The Buffering Balance Modeling Arctic river total inorganicand

The Buffering Balance: Modeling Arctic river total-, inorganicand organic-alkalinity fluxes C. W. Hunt, J. E. Salisbury, W. Wollheim, M. Mineau, and R. J. Stewart. University of New Hampshire
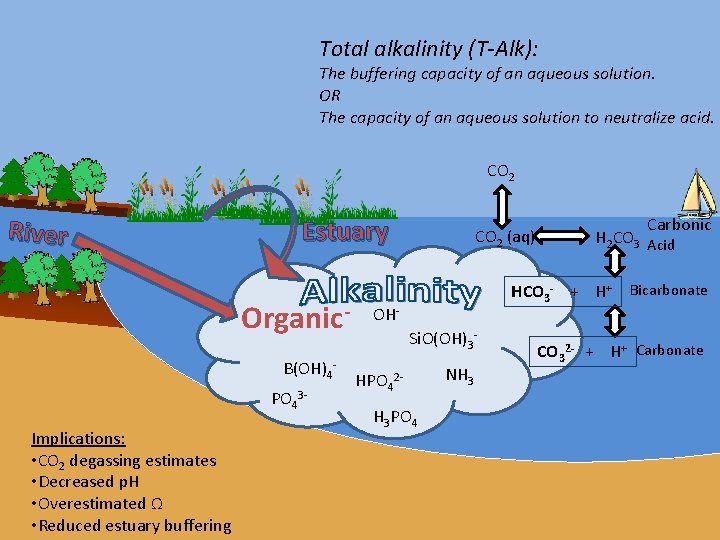
Total alkalinity (T-Alk): The buffering capacity of an aqueous solution. OR The capacity of an aqueous solution to neutralize acid. CO 2 River Estuary Organic. B(OH)4 PO 43 Implications: • CO 2 degassing estimates • Decreased p. H • Overestimated Ω • Reduced estuary buffering CO 2 (aq) Carbonic H 2 CO 3 Acid HCO 3 - + H+ Bicarbonate OHSi. O(OH)3 HPO 42 H 3 PO 4 NH 3 CO 32 - + H+ Carbonate
![How is alkalinity defined? Acid-Neutralizing Definition: T-Alk = [HCO 3−] + 2[CO 3− 2] How is alkalinity defined? Acid-Neutralizing Definition: T-Alk = [HCO 3−] + 2[CO 3− 2]](http://slidetodoc.com/presentation_image/26423670b1b032de1fa45b94906d8aa9/image-3.jpg)
How is alkalinity defined? Acid-Neutralizing Definition: T-Alk = [HCO 3−] + 2[CO 3− 2] + [B(OH)4−] + 2[PO 4− 3] + [HPO 4− 2] + [Si. O(OH)3−] …+ [Organic-] Ion Balance Definition [HCO 3−] + 2[CO 3− 2] +[OH-]- [H+ ] = [Na+]+[K+]+ 2[Ca+2] + 2[Mg+2] - [Cl-] - 2[SO 4 -2] - [Organic-] Working Definitions: C-Alk = [HCO 3−] + 2[CO 3− 2] ≈ [HCO 3−] NC-Alk = [B(OH)4−] + 2[PO 4− 3] + [HPO 4− 2] + [Si. O(OH)3−] + [Organic-] Org-Alk = [Organic-] ≈ NC-Alk Org-Alk% = [Org-Alk] / [Total Alk]
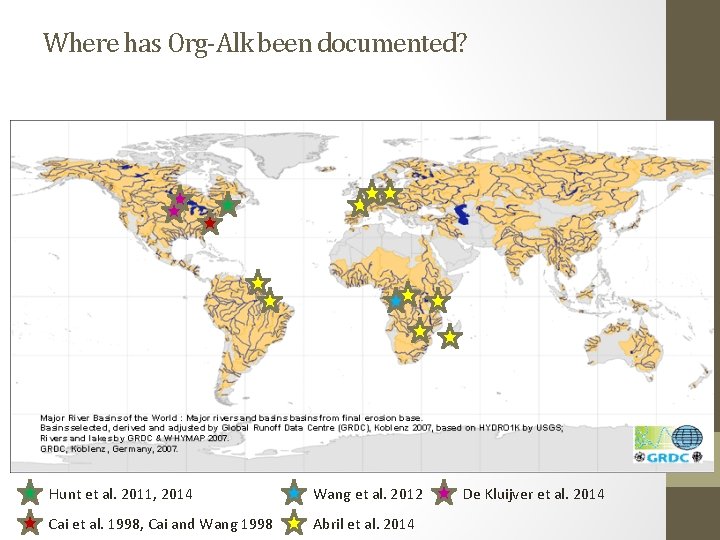
Where has Org-Alk been documented? Hunt et al. 2011, 2014 Wang et al. 2012 Cai et al. 1998, Cai and Wang 1998 Abril et al. 2014 De Kluijver et al. 2014
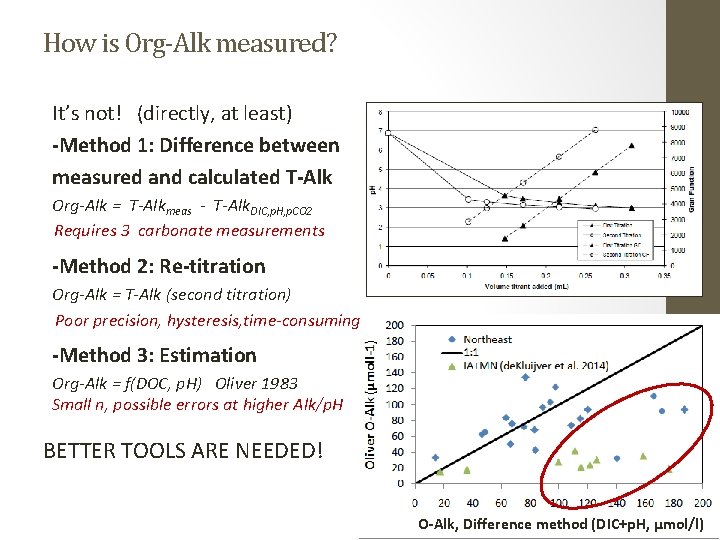
How is Org-Alk measured? It’s not! (directly, at least) -Method 1: Difference between measured and calculated T-Alk Org-Alk = T-Alkmeas - T-Alk. DIC, p. H, p. CO 2 Requires 3 carbonate measurements -Method 2: Re-titration Org-Alk = T-Alk (second titration) Poor precision, hysteresis, time-consuming -Method 3: Estimation Org-Alk = f(DOC, p. H) Oliver 1983 Small n, possible errors at higher Alk/p. H BETTER TOOLS ARE NEEDED! O-Alk, Difference method (DIC+p. H, µmol/l)
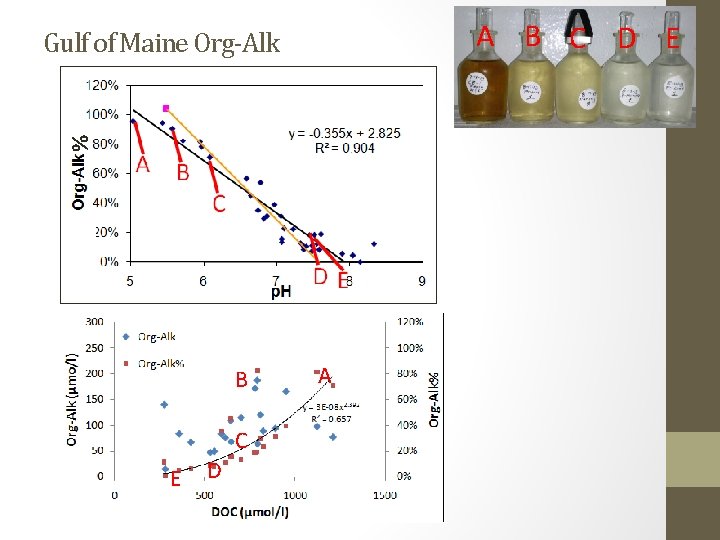
A B C D E % Gulf of Maine Org-Alk B C E D A
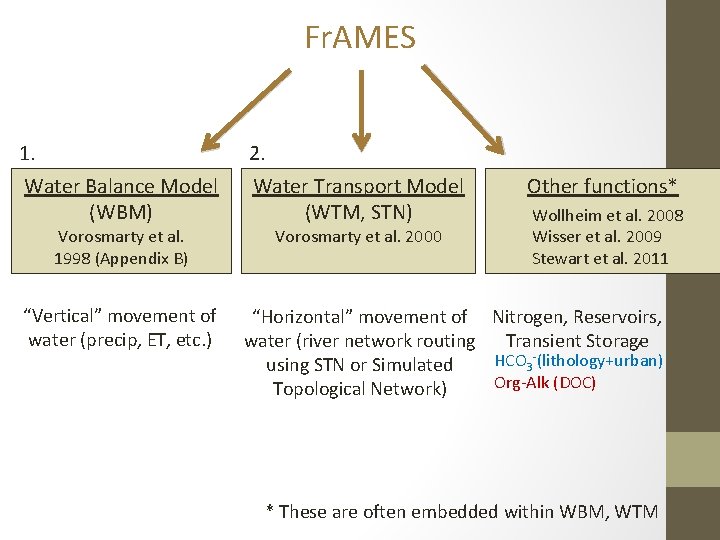
Fr. AMES 1. 2. Water Balance Model (WBM) Water Transport Model (WTM, STN) “Vertical” movement of water (precip, ET, etc. ) “Horizontal” movement of water (river network routing using STN or Simulated Topological Network) Vorosmarty et al. 1998 (Appendix B) Vorosmarty et al. 2000 Other functions* Wollheim et al. 2008 Wisser et al. 2009 Stewart et al. 2011 Nitrogen, Reservoirs, , Transient Storage HCO 3 -(lithology+urban) Org-Alk (DOC) * These are often embedded within WBM, WTM
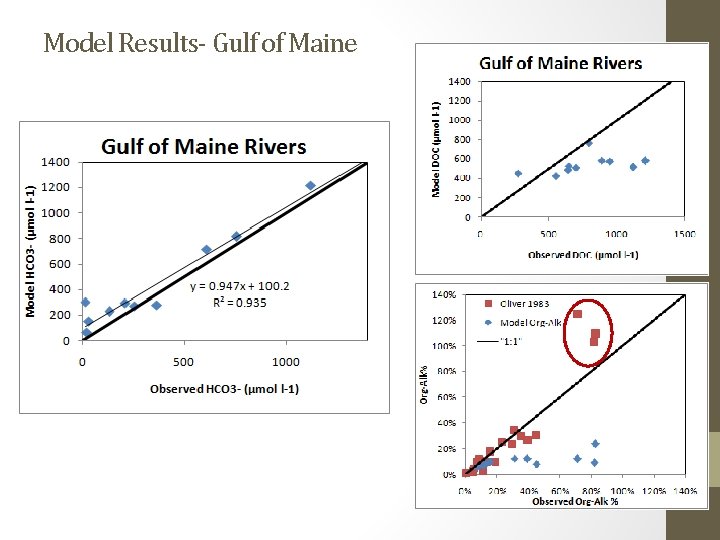
Model Results- Gulf of Maine
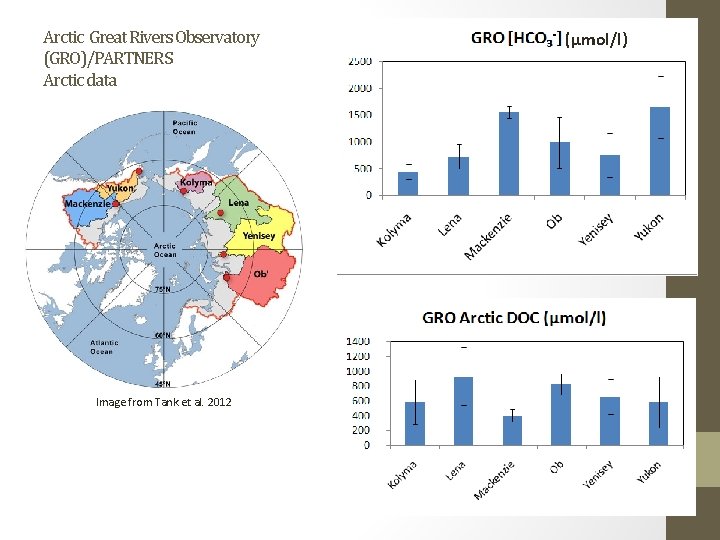
Arctic Great Rivers Observatory (GRO)/PARTNERS Arctic data Image from Tank et al. 2012 (µmol/l)
![Arctic Great Rivers Observatory (GRO)/PARTNERS Arctic data Oliver 1983: [Org-Alk] = (10 -p. H Arctic Great Rivers Observatory (GRO)/PARTNERS Arctic data Oliver 1983: [Org-Alk] = (10 -p. H](http://slidetodoc.com/presentation_image/26423670b1b032de1fa45b94906d8aa9/image-10.jpg)
Arctic Great Rivers Observatory (GRO)/PARTNERS Arctic data Oliver 1983: [Org-Alk] = (10 -p. H )(DOC*10) (10 -p. H ) + K Kolyma Yenisey Yukon Mackenzie DOC (µmol C/l) Ob Lena
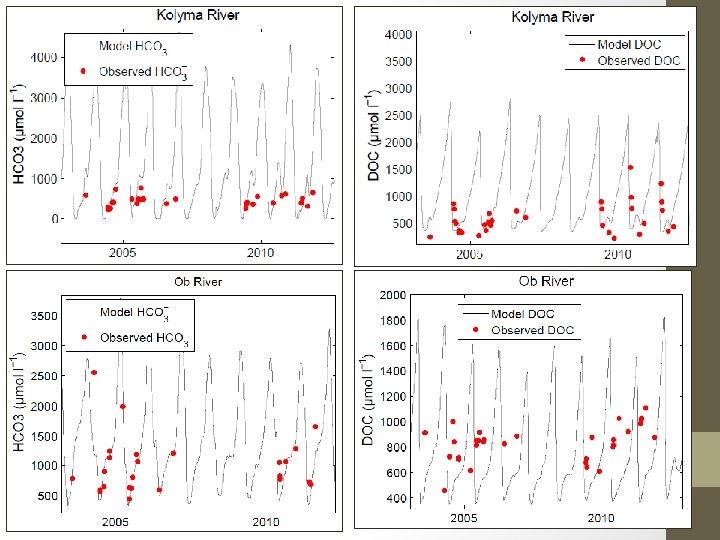
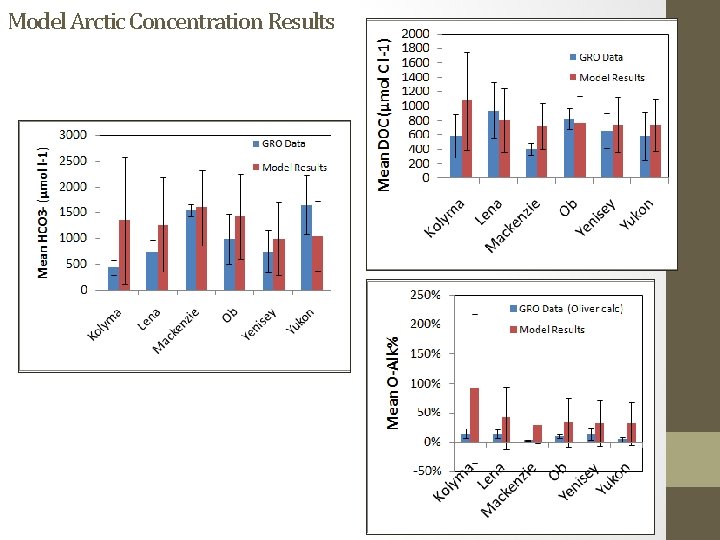
Model Arctic Concentration Results
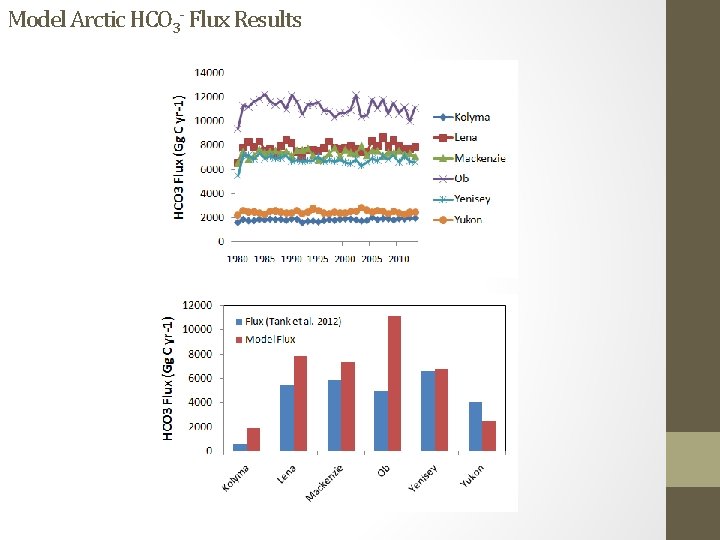
Model Arctic HCO 3 - Flux Results
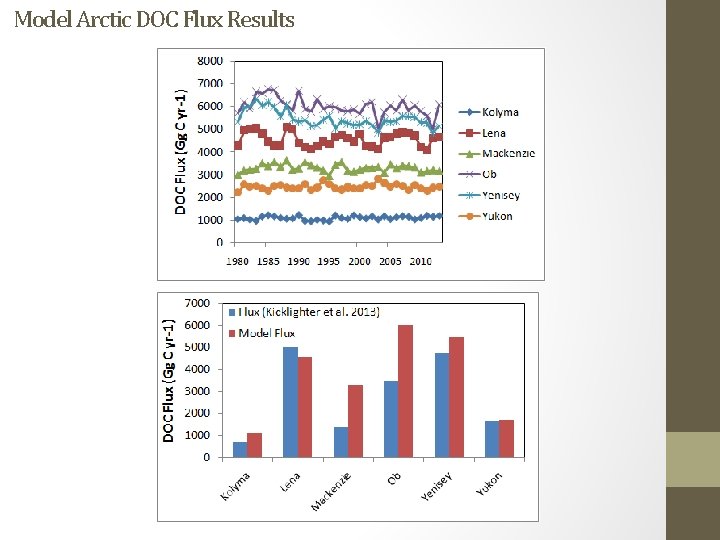
Model Arctic DOC Flux Results

Conclusions • Gulf of Maine DOC/Org-Alk% relationship most likely not appropriate for Arctic rivers • More Arctic validation data needed, especially DIC or p. CO 2 Future Goals • Hope to incorporate DOC quality- remote sensing opportunities • Calibrate model coefficients for Arctic setting • Including a permafrost parameter

Arctic - COLORS Arctic-Coastal Land Ocean Interactions A NASA Scoping Study Grants NASA NNX 14 AD 75 G and NNX 09 AU 89 G

Questions? • I have many, such as: • What factors are missing in the model for DOC, HCO 3? • Is there a better way to model Arctic O-Alk? • We eventually need DOC quality in order to understand DOC color signature. Can SUVA get us there? • What data are needed to improve understanding of Artic O-Alk? • Strategies for modeling Arctic river p. H? • How can we simulate the potential release of DOC from thawing permafrost?

References • • • • Abril, G. , S. Bouillon, F. Darchambeau, C. R. Teodoru, T. R. Marwick, F. Tamooh, F. Ochieng Omengo, N. Geeraert, L. Deirmendjian, P. Polsenaere, and A. V. Borges. 2014. Technical Note: Large overestimation of p. CO 2 calculated from p. H and alkalinity in acidic, organic-rich freshwaters. Biogeosciences bg-2014 -341 Amiotte-Suchet, P. , J. -L. Probst, and W. Ludwig (2003), Worldwide distribution of continental rock lithology: Implications for the atmospheric/soil CO 2 uptake by continental weathering and alkalinity river transport to the oceans, Global Biogeochem. Cycles, 17(2), 1038, doi: 10. 1029/2002 GB 001891. Cai, W. -J. and Wang, Y. : The chemistry, fluxes and sources of carbon dioxide in the estuarine waters of the Satilla and Altamaha Rivers, Georgia, Limnol. Oceanogr. , 43, 657– 668, 1998. Cai, W. -J. , Wang, Y. , and Hodson, R. E. : Acid-base properties of dissolved organic matter in the estuarine waters of Georgia, USA, Geochim. Cosmochim. Ac. , 62, 473– 483, 1998. de Kluijver, A. , Schoon, P. L. , Downing, J. A. , Schouten, S. , and Middelburg, J. J. : Stable carbon isotope biogeochemistry of lakes along a trophic gradient, Biogeosciences, 11, 6265 -6276, doi: 10. 5194/bg-11 -6265 -2014, 2014. Hartmann, Jörg; Moosdorf, Nils (2012): Global Lithological Map Database v 1. 0 (gridded to 0. 5° spatial resolution). doi: 10. 1594/PANGAEA. 788537, Supplement to: Hartmann, Jens; Moosdorf, Nils (2012): The new global lithological map database GLi. M: A representation of rock properties at the Earth surface. Geochemistry, Geophysics, Geosystems, 13, Q 12004, doi: 10. 1029/2012 GC 004370 Hunt, C. W. , J. E. Salisbury and D. Vandemark. (2013) CO 2 Input Dynamics and Air-Sea Exchange in a Large New England Estuary. Estuaries and Coasts 37(5): 1078 -1091 C. W. Hunt, J. E. Salisbury, D. Vandemark. (2011) Contribution of non-carbonate anions to total alkalinity and overestimation of p. CO 2 in New England New Brunswick rivers. Biogeosciences, doi: 10. 5194/bg-8 -3069 -2011 Kicklighter, DW, Hayes, DJ, Mc. Clelland, JW, Peterson, BJ, Mc. Guire, AD and JM Melillo. 2013. Insights and issues with simulating terrestrial DOC loading of Arctic river networks. Ecological Applications 23(8): 1817 -1836. Tank, S. E. , P. A. Raymond, R. G. Striegl, J. W. Mc. Clelland, R. M. Holmes, G. J. Fiske, and B. J. Peterson (2012), A land-to-ocean perspective on the magnitude, source and implication of DIC flux from major Arctic rivers to the Arctic Ocean, Global Biogeochem. Cycles, 26, GB 4018, doi: 10. 1029/2011 GB 004192. Wang, Z. A. , D. J. Bienvenu, P. J. Mann, K. A. Hoering, J. R. Poulsen, R. G. M. Spencer, and R. M. Holmes (2013), Inorganic carbon speciation and fluxes in the Congo River, Geophys. Res. Lett. , 40, 511– 516, doi: 10. 1002/grl. 50160.

Loading equation: DOC (µmol/l) = Constant * ROSlope =1 - (0. 0001 WL 2 – 0. 0193*WL + 1. 4237) Constant = -0. 002 WL 2 + 0. 4552*WL + 7. 275 RO=Runoff (mm/d), WL=Wetland% HCO 3 (µmol/l) = Lithological HCO 3 - load (Amiotte-Suchet et al. 2003) Global 1° lithology (Hartmann and Moosdorf 2012) Respiration Removal of DOC: Resp. Vf (m/d) = KResp * Resp. Q 10(water. T - Resp_Tref) / 10 Resp. Removal = DOC * (1 - 10(-Resp. Vf / HL)) HL=Hydraulic Load (m/d) Photolysis Removal of DOC: Photo. Vf (m/d) = KPhoto * PAR * (Photo. Depth * Channel. Depth) Photo. Removal = HPOA * (1 - 10(-Photo. Vf / HL))
- Slides: 19