The Breathalyzer The Breathalyzer The Breathalyzer Reaction C

The Breathalyzer

The Breathalyzer

The Breathalyzer Reaction C 2 H 5 OH + Cr 2 O 72 - + H+ → CH 3 COOH + Cr 3+ + H 2 O Ethanol Orange Acid Acetic Acid Green Water If the orange colour decreases there is alcohol present. It is measured with a spectrophotometer. The more it decreases, the higher the blood alcohol content. Legal Limit Class 5 Licence 0. 08 mg/m. L Legal Limit New Driver 0. 00 mg/m. L

Test tubes each contain 5 m. L 0. 25 M K 2 Cr 2 O 7 + 5 m. L of 6. 0 M H 2 SO 4 + 1 drop of 0. 10 M Ag. NO 3 (catalyst) and 0, 1, 2, 3, 4, drops respectively of 10. 0 % ethanol. 0. 000 % 0. 05 % 0. 10 % 0. 15 % 0. 20 %

Redox Titrations A redox titration is the same as an acid-base titration except it involves a redox reaction. Reagents are chosen so that the reaction is spontaneous. Pick a suitable reagent for redox titration involving IO 3 - in acid solution. A FB I- C SO 42 D Cl

Pick the spontaneous reaction

6. 75 m. L of 0. 100 M KMn. O 4 is required to titrate 25. 0 m. L of Fe. Cl 2. Calculate the [Fe 2+]. Mn. O 4 - + 8 H+ + 5 Fe 2+ → Mn 2+ + 4 H 2 O + 5 Fe 3+ 0. 00675 L 0. 0250 L 0. 100 M ? M 0. 00675 L Mn. O 4 - x 0. 100 mole x 5 moles Fe 2+ 1 L 1 mole Mn. O 4 [Fe 2+] = 0. 0250 L = 0. 135 M
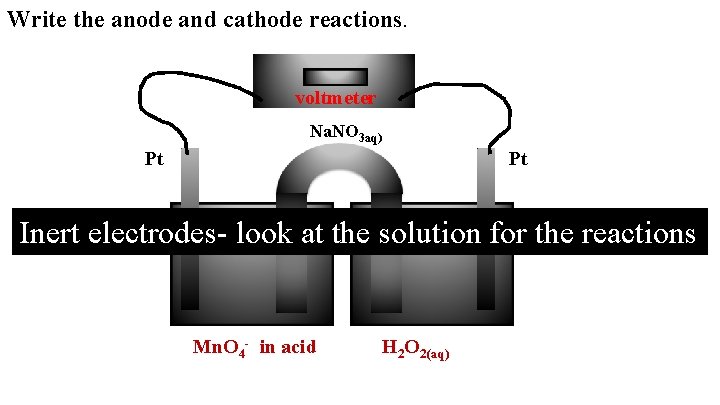
Write the anode and cathode reactions. voltmeter Na. NO 3 aq) Pt Pt Inert electrodes- look at the solution for the reactions Mn. O 4 - in acid H 2 O 2(aq)
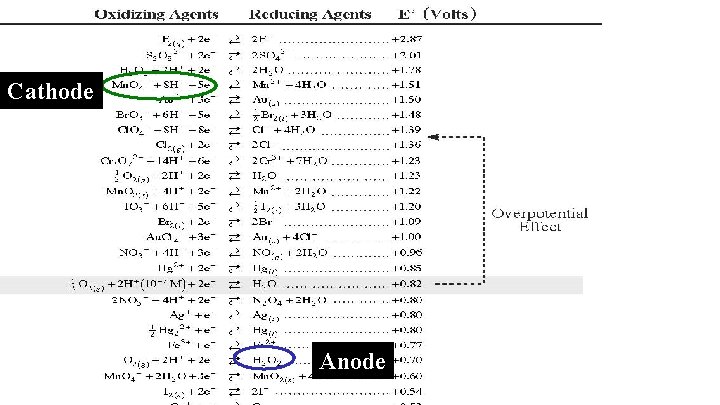
Cathode Anode

Cathode: Mn. O 4 - + 8 H+ + 5 e- → Anode: H 2 O 2 → O 2 + 2 H+ + 2 e- What happens to the mass of the cathode? Constant What happens to the mass of the anode? Constant What happens to the p. H of the cathode? Increases What happens to the p. H of the anode? Decreases Mn 2+ + 4 H 2 O
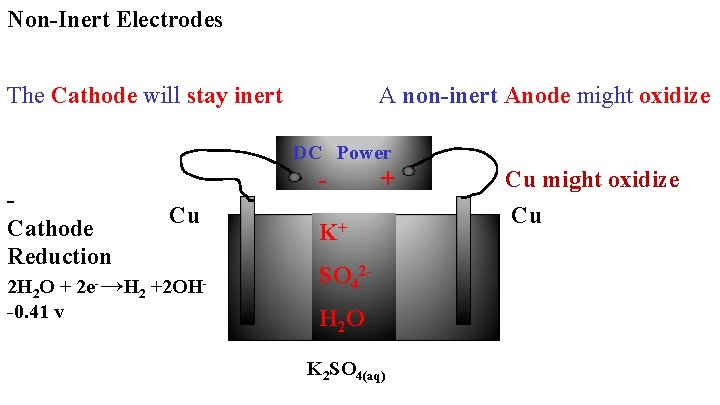
Non-Inert Electrodes The Cathode will stay inert A non-inert Anode might oxidize DC Power Cu Cathode Reduction 2 H 2 O + 2 e-→H 2 +2 OH-0. 41 v - + K+ SO 42 H 2 O K 2 SO 4(aq) Cu might oxidize Cu

You must look at the possible oxidation of: SO 42 - H 2 O Cu Strongest Reducing Agent
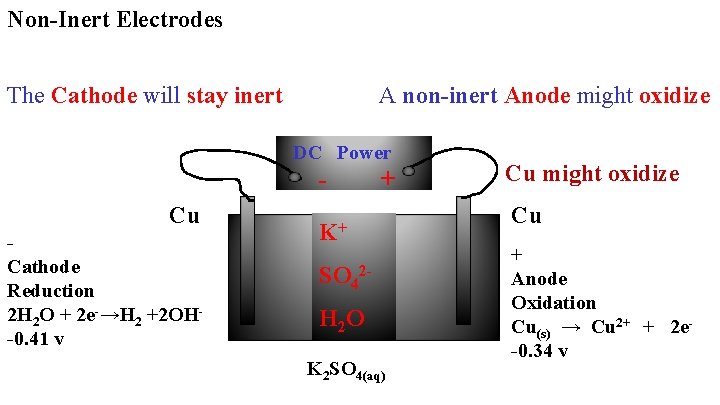
Non-Inert Electrodes The Cathode will stay inert A non-inert Anode might oxidize DC Power - + Cu Cathode Reduction 2 H 2 O + 2 e-→H 2 +2 OH-0. 41 v K+ SO 42 H 2 O K 2 SO 4(aq) Cu might oxidize Cu + Anode Oxidation Cu(s) → Cu 2+ + 2 e-0. 34 v

Review of Cells Electrochemical Is a power supply Spontaneous (+) Makes electricity Electrolytic Requires power supply Nonspontaneous (-) Makes chemicals Reduction is highest on Chart Reduction is the –ve

For all cells: Cations migrate to the cathode, which is the site of reduction. Anions migrate to the anode, which is the site of oxidation. Electrons travel through the wire from anode to cathode.

Complete the Chart Electrochemical Cell: Zn, Zn(NO 3)2 II Cu, Cu. SO 4 Anode: Zn Reaction: Zn(s) → Zn 2+ + 2 e- 0. 76 v Cathode: Cu Reaction: Cu 2+ + 2 e- → Cu(s) 0. 34 v E 0 1. 10 v = Higher on reduction Chart

Electrolytic Cell: Molten Al. Cl 3 Anode: Reaction: C Cathode: Reaction: C E 0 = -3. 02 v Al 3+ Cl - → Cl 2(g) + 2 e 2 Cl -1. 36 v Al 3+ + 3 e- → Al(s) -1. 66 v MTV = The anode and cathode are inert C or Pt Vowels: Anode Anion Consonants: Cathode Cation +3. 02 v Oxidation Reduction

Electrolytic Cell: KBr(aq) Anode: Cathode: K+ Br- H 2 O C C Anode Oxidation Anion or Water
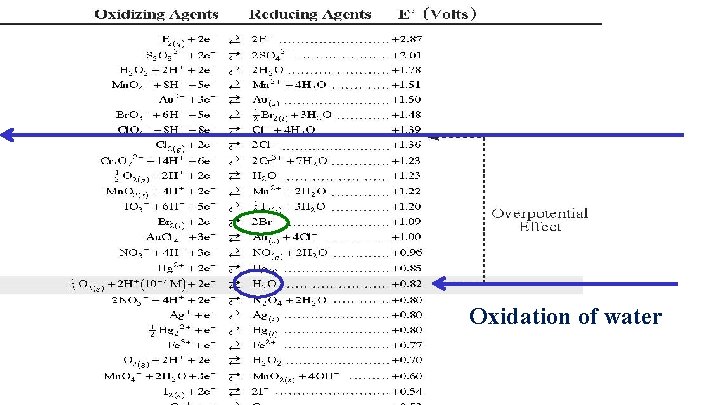
Oxidation of water

Electrolytic Cell: KBr(aq) K+ Anode: Cathode: C 2 Br- → Br 2(g) + 2 e- Br- H 2 O C Anode Oxidation Cathode Reduction Anion or Water Cation or water -1. 09 v


Electrolytic Cell: KBr(aq) K+ Anode: Cathode: E 0 = Br- H 2 O C 2 Br- → Br 2(g) + 2 e- -1. 09 v C 2 H 2 O + 2 e- → H 2(g) + 2 OH- -0. 41 v -1. 50 v Anode Oxidation Cathode Reduction MTV = +1. 50 v Anion or Water Cation or water

Is Al a reactive or non-reactive metal? Look on page 8 Reactive as Al is a relatively strong reducing agent. Why is Al used for boats, patio furniture, swing sets, and trucks boxes? Al makes a clear transparent Al 2 O 3 paint like coating that prevents further oxidation.

How to Fail a Breathalyzer Video 1 Breathalyzer Video 2 Drunks Jeff Dunham
- Slides: 24