THE BUILDING BLOCKS OF LIFE Macromolecules CARBON Carbon
THE BUILDING BLOCKS OF LIFE Macromolecules

CARBON Carbon is a component of almost all biological molecules Life on earth is considered “carbon- based”.
MACROMOLECULES Large molecules that are formed by joining smaller organic molecules together Polymer – made from repeating subunits Important Macromolecules: � Carbohydrates � Proteins � Fats or Lipids � Nucleic Acids

ACTIVITY – MACROMOLECULES USE PP. 166 -171 Obtain four index cards and 5 color pencils Write the name of a macromolecule in big letters on each of the index cards Draw a labeled diagram of the molecule. Carbohydrates - draw the ring and chain structure of a monosaccharide. Draw a disaccharide. Proteins – Draw two amino acids joined together. Label the Carboxyl groups, Amino groups, and a peptide bond. Nucleic acid – Draw a simple nucleotide (sugar, phosphate, base) Lipids – Draw saturated and unsaturated and label important subunits. On the other side: � � Subunits: Elements present: Identify different types: (carbs, lipids, nucleic acids only) Give examples and their function in living organisms:
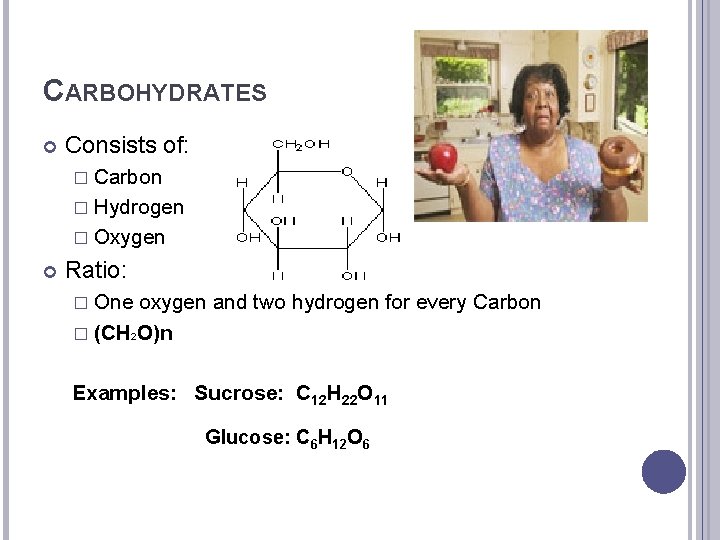
CARBOHYDRATES Consists of: � Carbon � Hydrogen � Oxygen Ratio: � One oxygen and two hydrogen for every Carbon � (CH 2 O)n Examples: Sucrose: C 12 H 22 O 11 Glucose: C 6 H 12 O 6
CARBOHYDRATES Types: � Monosaccharide Glucose – provides energy to most living organisms � Disacharide – simple sugar – Two simple sugars bound together Lactose and sucrose � Polysaccaride - long chain “many” Glycogen – storage in liver or muscle Starch – storage in plants Cellulose - Makes up outer boundary of a plant cell
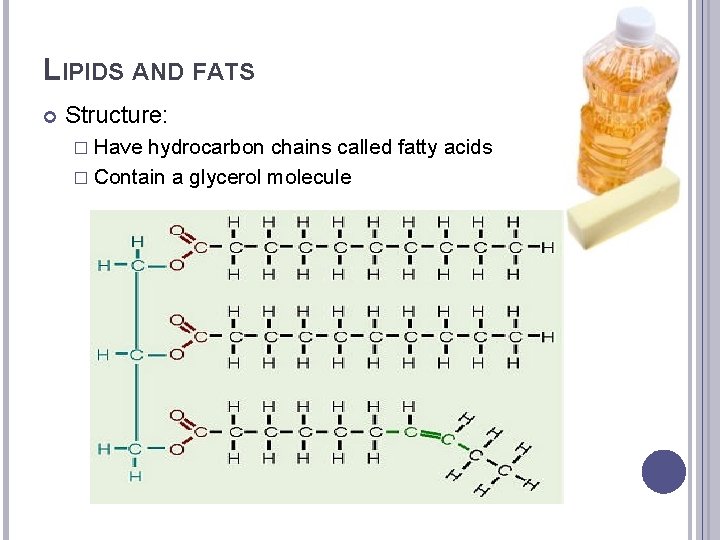
LIPIDS AND FATS Structure: � Have hydrocarbon chains called fatty acids � Contain a glycerol molecule

LIPIDS AND FATS Types: � Saturated – Animals store energy as saturated fats. The fatty acids stick to arterial walls and can lead to cardiovascular disease if not consumed in a cautious manner. � Unsaturated Fats: Plants store energy in unsaturated fats. These fats don’t stick to arterial walls in the same way as saturated fats do.

LIPIDS AND FATS Roles in living organisms: � Energy storage � Insulation � Structure of the cell membrane (phospholipid) � Steroids Starting point for vitamin D and hormones (estrogen and testosterone) Adds stability to the cell membrane
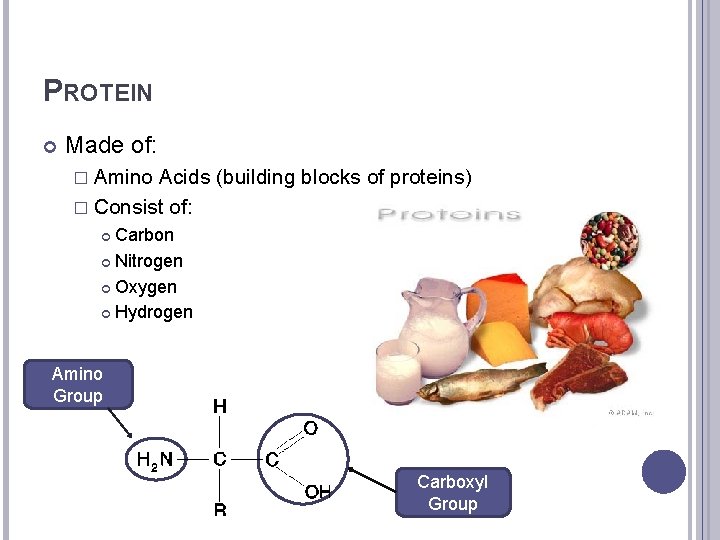
PROTEIN Made of: � Amino Acids (building blocks of proteins) � Consist of: Carbon Nitrogen Oxygen Hydrogen Amino Group Carboxyl Group
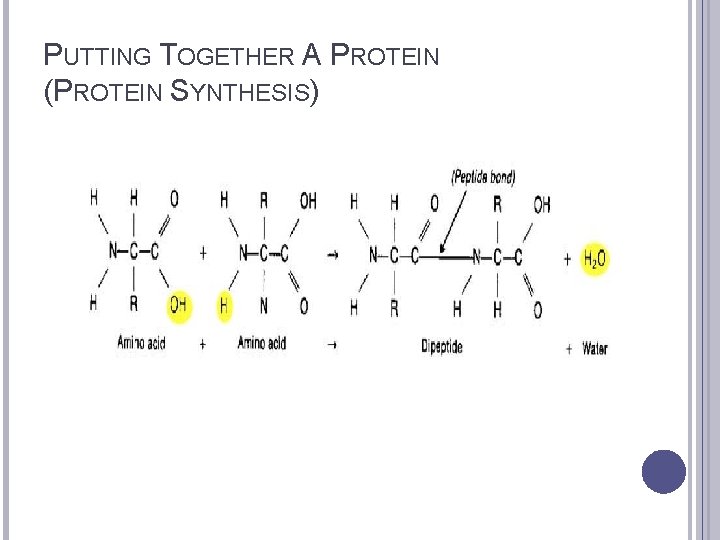
PUTTING TOGETHER A PROTEIN (PROTEIN SYNTHESIS)
PROTEIN Examples and Function � Speed up reactions – enzymes in digestion � Transport substances (hemoglobin) � Provide structural support (collagen) � Make hormones – (estrogen, testosterone) � IMPROTANT: Amino Acids are the building blocks of proteins!

ENZYMES Substances that help speed up chemical reactions Substrate: The compound to be broken down or put together http: //highered. mcgrawhill. com/sites/0072495855/student_view 0/chapter 2/ animation__how_enzymes_work. html
NUCLEIC ACIDS Store and transmit genetic information Examples: DNA & RNA Made of nucleotides that contain: � Carbon � Oxygen � Nitrogen � Phosphorus � Hydrogen
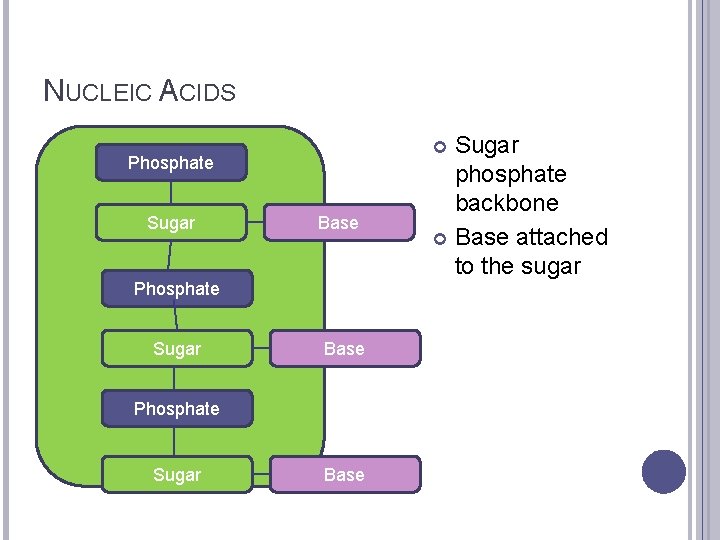
NUCLEIC ACIDS Phosphate Sugar Base Phosphate Sugar phosphate backbone Base attached to the sugar Base
- Slides: 15