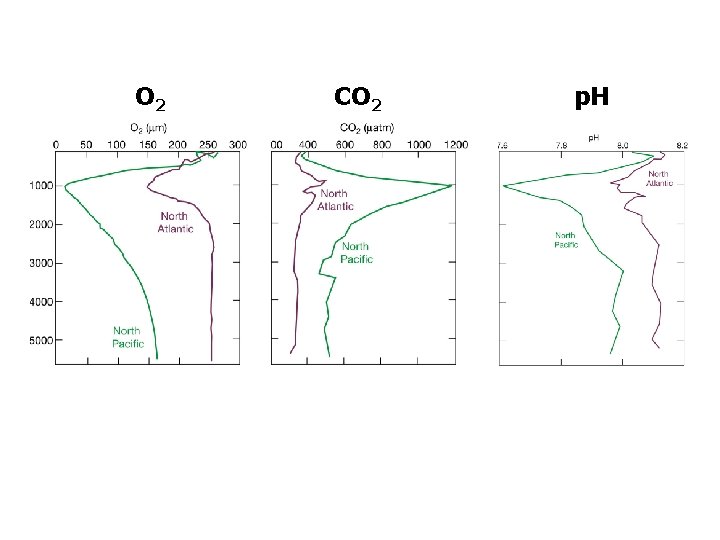
O 2 CO 2 p. H

The Carbonate System sources of inorganic carbon from dissolved CO 2 gas from dissolution of Calcium Carbonate NOTE: Biology and Physics participate in the equilibrium of the carbonate system
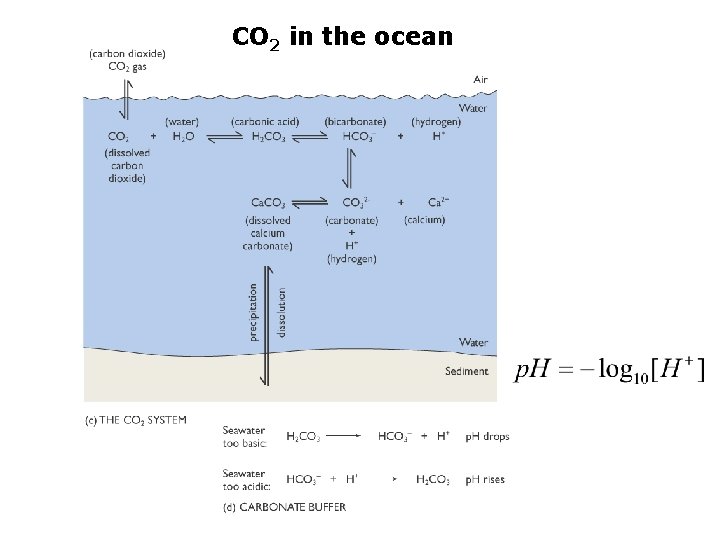
CO 2 in the ocean

Total dissolved inorganic carbon this is very small Total dissolved inorganic carbon not found in this form
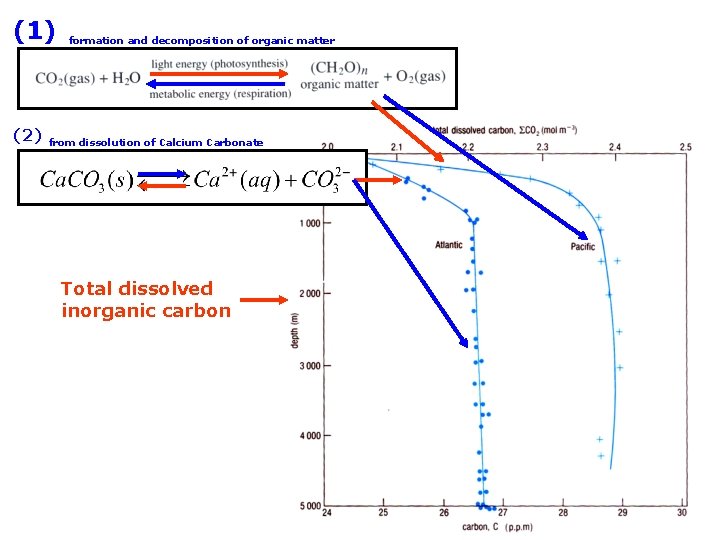
(1) (2) formation and decomposition of organic matter from dissolution of Calcium Carbonate Total dissolved inorganic carbon
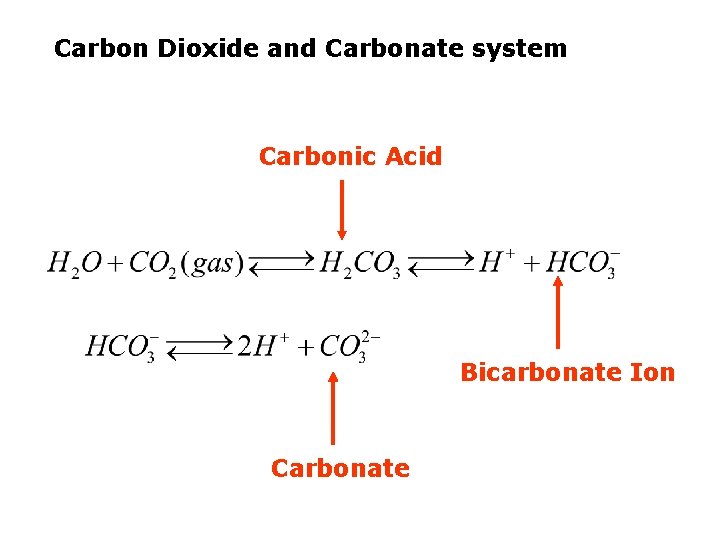
Carbon Dioxide and Carbonate system Carbonic Acid Bicarbonate Ion Carbonate
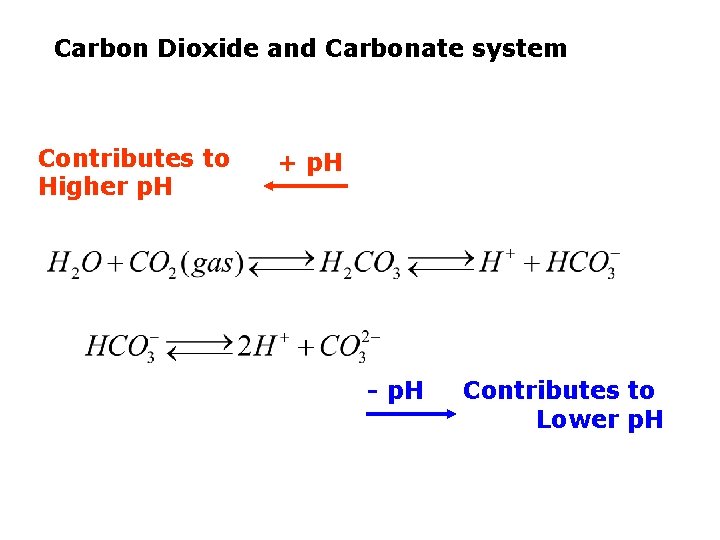
Carbon Dioxide and Carbonate system Contributes to Higher p. H + p. H - p. H Contributes to Lower p. H
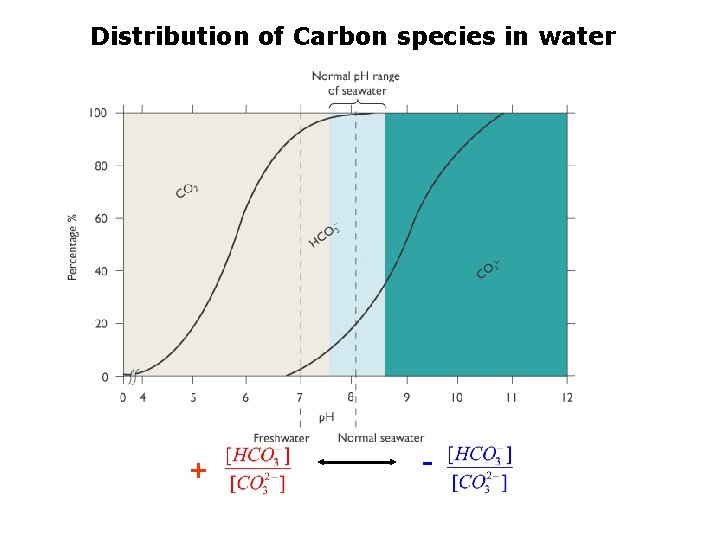
Distribution of Carbon species in water + -
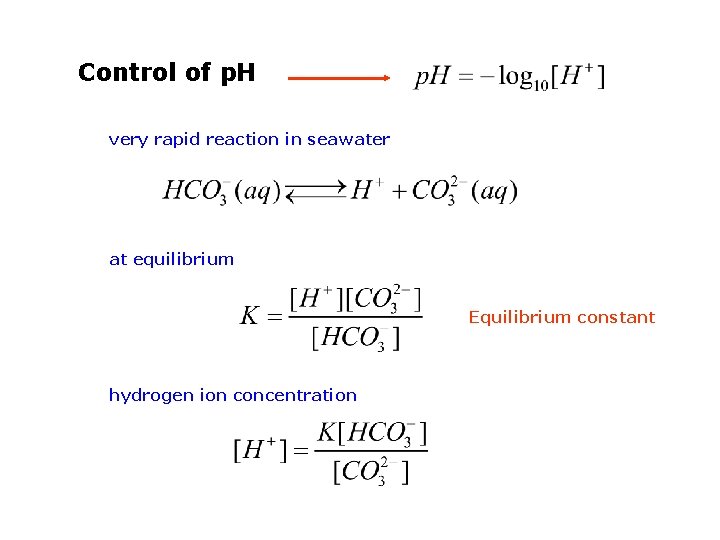
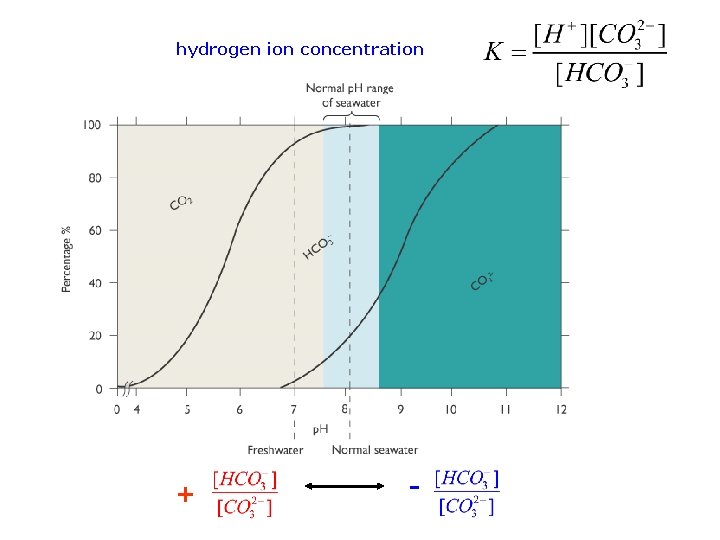
Control of p. H very rapid reaction in seawater at equilibrium Equilibrium constant hydrogen ion concentration
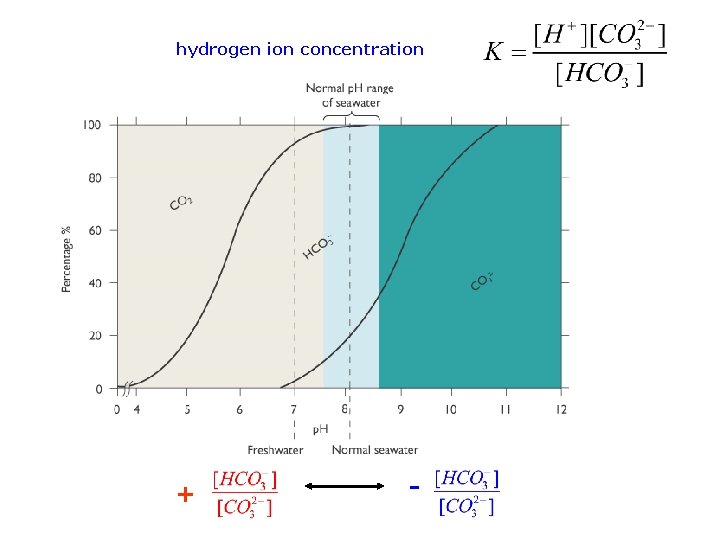
hydrogen ion concentration + -

Why is the p. H of seawater close to neutral? seawater p. H=7. 5 -8. 5
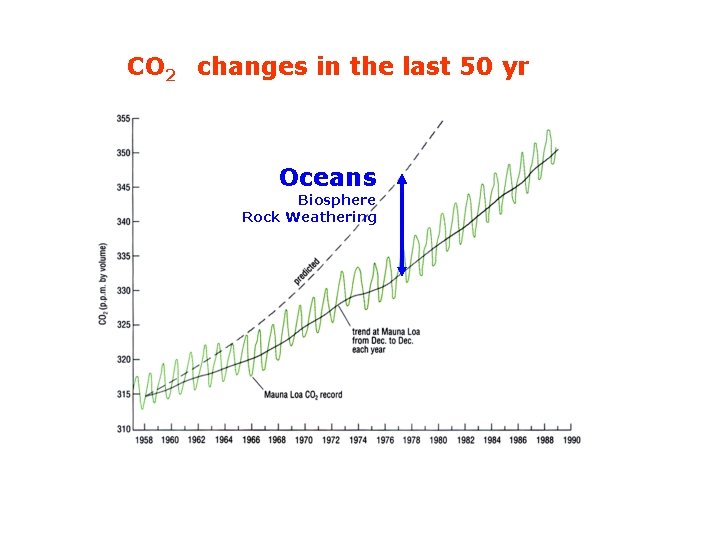
CO 2 changes in the last 50 yr Oceans Biosphere Rock Weathering

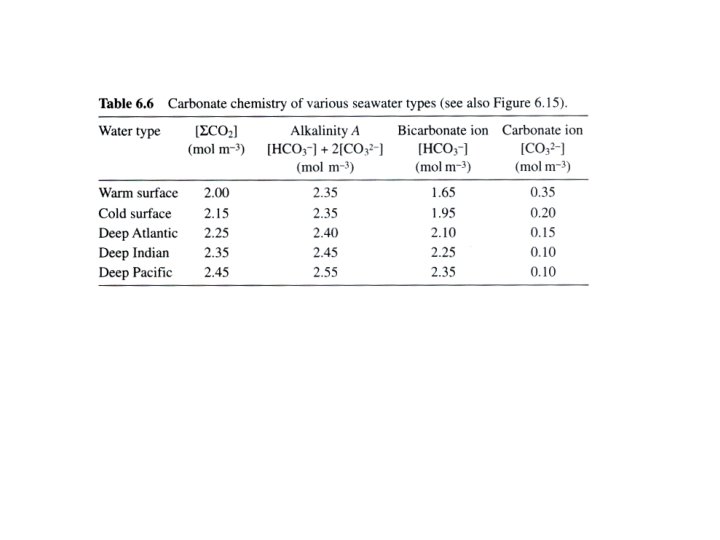
What control the absorption of Carbon Dioxide in the ocean? The carbonate system chemistry in seawater How can we measure it? The concept of alkalinity, p. H …

Atomic mass sodium 23 g/mol Atomic mass chloride 35. 5 g/mol

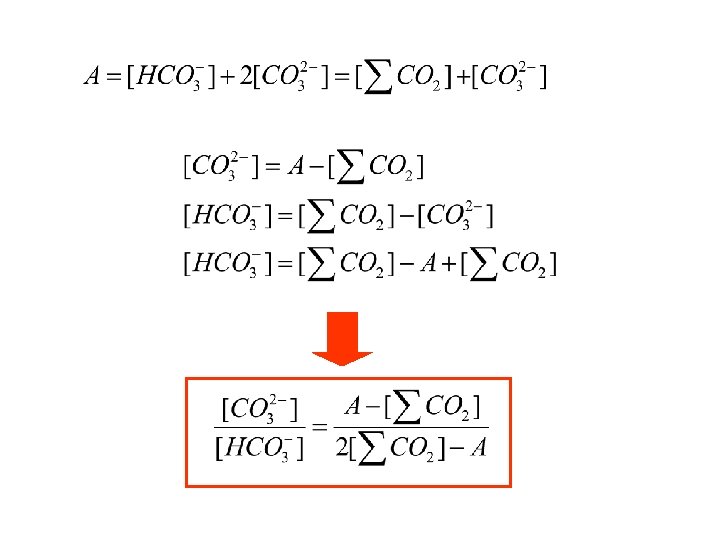
Def: Alkalinity: is the net molar concentration, in chargeequivalents, of the cations of strong bases in excess of the anions of strong acids Strong base cations Strong acid anions

What happens to this balance? Strong base cations Strong acid anions
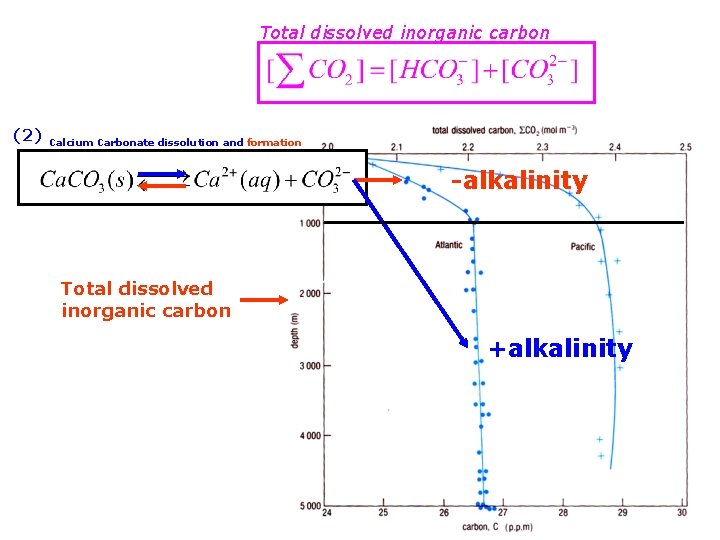
Total dissolved inorganic carbon (2) Calcium Carbonate dissolution and formation -alkalinity Total dissolved inorganic carbon +alkalinity

Strong base cations Strong acid anions This excess of + charges turns out to be equal to the week acid anions of the carbonate system

Strong base cations Strong acid anions Bicarbonate and Carbonate Ions

Alkalinity can be measured by titration

What happens if I add or remove CO 2 by photosynthesis and respiration?
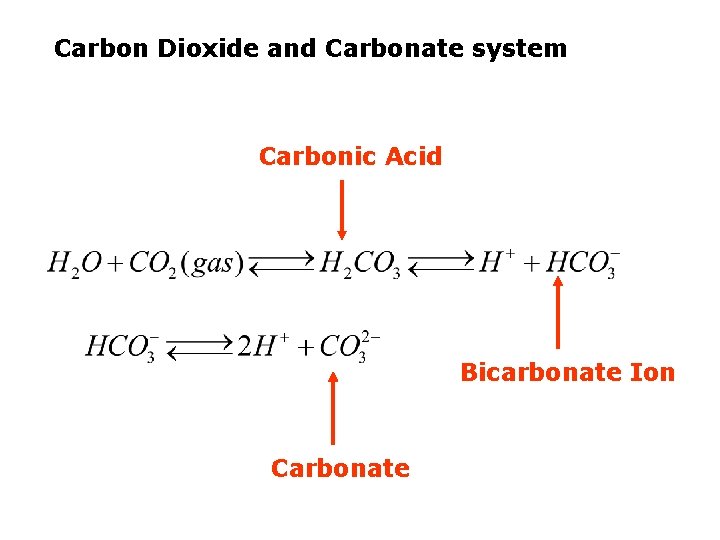
Carbon Dioxide and Carbonate system Carbonic Acid Bicarbonate Ion Carbonate
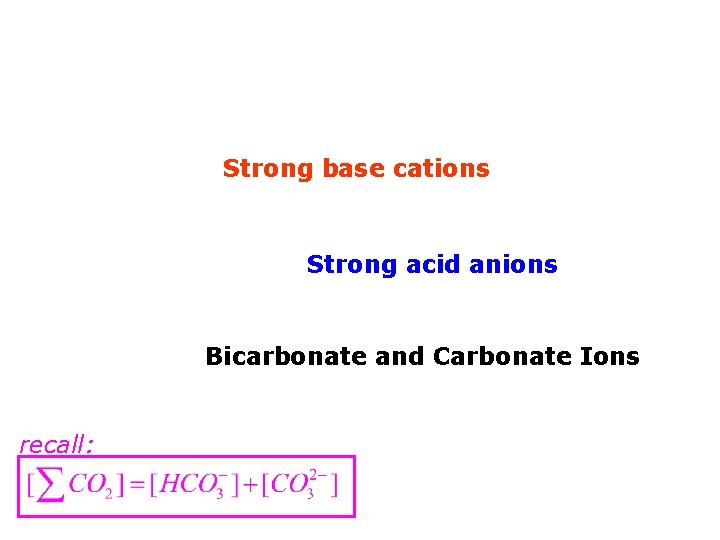
Strong base cations Strong acid anions Bicarbonate and Carbonate Ions recall:

Strong base cations Strong acid anions Bicarbonate and Carbonate Ions

Strong base cations Strong acid anions Bicarbonate and Carbonate Ions
hydrogen ion concentration + -
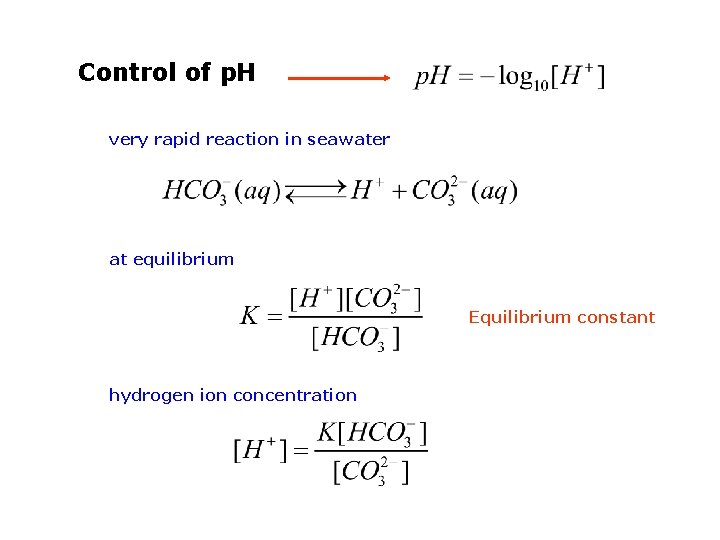
Control of p. H very rapid reaction in seawater at equilibrium Equilibrium constant hydrogen ion concentration
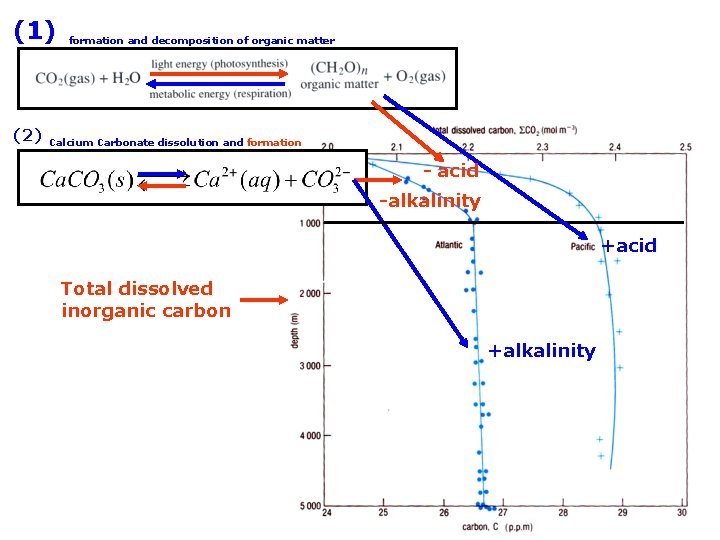
(1) (2) formation and decomposition of organic matter Calcium Carbonate dissolution and formation - acid -alkalinity +acid Total dissolved inorganic carbon +alkalinity
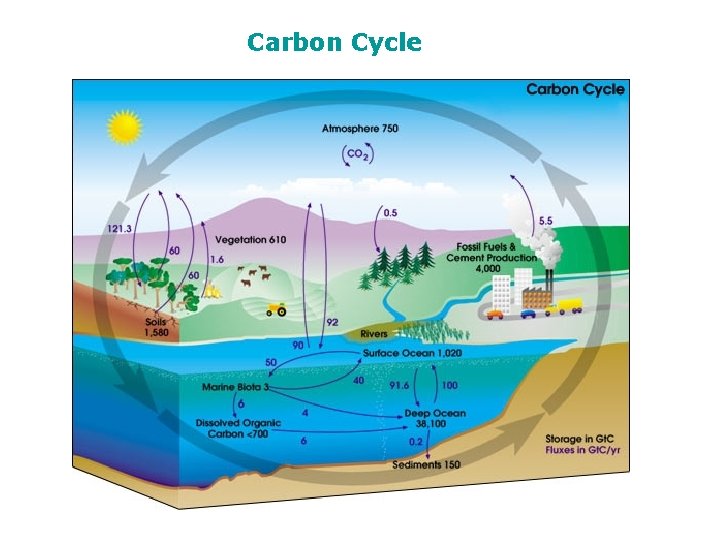
Carbon Cycle
- Slides: 44