The Bohr Model of the Atom DO NOT

The Bohr Model of the Atom DO NOT WRITE ON THESE NOTES COPY THESE NOTES IN CORNELL STYLE IN YOURBINDER.

What do we need to know? • Atoms have 2 main parts – Electron cloud -- contains electrons – Nucleus – central part of the atom, contains protons and neutrons

Subatomic particles • Symbols for each subatomic particle – Electrons = e– Protons = p+ – Neutrons = n 0 (found in the electron cloud) (found in the nucleus)

What do you need to know? • Orbitals are found around the nucleus – There are 7 possible orbitals – Orbitals are determined by looking at what PERIOD (row) the element is in – The orbitals hold a specific number of electrons (which is the same as the atomic number) Period 1 Period 2 Period 3 Period 4 Period 5 Period 6 Period 7

How many electrons per orbital? Period 1 orbital (ring) 1: 2 e- Period 2 orbital (ring) 1: 2 eorbital (ring) 2: 8 e- Period 3 orbital (ring) 1: 2 eorbital (ring) 2: 8 eorbital (ring) 3: 8 e- Period 4 orbital (ring) 1: 2 eorbital (ring) 2: 8 eorbital (ring) 3: 18 eorbital (ring) 4: 8 e-
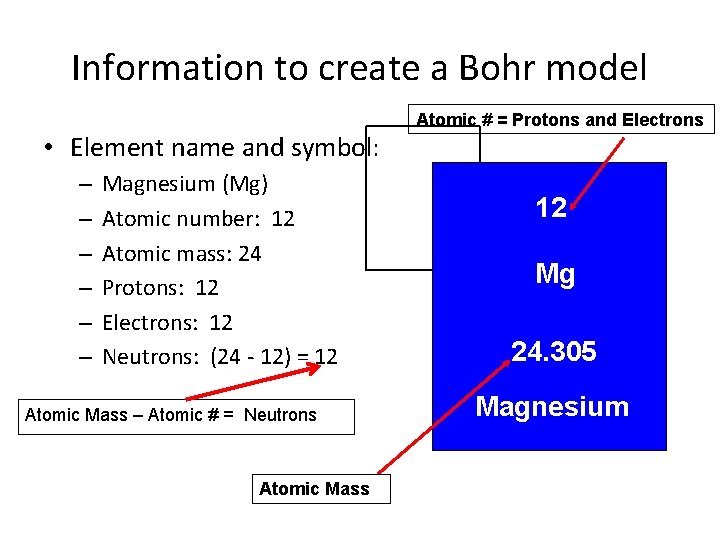
Information to create a Bohr model • Element name and symbol: – – – Magnesium (Mg) Atomic number: 12 Atomic mass: 24 Protons: 12 Electrons: 12 Neutrons: (24 - 12) = 12 Atomic Mass – Atomic # = Neutrons Atomic Mass Atomic # = Protons and Electrons 12 Mg 24. 305 Magnesium
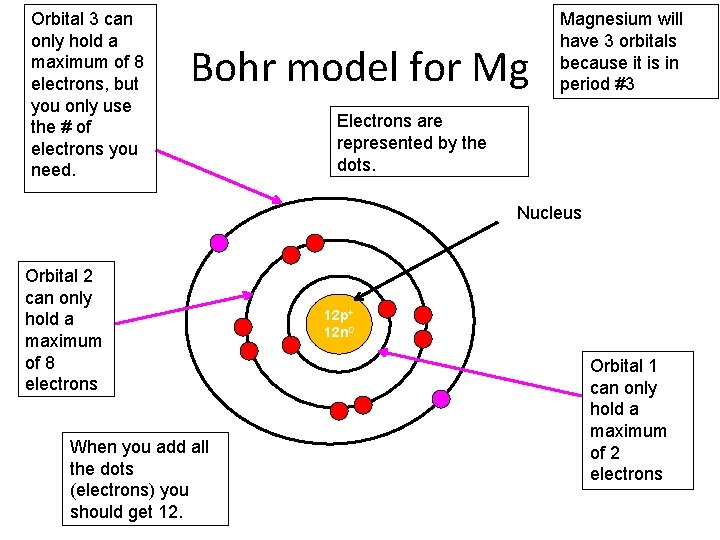
Orbital 3 can only hold a maximum of 8 electrons, but you only use the # of electrons you need. Bohr model for Mg Magnesium will have 3 orbitals because it is in period #3 Electrons are represented by the dots. Nucleus Orbital 2 can only hold a maximum of 8 electrons When you add all the dots (electrons) you should get 12. 12 p+ 12 n 0 Orbital 1 can only hold a maximum of 2 electrons
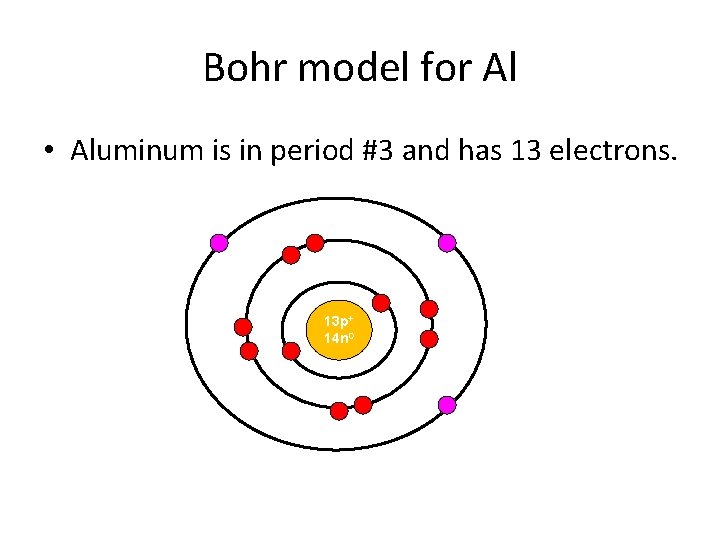
Bohr model for Al • Aluminum is in period #3 and has 13 electrons. 13 p+ 14 n 0

Your Assignment! This goes on a separate piece of paper! • Create the Bohr model for elements 1 -20 on the periodic table. (Hydrogen (H) – Calcium (Ca)) • For each element provide the: – Write the element name: _________ – Protons= ____ – Electrons= ____ – Neutrons= ____ – Period = _____ = Orbitals= _____ – Bohr Model:
- Slides: 9