The Bypass Angioplasty Revascularization Investigation 2 Diabetes Trial

The Bypass Angioplasty Revascularization Investigation 2 Diabetes Trial BARI 2 D Trial Presented at the American Diabetes Association (ADA) Annual Scientific Sessions 2009 in New Orleans Copyleft Clinical Trial Results. You Must Redistribute Slides

BARI 2 D Trial: Background • Patients with Type 2 diabetes have an increased risk of suffering a cardiovascular event over non-diabetic patients. • The success of coronary revascularization in reducing myocardial infarction and death in diabetic patients with chronic stable angina has not been established. • Similarly, it is unclear if insulin sensitization therapy offers benefits over insulin provision therapy in reducing cardiovascular events. Copyleft Clinical Trial Results. You Must Redistribute Slides BARI 2 D Study Group, NEJM 2009
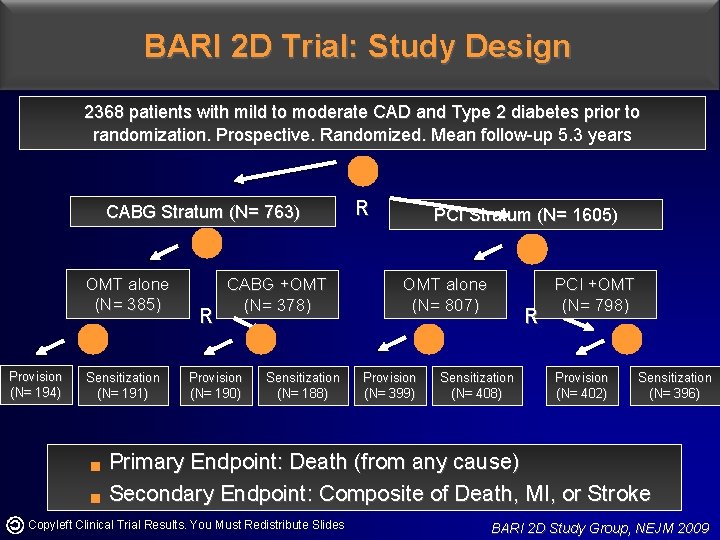
BARI 2 D Trial: Study Design 2368 patients with mild to moderate CAD and Type 2 diabetes prior to randomization. Prospective. Randomized. Mean follow-up 5. 3 years CABG Stratum (N= 763) OMT alone (N= 385) Provision (N= 194) Sensitization (N= 191) g g R CABG +OMT (N= 378) Provision (N= 190) Sensitization (N= 188) R PCI Stratum (N= 1605) OMT alone (N= 807) Provision (N= 399) R Sensitization (N= 408) PCI +OMT (N= 798) Provision (N= 402) Sensitization (N= 396) Primary Endpoint: Death (from any cause) Secondary Endpoint: Composite of Death, MI, or Stroke Copyleft Clinical Trial Results. You Must Redistribute Slides BARI 2 D Study Group, NEJM 2009
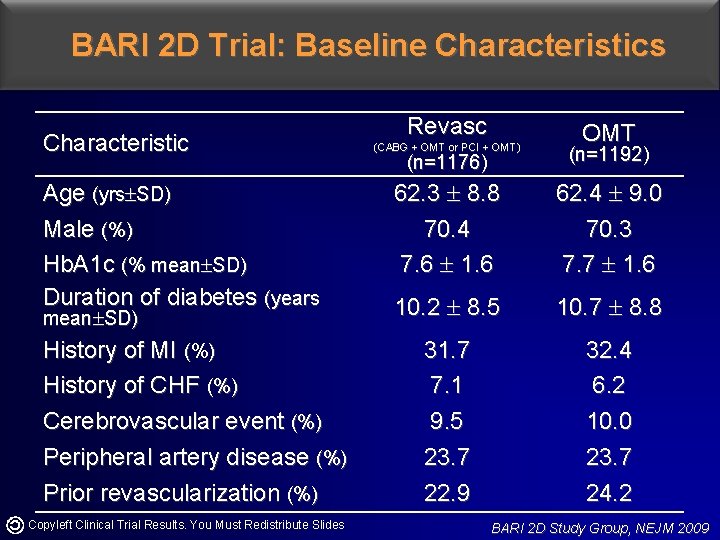
BARI 2 D Trial: Baseline Characteristics Characteristic Age (yrs SD) Male (%) Hb. A 1 c (% mean SD) Duration of diabetes (years mean SD) History of MI (%) History of CHF (%) Cerebrovascular event (%) Peripheral artery disease (%) Prior revascularization (%) Copyleft Clinical Trial Results. You Must Redistribute Slides Revasc OMT (CABG + OMT or PCI + OMT) (n=1176) (n=1192) 62. 3 8. 8 70. 4 7. 6 1. 6 62. 4 9. 0 70. 3 7. 7 1. 6 10. 2 8. 5 10. 7 8. 8 31. 7 7. 1 9. 5 23. 7 22. 9 32. 4 6. 2 10. 0 23. 7 24. 2 BARI 2 D Study Group, NEJM 2009
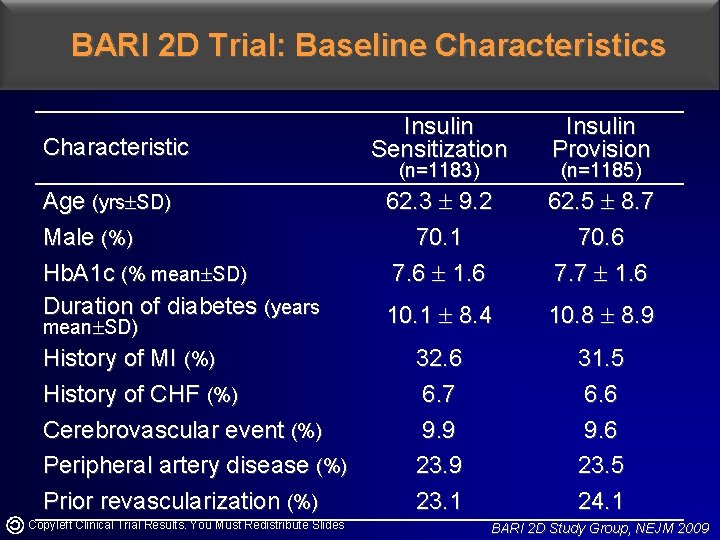
BARI 2 D Trial: Baseline Characteristics Characteristic Age (yrs SD) Male (%) Hb. A 1 c (% mean SD) Duration of diabetes (years mean SD) History of MI (%) History of CHF (%) Cerebrovascular event (%) Peripheral artery disease (%) Prior revascularization (%) Copyleft Clinical Trial Results. You Must Redistribute Slides Insulin Sensitization Insulin Provision 62. 3 9. 2 70. 1 7. 6 1. 6 62. 5 8. 7 70. 6 7. 7 1. 6 10. 1 8. 4 10. 8 8. 9 32. 6 6. 7 9. 9 23. 1 31. 5 6. 6 9. 6 23. 5 24. 1 (n=1183) (n=1185) BARI 2 D Study Group, NEJM 2009
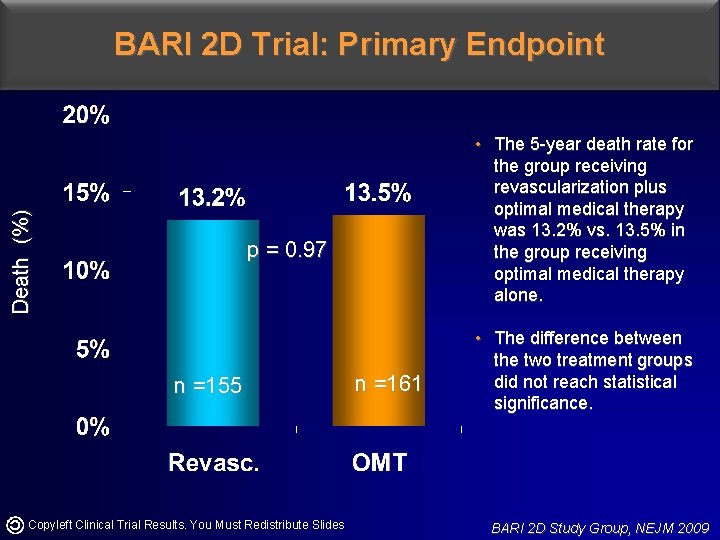
Death (%) BARI 2 D Trial: Primary Endpoint • The 5 -year death rate for the group receiving revascularization plus optimal medical therapy was 13. 2% vs. 13. 5% in the group receiving optimal medical therapy alone. p = 0. 97 n =155 Copyleft Clinical Trial Results. You Must Redistribute Slides n =161 • The difference between the two treatment groups did not reach statistical significance. BARI 2 D Study Group, NEJM 2009
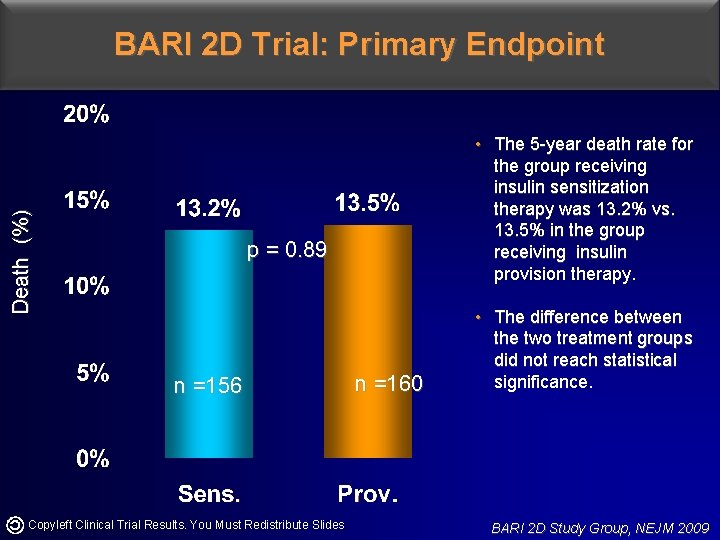
Death (%) BARI 2 D Trial: Primary Endpoint • The 5 -year death rate for the group receiving insulin sensitization therapy was 13. 2% vs. 13. 5% in the group receiving insulin provision therapy. p = 0. 89 n =156 Copyleft Clinical Trial Results. You Must Redistribute Slides n =160 • The difference between the two treatment groups did not reach statistical significance. BARI 2 D Study Group, NEJM 2009
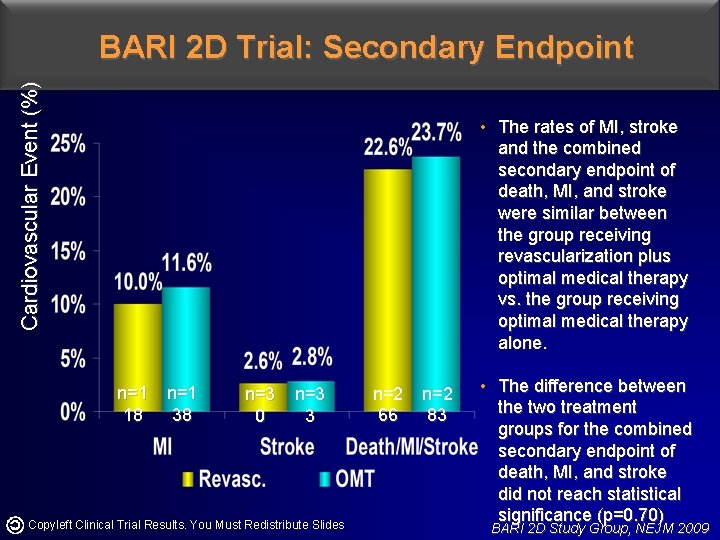
Cardiovascular Event (%) BARI 2 D Trial: Secondary Endpoint • The rates of MI, stroke and the combined secondary endpoint of death, MI, and stroke were similar between the group receiving revascularization plus optimal medical therapy vs. the group receiving optimal medical therapy alone. n=1 18 n=1 38 n=3 0 n=3 3 Copyleft Clinical Trial Results. You Must Redistribute Slides n=2 66 n=2 83 • The difference between the two treatment groups for the combined secondary endpoint of death, MI, and stroke did not reach statistical significance (p=0. 70) BARI 2 D Study Group, NEJM 2009
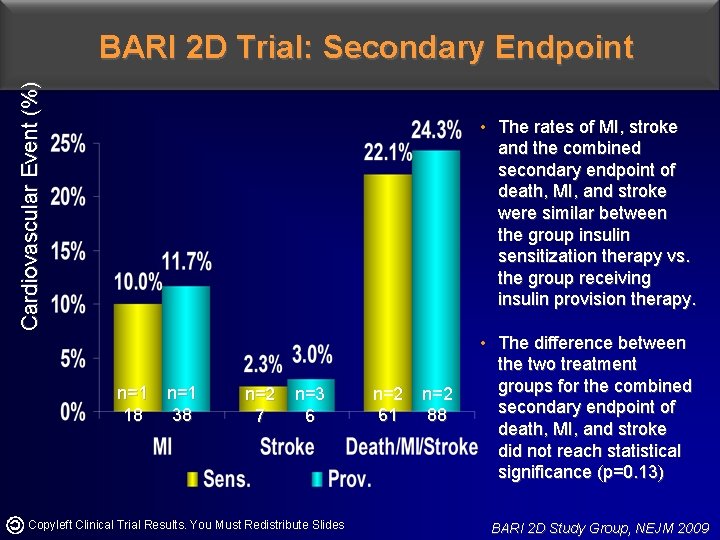
Cardiovascular Event (%) BARI 2 D Trial: Secondary Endpoint • The rates of MI, stroke and the combined secondary endpoint of death, MI, and stroke were similar between the group insulin sensitization therapy vs. the group receiving insulin provision therapy. n=1 18 n=1 38 n=2 7 n=3 6 Copyleft Clinical Trial Results. You Must Redistribute Slides n=2 61 n=2 88 • The difference between the two treatment groups for the combined secondary endpoint of death, MI, and stroke did not reach statistical significance (p=0. 13) BARI 2 D Study Group, NEJM 2009

BARI 2 D Trial: Limitations • Patients who are at high risk for MI and, therefore, stand to benefit the most from revascularization were excluded from the trial. • The broad applicability of BARI 2 D is limited by the fact that the patient population selected represents only a small subset of patients with diabetes and coronary artery disease. Copyleft Clinical Trial Results. You Must Redistribute Slides BARI 2 D Study Group, NEJM 2009

BARI 2 D Trial: Summary • Neither revascularization nor optimal medical therapy demonstrated a significant reduction in the occurrence of death or major cardiovascular events at 5 years. • Likewise, there was no demonstrated difference between a strategy of insulin sensitization and insulin provision. • Further research into defining optimal medical therapy for this group of patients is needed, as evidenced by the fact that 42. 1% of patients in the optimal medical therapy alone treatment group eventually required revascularization within 5 years. Copyleft Clinical Trial Results. You Must Redistribute Slides BARI 2 D Study Group, NEJM 2009
- Slides: 11