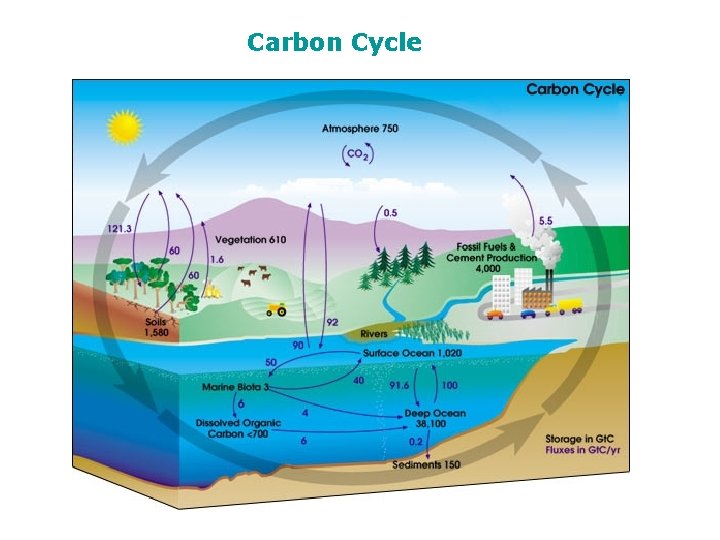
The Carbon Cycle Carbon Dioxide and Carbonate system

The Carbon Cycle

Carbon Dioxide and Carbonate system Why is it important? 1. CO 2 regulates temperature of the planet 2. Important for life in the ocean 3. Regulates the p. H of water
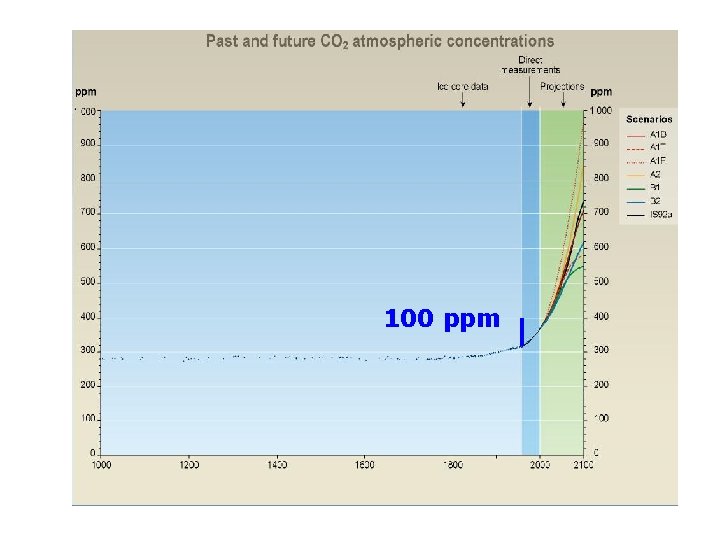
100 ppm
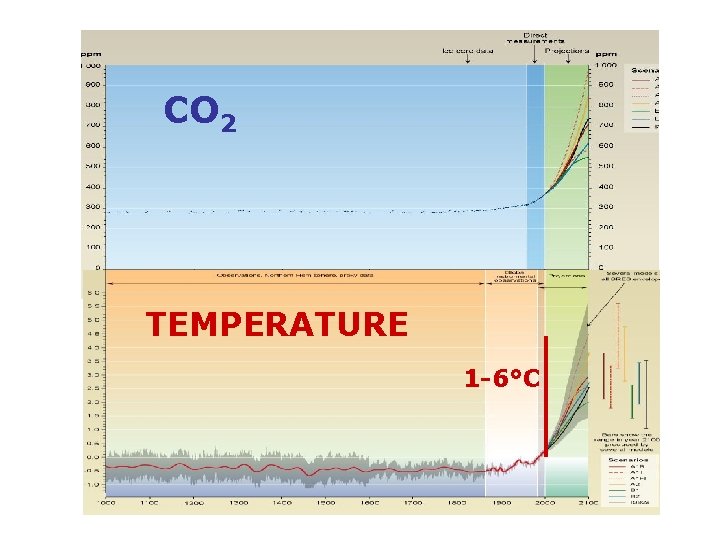
CO 2 TEMPERATURE 1 -6°C
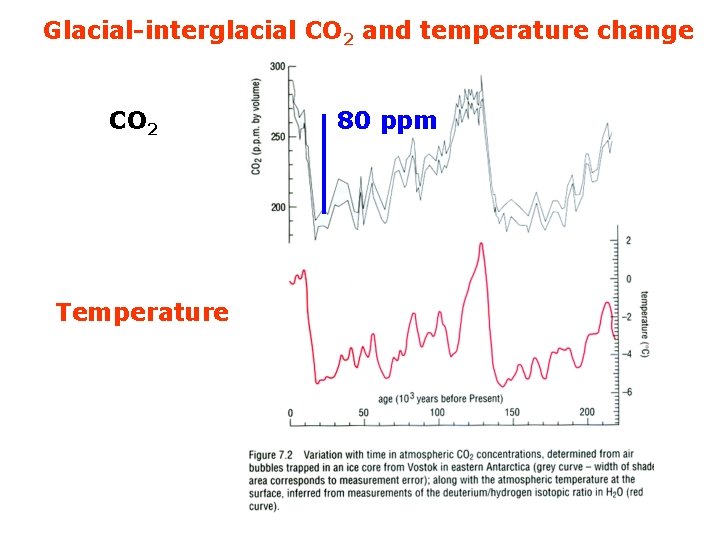
Glacial-interglacial CO 2 and temperature change CO 2 Temperature 80 ppm
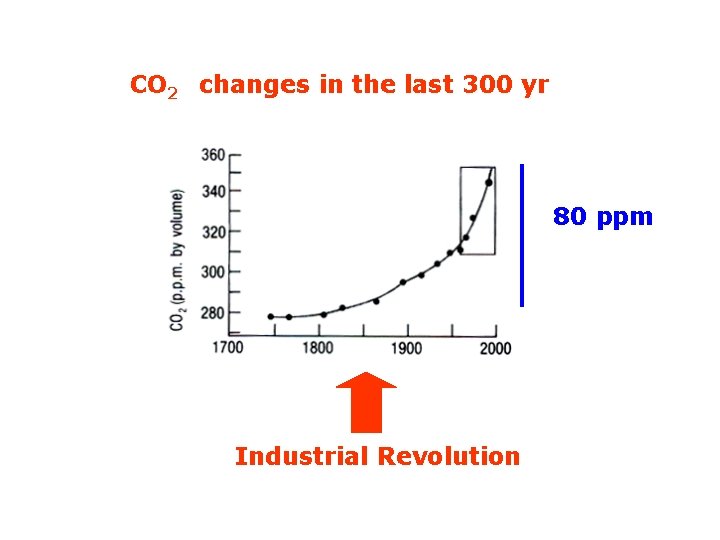
CO 2 changes in the last 300 yr 80 ppm Industrial Revolution
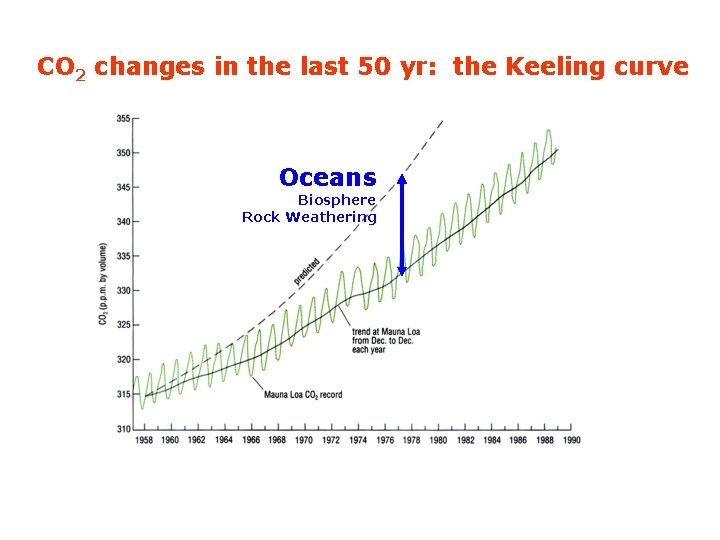
CO 2 changes in the last 50 yr: the Keeling curve Oceans Biosphere Rock Weathering

How much CO 2 can you sink in the ocean? Chemical Process that control CO 2 absorption in the ocean Biological Physical Carbon Cycle
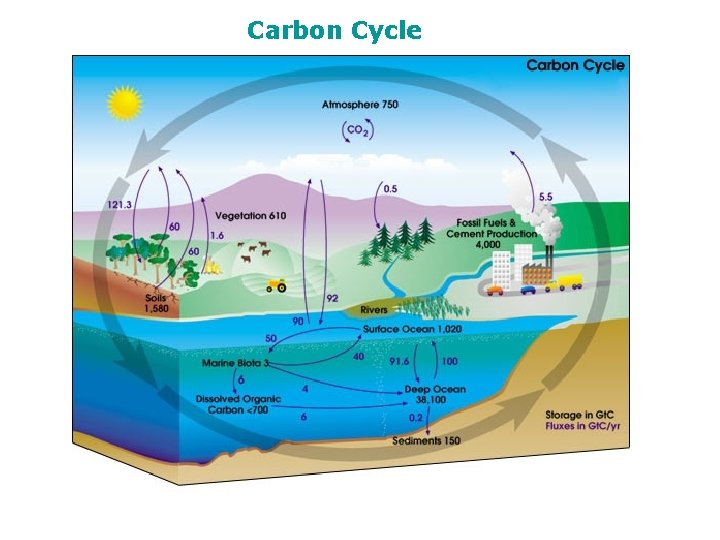
Carbon Cycle
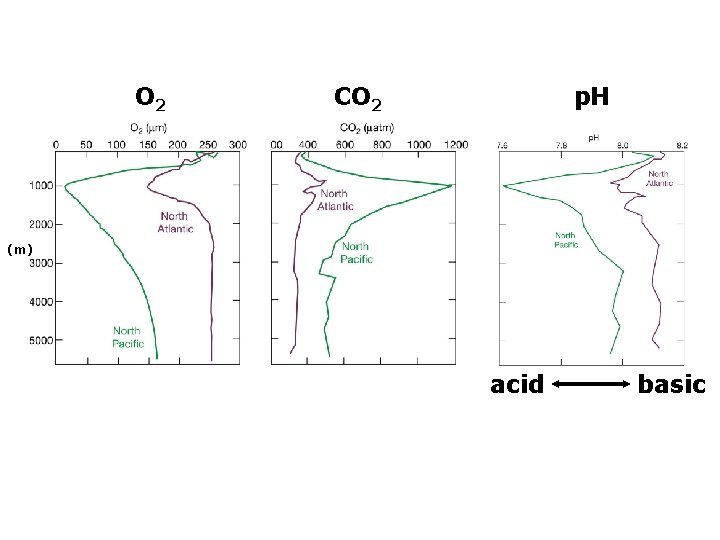
O 2 CO 2 p. H (m) acid basic
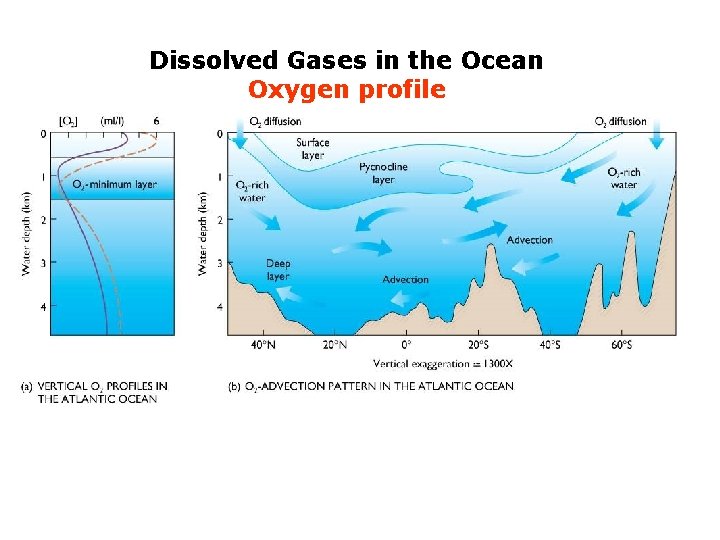
Dissolved Gases in the Ocean Oxygen profile
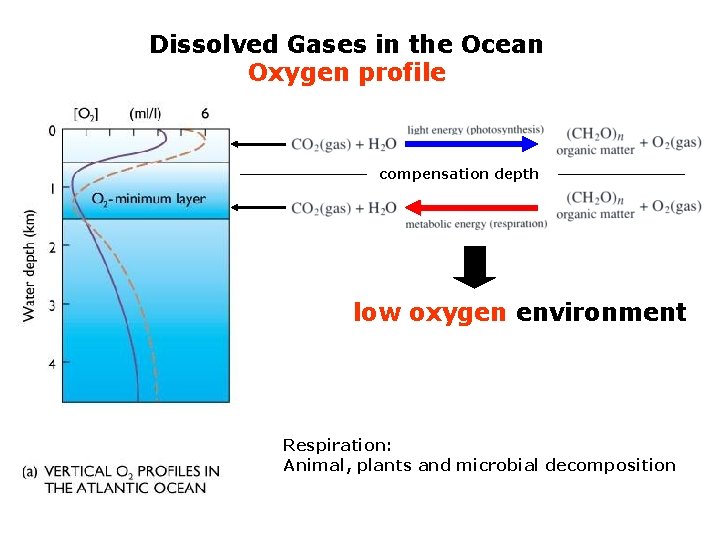
Dissolved Gases in the Ocean Oxygen profile compensation depth low oxygen environment Respiration: Animal, plants and microbial decomposition

The Carbonate System sources of inorganic carbon from dissolved CO 2 gas from dissolution of Calcium Carbonate
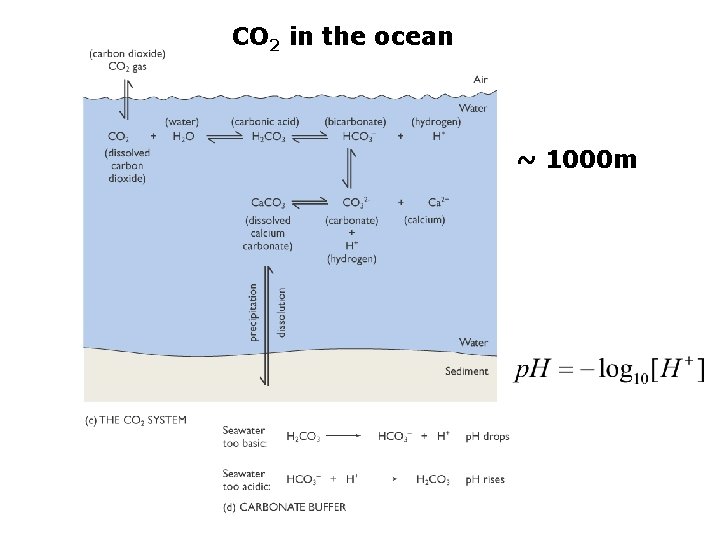
CO 2 in the ocean ~ 1000 m

Total dissolved inorganic carbon this is very small Total dissolved inorganic carbon not found in this form
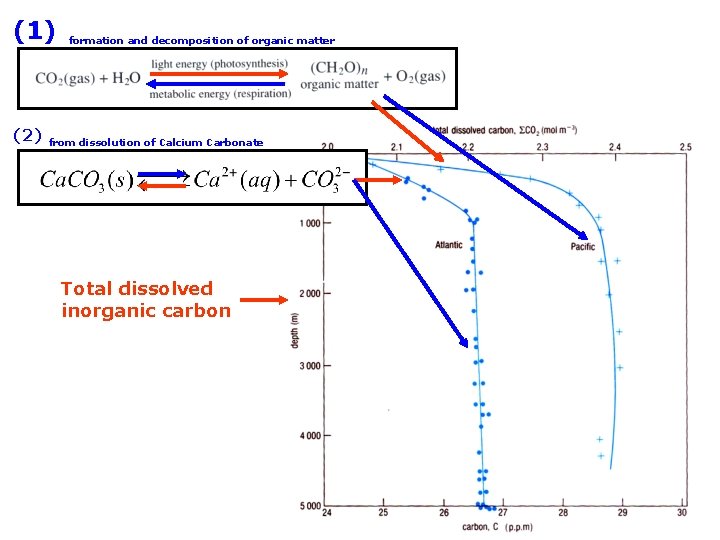
(1) (2) formation and decomposition of organic matter from dissolution of Calcium Carbonate Total dissolved inorganic carbon
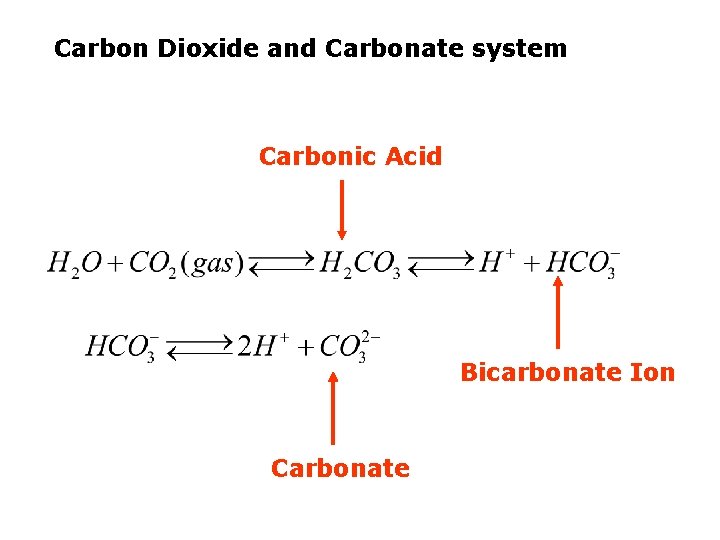
Carbon Dioxide and Carbonate system Carbonic Acid Bicarbonate Ion Carbonate
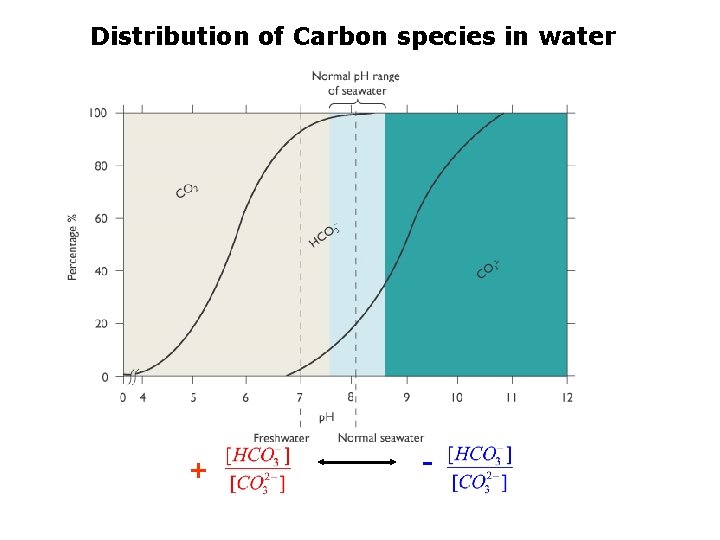
Distribution of Carbon species in water + -

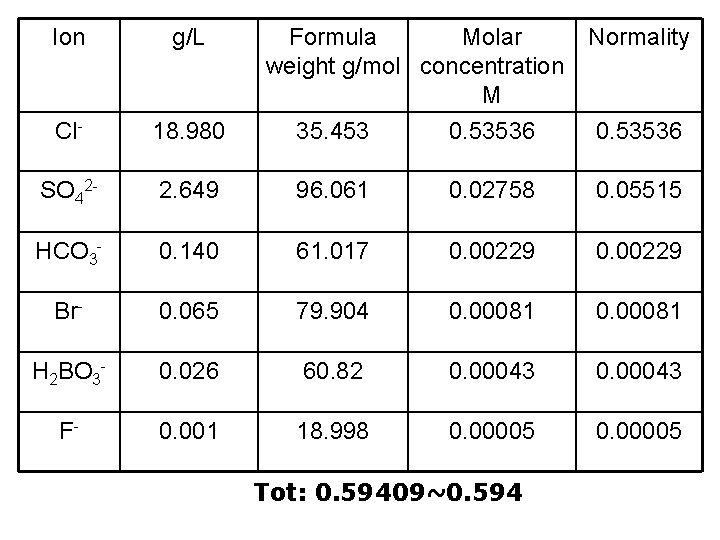
Ion g/L Formula Molar weight g/mol concentration M 35. 453 0. 53536 Cl- 18. 980 SO 42 - 2. 649 96. 061 0. 02758 0. 05515 HCO 3 - 0. 140 61. 017 0. 00229 Br- 0. 065 79. 904 0. 00081 H 2 BO 3 - 0. 026 60. 82 0. 00043 F- 0. 001 18. 998 0. 00005 Tot: 0. 59409~0. 594 Normality 0. 53536
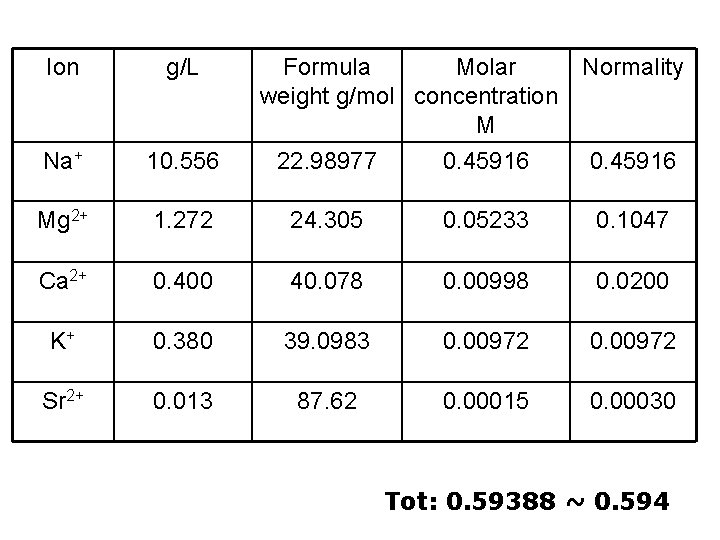
Ion g/L Formula Molar weight g/mol concentration M 22. 98977 0. 45916 Normality Na+ 10. 556 Mg 2+ 1. 272 24. 305 0. 05233 0. 1047 Ca 2+ 0. 400 40. 078 0. 00998 0. 0200 K+ 0. 380 39. 0983 0. 00972 Sr 2+ 0. 013 87. 62 0. 00015 0. 00030 0. 45916 Tot: 0. 59388 ~ 0. 594

What control the absorption of Carbon Dioxide in the ocean? The carbonate system chemistry in seawater How can we measure it? The concept of alkalinity, p. H …

Def: Alkalinity: is the net molar concentration, in chargeequivalents, of the cations of strong bases in excess of the anions of strong acids Strong base cations (less) strong acid anions in simple terms represents the degree to which a solution resists a decrease in p. H when acid is added

Alkalinity • Alkalinity is fundamental for hard corals and coralline algae to properly secrete new skeletal material. When alkalinity levels drop, the carbonate ions needed are not available and the process slows or stops. • Alkalinity is measured in one of two units: milliequivalents per liter (meq/l), or parts per million of calcium carbonate (ppm Ca. CO 3). • The 'ppm Ca. CO 3' unit reports the concentration of Ca. CO 3 in pure water that would provide the same buffering capacity as the water sample in question. This does not mean the sample contains that much Ca. CO 3. • In natural seawater, though, carbonates make up 96% of the alkalinity so equating alkalinity with carbonate hardness isn't too far off.
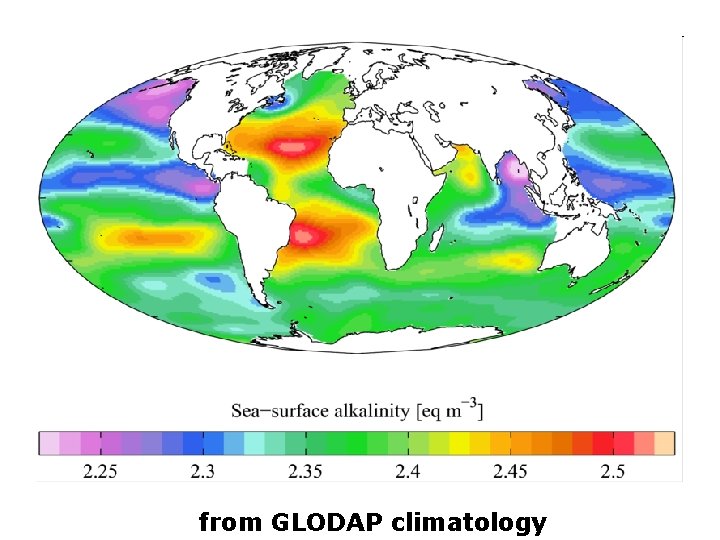
from GLODAP climatology

What happens to this balance? Strong base cations (less) Strong acid anions
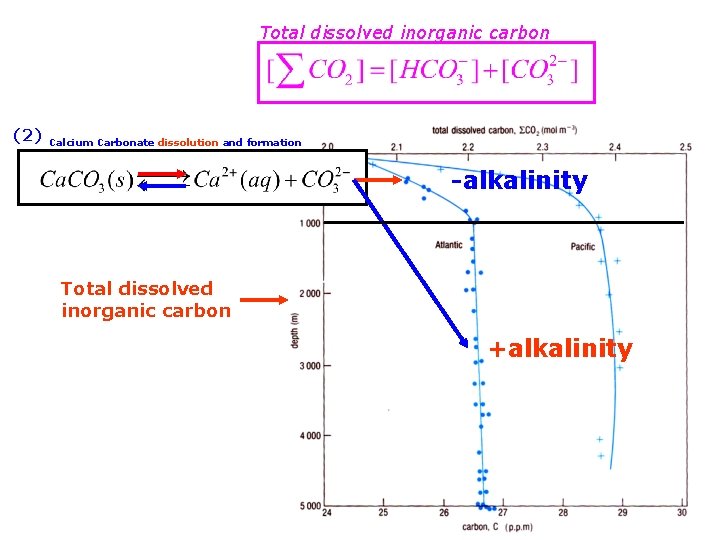
Total dissolved inorganic carbon (2) Calcium Carbonate dissolution and formation -alkalinity Total dissolved inorganic carbon +alkalinity

What happens if I add or remove CO 2 by photosynthesis and respiration?
Carbon Cycle
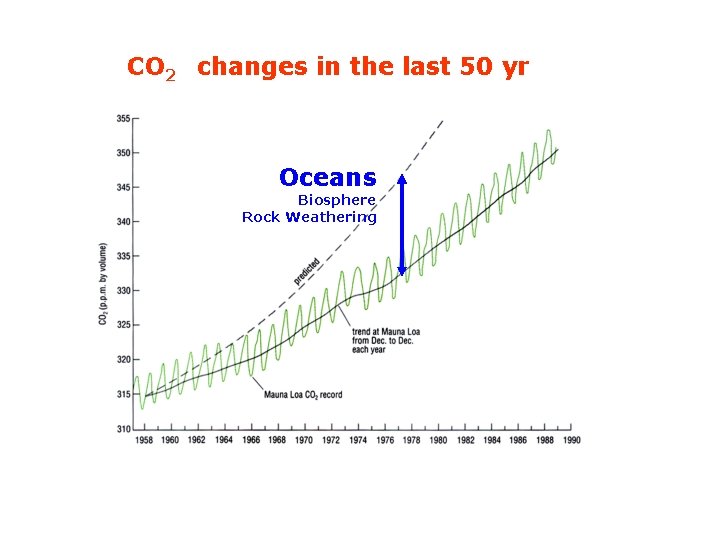
CO 2 changes in the last 50 yr Oceans Biosphere Rock Weathering
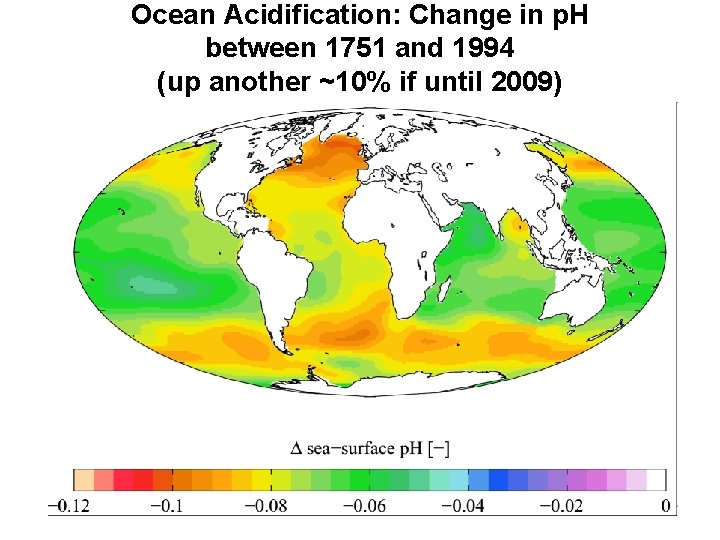
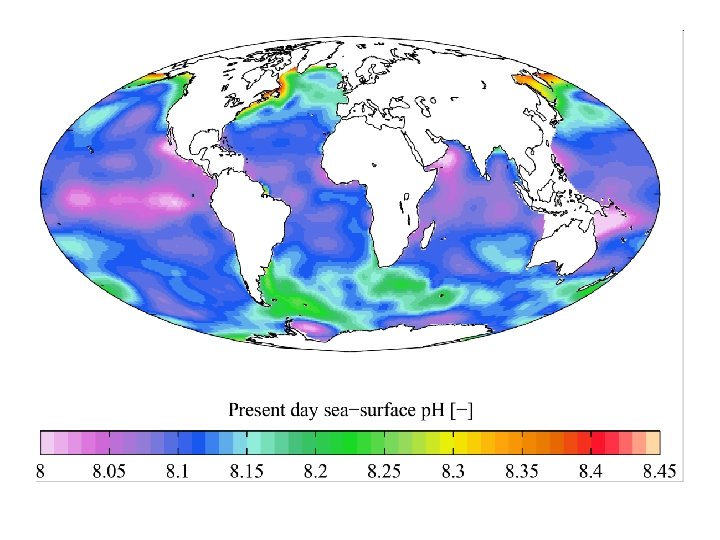
Ocean Acidification: Change in p. H between 1751 and 1994 (up another ~10% if until 2009)
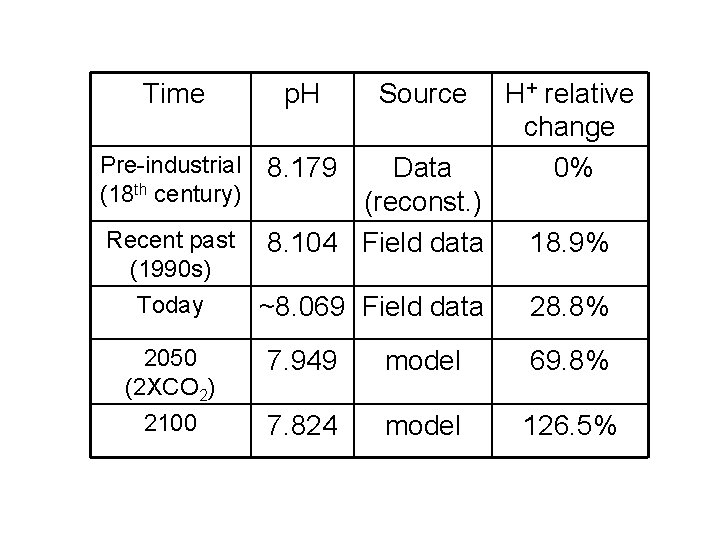
Time p. H Pre-industrial (18 th century) 8. 179 Recent past (1990 s) Today 2050 (2 XCO 2) 2100 Source Data (reconst. ) 8. 104 Field data H+ relative change 0% 18. 9% ~8. 069 Field data 28. 8% 7. 949 model 69. 8% 7. 824 model 126. 5%

several important articles in 2010 summarized by Scientific American: • July 2010: William Howard (ACEC Research Center in Hobart, Tasmania) states that "the current rate of ocean acidification is about a hundred times faster than the most rapid events" in the geologic past. • August 2010: Marah J. Hardt and Carl Safina (USF) make the case that in the 15 -year period 1995 -2010, acidity has increased 6 percent in the upper 100 meters of the Pacific Ocean from Hawaii to Alaska

A summary of studies indicate that almost all calcifying species examined to date have shown reduced calcification rates at higher p. CO 2 levels, Today‘s world High-CO 2 world p. CO : 280 -380 ppm. V 2 p. CO 2: 580 -720 ppm. V Gephyrocapsa oceanica Emiliania huxleyi Calcidiscus leptoporus but only a small fraction of the natural species have been tested.

Consensus among the participants was that higher CO 2 levels will generally be detrimental to calcifying organisms and that food web structures and biodiversity will likely change, Coccolithophores calcite Forams calcite Corals aragonite but it is not clear how this might impact overall productivity and top level predators (e. g. fish).


Ocean acidification: What corals are dying to tell us slides from Ken Caldeira
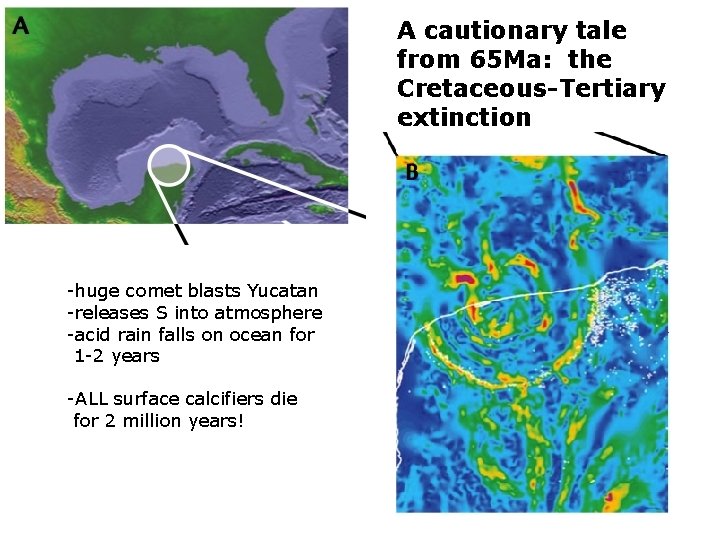
A cautionary tale from 65 Ma: the Cretaceous-Tertiary extinction -huge comet blasts Yucatan -releases S into atmosphere -acid rain falls on ocean for 1 -2 years -ALL surface calcifiers die for 2 million years!
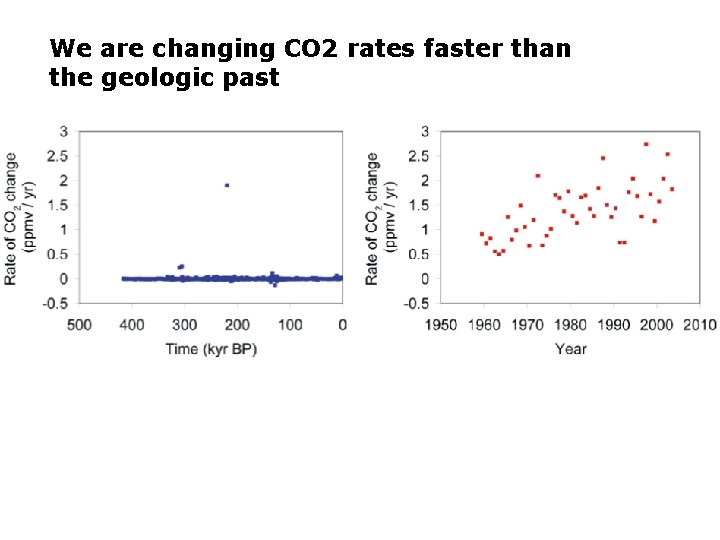
We are changing CO 2 rates faster than the geologic past
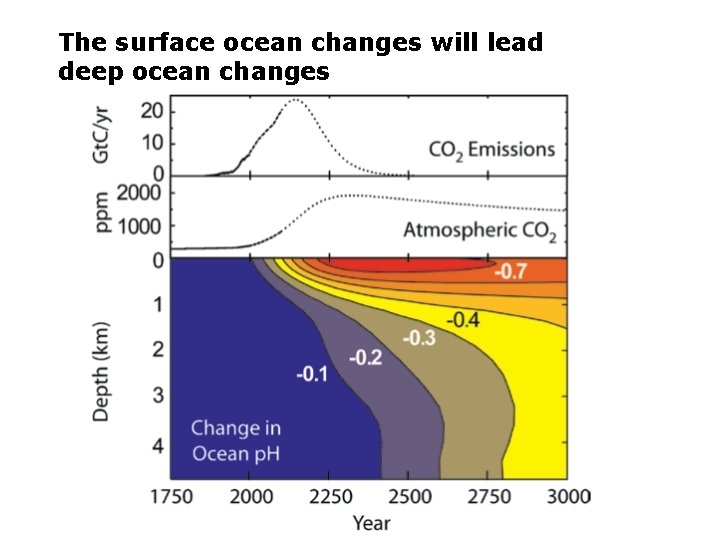
The surface ocean changes will lead deep ocean changes
- Slides: 42