The Bohr Model 1913 1913 A Danish scientist

The Bohr Model (1913) • 1913 – A Danish scientist Niels Bohr suggested revolve around the that the electrons ______ nucleus like the planets revolve around the sun _______. • In Bohr’s model each electron has a certain energy that is defined by its amount of ______ path around the nucleus. These defined orbits are called energy levels.

Bohr’s theory Planetary Model: Three rules: • Electrons orbit the nucleus like planets orbit the sun • Electrons can “jump” between orbits by absorbing or _____ emitting _____ photons • Theory explained the line spectra of H 1. Electrons only exist in certain allowed orbits 2. Within an orbit, the electron does not _______ radiate 3. Radiation (light) is emitted or absorbed when changing orbits
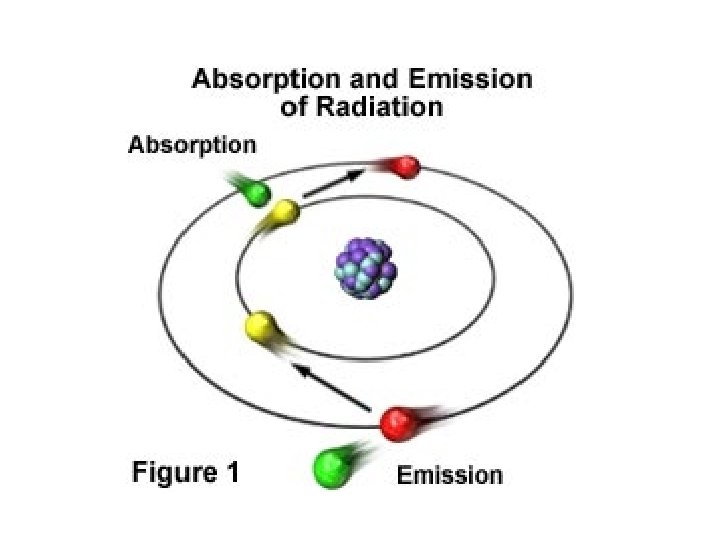
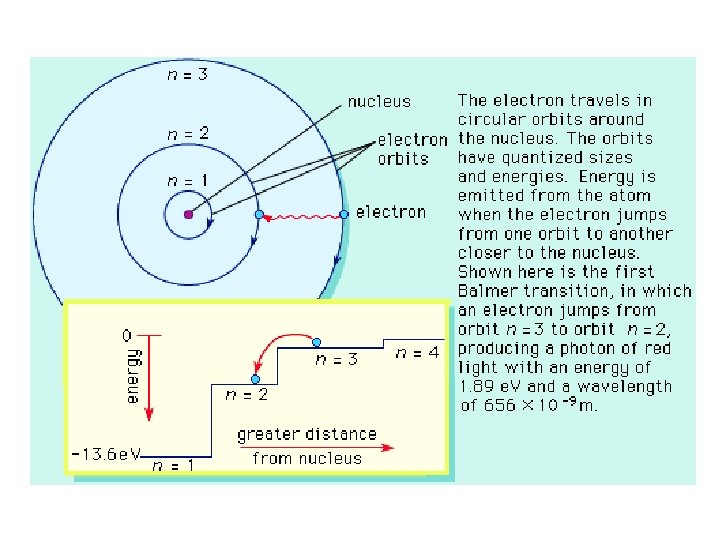

Atomic Spectra • When electrons absorb energy they move to higher energy levels, and when electrons lose energy by emitting light they return to their ground state.
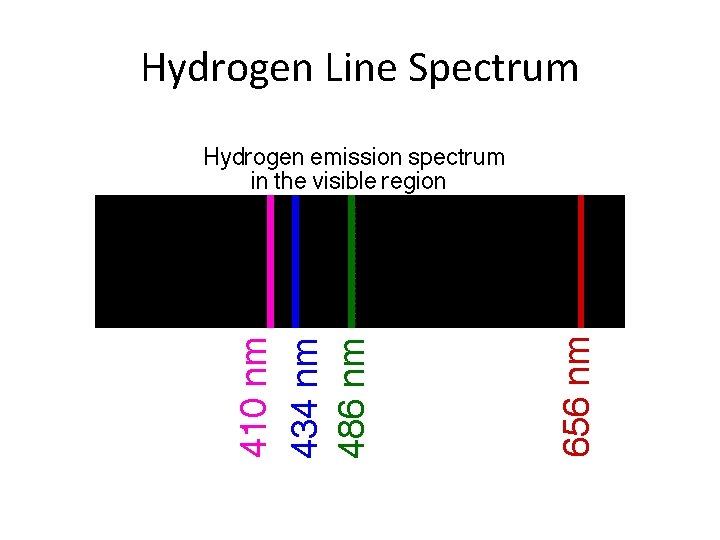
Hydrogen Line Spectrum
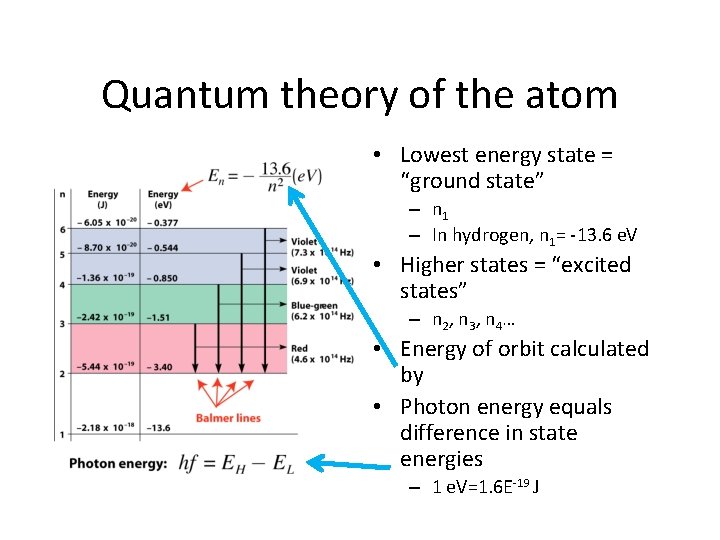
Quantum theory of the atom • Lowest energy state = “ground state” – n 1 – In hydrogen, n 1= -13. 6 e. V • Higher states = “excited states” – n 2 , n 3 , n 4 … • Energy of orbit calculated by • Photon energy equals difference in state energies – 1 e. V=1. 6 E-19 J

The Energy of a Photon • The energy of a photon is proportional to the frequency of light and can be calculated using Plank’s constant • E = hf, where h = 6. 63 x 10 -34 Js • How much energy would red light have with a frequency of 4. 57 x 1014 Hz?

How does this tie in with Bohr’s Model • The light emitted from an electron moving from a higher to a lower energy level has a frequency proportional ( ) to the energy change of the electron moving between energy levels

An electron in a hydrogen atom jumps from the exited energy level n = 4 to n = 2. What is the frequency and wavelength of the emitted photon?
- Slides: 10