The biotech market today and the capacity crunch






















契機 The biotech market today and the ”capacity crunch” n Why outsource and factors in choosing a CMO n Sound process development n Increasing regulatory requirements n 22

Capacity crunch “We estimate that demand for manufacturing capacity will exceed current capacity by a factor of four by 2005” J. P. Morgan. The State of Biologics Manufacturing, 2001 n ”. . . the expected needs for increased capacity would be almost fourfold for fermentation and monoclonal antibodies and ten fold for mammalian cell culture. Given that Contract biomanufacturers only have an increase of one to two fold planned, this shortfall in capacity could become significantly painful” n High Tech Business Decisions, 2001 23
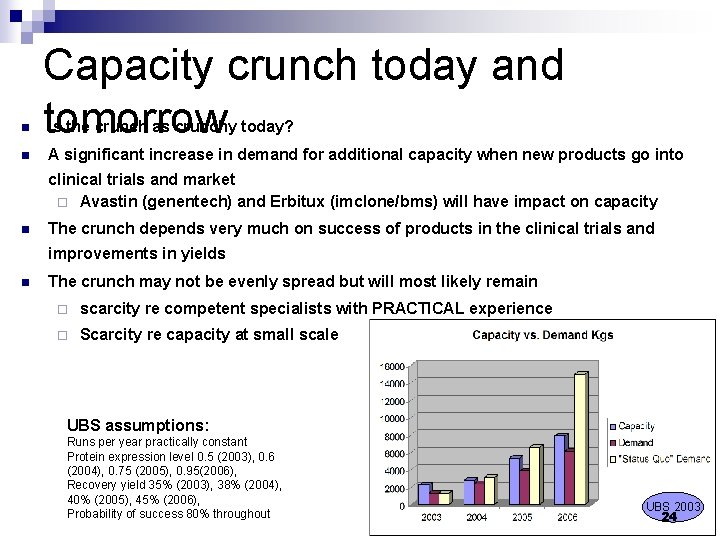
n n Capacity crunch today and tomorrow Is the crunch as crunchy today? A significant increase in demand for additional capacity when new products go into clinical trials and market ¨ Avastin (genentech) and Erbitux (imclone/bms) will have impact on capacity n The crunch depends very much on success of products in the clinical trials and improvements in yields n The crunch may not be evenly spread but will most likely remain ¨ scarcity re competent specialists with PRACTICAL experience ¨ Scarcity re capacity at small scale UBS assumptions: Runs per year practically constant Protein expression level 0. 5 (2003), 0. 6 (2004), 0. 75 (2005), 0. 95(2006), Recovery yield 35% (2003), 38% (2004), 40% (2005), 45% (2006), Probability of success 80% throughout UBS 2003 24

Solutions for meeting capacity demands n Long term ¨ Expanding capacity or building new facilities n Short term ¨ Improving process development ¨ Increasing staff and adding shifts ¨ Develop better expression systems, optimize media selection, simplify purification, improve processes and technologies 25

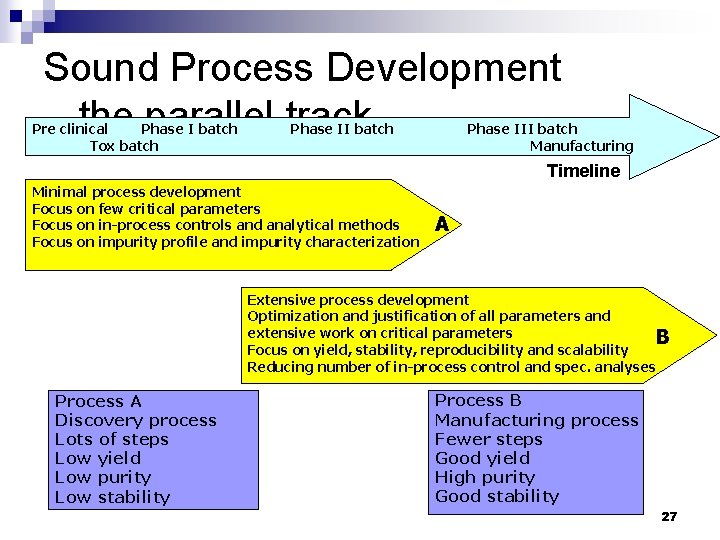
Sound Process Development - the parallel track Pre clinical Phase I batch Tox batch Phase III batch Manufacturing Timeline Minimal process development Focus on few critical parameters Focus on in-process controls and analytical methods Focus on impurity profile and impurity characterization A Extensive process development Optimization and justification of all parameters and extensive work on critical parameters B Focus on yield, stability, reproducibility and scalability Reducing number of in-process control and spec. analyses Process A Discovery process Lots of steps Low yield Low purity Low stability Process B Manufacturing process Fewer steps Good yield High purity Good stability 27

”To build or not to build” n n n - Investment of 200 -500 million € for new commercial facility 3 -6 years completion from design to ”on-stream” In the end time and money is more than budgeted Need to employ >many competent employees as biomanufacturing is complex Easier access to capacity through partnering between haves and haves-not Agonist (small quantities) vs. antagonists (large quantities) considerations Timeline: Too early? What if candidate fails in CTs Too late? Lack of revenue stream 28

THINK PRODUCTION SCALE FROM EARLY DEVELOPMENT: Consider environmental, safety and economical issues Use well established techniques – easy to explain to authorities (and the buyer of the process) Design for low bio burden (Selection of buffers and CIP) Availability of raw material in large quantities Include production of stabile intermediate product Cleanability of equipment The validation task in production scale 29

n n Contract Manufacturing Organization (CMO) Provider of mammalian and microbial based contract services including development and manufacture ¨ ¨ ¨ n Cloning & transfection Fermentation/Cultivation Recovery Purification and API formulation Analytical development and QC Quality Assurance and Regulatory Support State-of-the-art new facility designed and constructed from green-field for flexible CMO services 30

Choosing a CMO • Focus on • • • Time, money and competence Drawback • Loss of control of the manufacturing process, is the quality level OK? , IP exclusivity • Dependent on the CMO for your success – better choose wisely (quality, science, ease of scope changes, avoidance of mouse-trapping). Benefits • Lack of in-house capacity • Avoid capital risk to build facility – postpone investment • Reduction of overhead costs, economy of scale • Access to CMO’s capacity, expertise (toolbox), experience and flexibility – saving internal resources for other activites • CMO typically masters many different technologies cost-efficiently • Complex biotech processes – avoid the learning curve and how to deal with regulatory bodies • Established companies: • Leveraging bottlenecks in capacity for development and manufacturing and acces to new technologies • Newly established biotech: • Lack of technology, CGMP and regulatory competencies and/or financial muscle for 31 financing own factory


CMO與CRO的發展 33













CMO? or CRO? 46
- Slides: 46