THE BOHR ATOM ESSENTIAL QUESTION How can properties

THE BOHR ATOM

ESSENTIAL QUESTION • How can properties of a wave be used in energy calculations?

THE BOHR ATOM • Protons and neutrons are located in the nucleus • Electrons orbit the nucleus • Also called the “Planetary model” because it is similar to the solar system
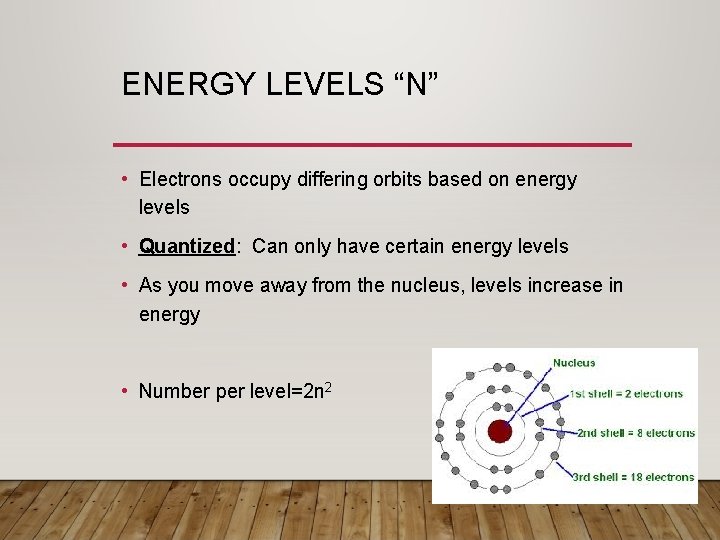
ENERGY LEVELS “N” • Electrons occupy differing orbits based on energy levels • Quantized: Can only have certain energy levels • As you move away from the nucleus, levels increase in energy • Number per level=2 n 2

LETS PRACTICE • Nitrogen: • How many electrons? • How many in the first energy level? • How many in the second energy level? • Draw the picture

YOU TRY • Calcium (Ca): • How many total electrons? • How many in the first energy level? • How many in the second energy level? • Third? • Draw the picture
TRY THESE! • Phosphorus: • Carbon: • Nitrogen:

• How are nitrogen and carbon similar? How are they different? • How are nitrogen and phosphorus similar? How are they different?
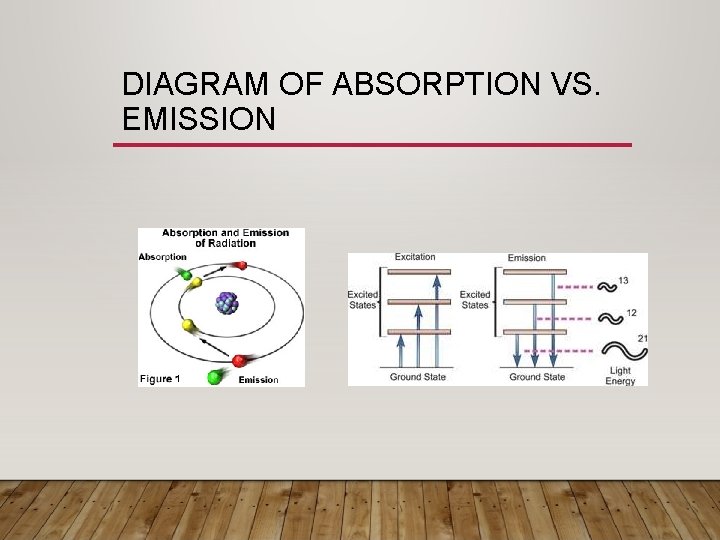
DIAGRAM OF ABSORPTION VS. EMISSION

ABSORPTION VS. EMISSION • Absorption • If energy is added, electrons can become excited and jump to higher energy levels. • They can move multiple energy levels • Emission: • Electrons are not stable in their excited state • Want to emit energy to return to their original state • Do this by emitting a photon (packet of light). • The color given off depends how many energy levels the electrons fall.

WHEN ELECTRONS ABSORB OR EMIT ENERGY, THEY ACT AS A WAVE

ELECTROMAGNETIC SPECTRUM

ESSENTIAL QUESTION • How can the properties of waves be used in energy calculations?

WHAT DO YOU ALREADY KNOW ABOUT THE ELECTROMAGNETIC SPECTRUM? ? • Based on the video, write down something you already know about the electromagnetic spectrum. We will turn and talk, then do a whip around. • https: //www. youtube. com/watch? v=P_PVz 8 Hrr. CI

VIDEO • In the video, write down something about each type of radiation. http: //www. youtube. com/watch? v=P_PVz 8 Hrr. CI • Gamma • X-rays • UV • visible • Infrared • Microwaves • Radio


VISIBLE LIGHT SPECTRUM
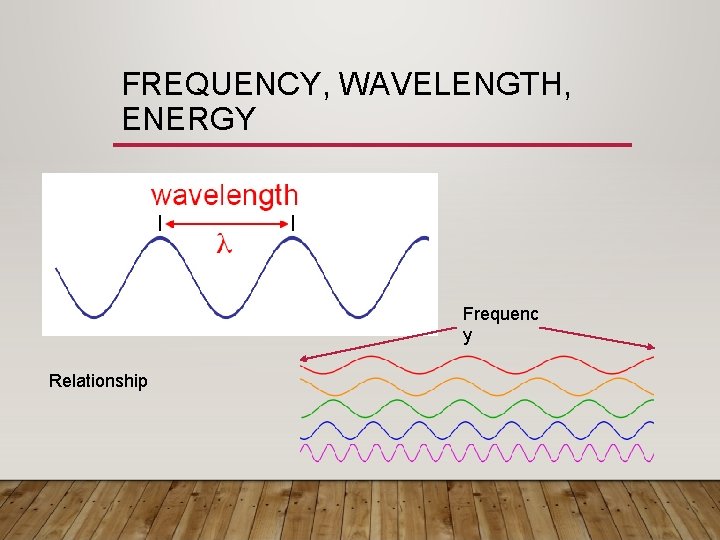
FREQUENCY, WAVELENGTH, ENERGY Frequenc y Relationship

HOW TO CONVERT BETWEEN THE TWO • C= λ · ν • c= speed of light (3. 00 x 108 m/s) • λ = wavelength (m) • ν = frequency (Hertz: Hz) • 1. 0 nm = 1 x 10 -9 m C • Let’s Practice! λ ν

WHY IS THIS IMPORTANT • Allows us to calculate the amount of energy released. • h= Planck’s Constant (6. 63 x 10 -34 J · s)

• REVIEW OF EQ: How can the properties of waves be used in energy calculations? • Without looking, put the following in order from longest wavelength, to shortest wavelength. • X-ray • Infrared Microwave Visible Radio Wave Gamma • Circle the one with the highest frequency? • Put a smiley face over the one with the most amount
- Slides: 21