The Biotech Industry Segmentation Scale and Regulatory Environment

The Biotech Industry: Segmentation, Scale and Regulatory Environment Mark A. Snyder, Ph. D. Bio-Rad Laboratories 1 of 39

My background • • B. S. in biochemistry (MIT) Ph. D. in biochemistry (UCB)—didn’t know what else to do, seemed like the logical thing Post-doctoral studies (UCSF)—what everyone else did Five years at Scios as Senior Scientist (molecular biology, b. FGF purification) Four years at XOMA (Manager, Process Development) Thirteen years at Bayer (Associate Director, Purification Process Development) Now at Bio-Rad (Manager, Process Applications R&D) 2 of 39

Industry Segmentation 3 of 39

Biotechnology market segments • Agricultural Biotech • Biofuels • Bioinformatics • Biomaterials • Biomedical Biotech • Bioprocess human diagnostics genomics proteomics therapeutics Antibiotics Antibodies Antisense Drug delivery Drug Development Drug Disccovery Gene/cell Therapy Phytotherapeutics Therapeutic Enzymes Small Molecule Drugs Engineering • Biotech Investment • Biotech Services • Biotech Supplier • Environmental Vaccines Biotech • Marine Biotech • Microarrays & Microfluidics • Nanobiotechnology • Tissue Engineering • Veterinary Biotech http: //www. argosbiotech. de/1000/gbcd_markets. htm 4 of 39

Where are the jobs? • A pharmaceutical company developing new ways to cure cancer • A chemical company making plastic from corn instead of petroleum • An environmental company finding new microorganisms to clean up oil spills • An agricultural company developing drought-resistant crops • An energy company using fermentation to make ethanol for fuel • Law enforcement using DNA analysis and other biotechnology tools to solve crimes. • Multi-national collaborations storing DNA samples of endangered species. • The Do. D making vaccines as a new defense against bioterrorism. 5 of 39

Fight, feed, save I Fighting disease: • Faster Diagnosis : Biotechnology has made it possible to diagnose strep throat in minutes, rather than days. • New Treatments : Biotechnology has delivered the first treatments for a variety of diseases (Gaucher’s, Fabry’s, etc. ). In the future, defective genes or damaged cells may be repaired or replaced. • Better Prevention: New vaccines help prevent hepatitis, meningitis, and influenza. New vaccines in food may eliminate the need for a trip to the doctor and a shot. 6 of 39

Fight, feed, save II Feeding the world: • Hardier Crops : Innovative biotechnology solutions are creating crops that are more resistant to insects, diseases, and harsh weather. • Healthier Animals : Biotechnology-engineered vaccines are available for parasites and infectious diseases. Even rare chicken might be possible. • Better Food: One of the first biotechnology foods was a tomato that could ripen on the vine for better flavor and still remain firm for shipping. Biotechnology can make food safer by reducing naturally-occurring toxins and allergens. 7 of 39

Fight, feed, save III Saving our environment: • New Fuels : New “designer” enzymes are being used to manufacture bioethanol from renewable resources such as corn or agricultural waste. • Cleaner air , water , and soil : Plants and bacteria can be used to safely clean up oil spills and remove toxic chemicals from the environment. • New Materials : Genetically-engineered cells use plant sugars instead of petrochemicals to create biodegradable plastics and polyesters. “Green plastics” from corn are used to manufacture packaging materials, clothing, and bedding. • Reduce Global Warming: Genetically-modified bacteria can turn CO 2 from the air into usable plastics and fuels. 8 of 39

A few of the disciplines involved • Agricultural Science • Chemistry • Microbiology • Epidemiology • Molecular Biology • Food Science • Pharmacology • Biochemistry • Bioinformatics • Functional Genomics • Physiology • Biostatistics • Genetics • Plant Pathology • Botany • Immunology • Toxicology • Cell Biology • Marine Biology • Virology • Zoology 9 of 39
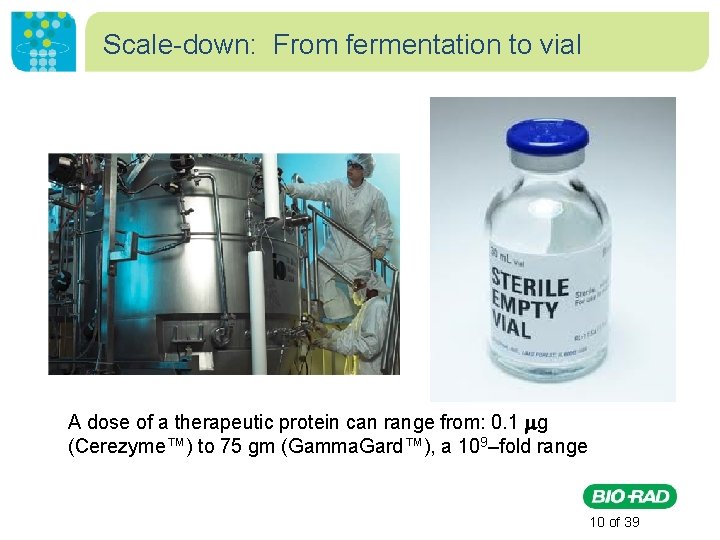
Scale-down: From fermentation to vial A dose of a therapeutic protein can range from: 0. 1 g (Cerezyme™) to 75 gm (Gamma. Gard™), a 109–fold range 10 of 39

Column scale-up: 1, 000 -fold 200 L; $30 200+ L; $2 MM. 11 of 39

Scale-up: from lab bench to production floor • • Ultimate scale-up can be 106 -fold Small-scale process *must* mirror the large-scale – Needed for viral clearance studies – Needed for enabling full-scale validation studies – Needed for failure analysis Some of what should not change: – Buffer setpoints and tolerances (p. H, conductivity ranges) – Residence time during chromatography – Bed heights (more or less) – Quality inputs – Quality outputs Some of what will change: – Volumes – Times – People – Equipment 12 of 39

Working In A Regulated Industry 13 of 39

Section overview 1. 2. 3. 4. 5. 6. 7. 8. Biological products are not like aspirin Making biologics is not like making aspirin Why regulations are the way they are The biologic development process, and therefore the selling cycle, is long and perilous The license application: control addresses complexity Our annuity: changes after licensing aren’t easy Supply chain management is critical How we help our customers through their development process 14 of 39

CBER vs. CDER (as of June, 2003) • CDER (Center for Drug Evaluation and Research) – Regulates under FD&C Act • Monoclonal antibodies for in vivo use • Proteins (e. g. monoclonal antibodies) intended for therapeutic use, except for those that are specifically assigned to CBER (e. g. , vaccines and blood products) • Immunomodulators (non-vaccine and non-allergenic products intended to treat disease by inhibiting or modifying a pre-existing immune response) • CBER ((Center for Biologics Evaluation and Research – Regulates under PHS Act • Cellular products, including products composed of human, bacterial or animal cells. • Gene therapy products • Vaccines • Allergenic extracts used for the diagnosis and treatment of allergic diseases and allergen patch tests. • Antitoxins, antivenins, and venoms • Blood, blood components, plasma derived products, human or animal polyclonal antibody preparations http: //www. fda. gov/About. FDA/Centers. Offices/Officeof. Medical. Productsand. Tobacco/CBER/ucm 133463. htm 15 of 39

1. Biological products are not like aspirin • “In contrast to chemically synthesized small molecular weight drugs, which have a well-defined structure and can be thoroughly characterized, biological products are … complex in structure, and thus are usually not fully characterized. ” – Aspirin: one unique molecule – m. Ab: potentially >2 x 106 forms • >2 x 106 different potencies, safeties, efficacies and stabilities • Residual impurities can + or - affect drug properties • “What you can test for is a surrogate for what you cannot”—P. Burd http: //www. fda. gov/Drugs/Development. Approval. Process/%20 How. Drugsare. Developedand. Approved/Approval. Applications/Therapeutic. Biologic. Applications/ucm 113522. htm 16 of 39

Monoclonal does not mean identical • http: //www. lonza. com/custom-manufacturing/development-technologies/protein-and-vaccine-development-services/developabilityservices/manufacturability-assessment. aspx 17 of 39

2. Making biologics is not like making aspirin • “Because, in many cases, there is limited ability to identify the identity of the clinically active component(s) of a complex biological product, such products are often defined by their manufacturing processes. ” • Manufacturing consistency and QC testing are designed to ensure consistent safety and efficacy http: //www. fda. gov/Drugs/Development. Approval. Process/%20 How. Drugsare. Developedand. Approved/Approval. Applications/Therapeutic. Biologic. Applications/ucm 113522. htm 18 of 39

Making resins is not like making a widget • On the microscopic level, each resin bead is different – Degree of crosslinking – Ligand density – Ligand distribution – Number/diameter/length/tortuosity of pores – Surface roughness • Macroscopically, each lot of resin is different • Like biologics, our process is the product • Manufacturing consistency and QC testing are designed to ensure consistent performance 19 of 39

3. Why regulations are the way they are: 1820 - 1906 • 1820’s: British pharmacist Jacob Bell notes that drugs “reduced by decay or ingenuity” were still "good enough for America. “ • 1848: Drug Importation Act prohibited importation of unsafe or adulterated drugs • 1879: Peter Collier, chief chemist of the Department of Agriculture recommended passage of a national food and drug law. Such a bill was introduced during his tenure. It was defeated. • 1902: Biologics Control Act followed the death of 20 children from tetanus-contaminated diphtheria anti-toxin and smallpox vaccine (“Horse named Jim” disaster). Manufacturing sites had to be inspected and licensed. • 1906: Pure Food and Drug Act passed which forbade use of opiates, toxic preservatives (borax, formaldehyde) and high levels of alcohol in medicines https: //archive. org/details/61541320 R. nlm. nih. gov http: //www. fda. gov/About. FDA/What. We. Do/History/FOrgs. History/CDER/Centerfor. Drug. Evaluationand. Research. Brochureand. Chronology/ucm 114470. htm http: //samples. sainsburysebooks. co. uk/9781400860326_sample_640345. pdf 20 of 39

21 of 39

1938 • In 1937 the “Elixir Sulfanilamide” disaster, where the vehicle for a liquid formulation was ethylene glycol, killed more than 100 people in 15 states – “… Any doctor who has practiced more than a quarter of a century has seen his share of death. But to realize that six human beings, all of them my patients, one of them my best friend, are dead because they took medicine that I prescribed for them …well, that realization has given me such days and nights of mental and spiritual agony as I did not believe a human being could undergo and survive. I have known hours when death for me would be a welcome relief from this agony. " (Letter by Dr. A. S. Calhoun to FDR, October 22, 1937) • Food Drug and Cosmetics Act passed in 1938 (21 U. S. C. 301) – Oversees the safety of food, drugs, and cosmetics – Regulates “articles intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in man or other animals; and … articles (other than food) intended to affect the structure or any function of the body of man or other animals. ” – Amended many times – Gives authority for establishing c. GMP’s – Drugs must meet requirements for safety, identity, strength, quality and purity (§ 351) – Provides for seizures, fines, imprisonment, etc. 22 of 39

1962 • European thalidomide use leads to the death of approximately 2, 000 children and serious birth defects in more than 10, 000 • Entry into the U. S. singlehandedly prevented by Frances O. Kelsey (age 18) at the FDA – “I had the feeling throughout the day, that they were at no time being wholly frank with me … regarding this drug. ” • Dr. Frances O. Kelsey, remembering her meetings with William S. Merrell Drug Company – Hailed as a heroine by U. S. newspapers • FD&C Act amended to require clinical trials (Kefauver Harris Amendment) 23 of 39

Public Health Services Act • 1798: “Act for Relief of Sick and Disabled Seamen” is passed. • 1878: Fears of epidemics in the U. S. lead to passage of the National Quarantine Act. • 1944: PHSA passed. Essential parts for this discussion codified at 42 U. S. Code § 262. • Covers “any biological product” where: – “The term “biological product” means a virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or derivative, allergenic product, protein (except any chemically synthesized polypeptide… applicable to the prevention, treatment, or cure of a disease or condition of human beings. ” https: //www. law. cornell. edu/uscode/text/42/262 24 of 39

4. Regulations + biological complexity = long, perilous road to market and therefore the selling cycle • It may take a year before an initial resin sample is put into a lab-scale process. Failure means our resin didn’t work as well as someone else’s. • It may be another 3 years before meaningful quantities are ordered as the product enters pre-clinical studies. • Clinical trial orders can be large but are usually once per phase. Failure usually due to clinical outcomes, not resin performance. { 25 of 39
Biological drug development is expensive • The average cost of bringing a new drug to market is $5 billion—the highest cost is failure. • Astra. Zeneca spends $12 billion in research money for every new drug approved • Competition is fierce, especially now that biosimilars are beginning to be approved – Zarxio (Neupogen biosimilar) licensed 3/6/15 http: //www. advisory. com/daily-briefing/2013/08/15/forbes-it-can-cost-more-than-5 -billion-to-bring-a-drug-to-market 26 of 39

Selling resin for biopharmaceutical manufacturing selling laboratory products for life science research • • Long selling cycle (years vs. days/weeks) Exceedingly risk-averse customer Multiple customer stakeholders High public and legal scrutiny of drug safety and supply • Enormous consumer economic commitment (2¢/dose for aspirin; ~$2000/dose for Humira®) • Extraordinary level of regulation • Assured annuity sales (almost) 27 of 39
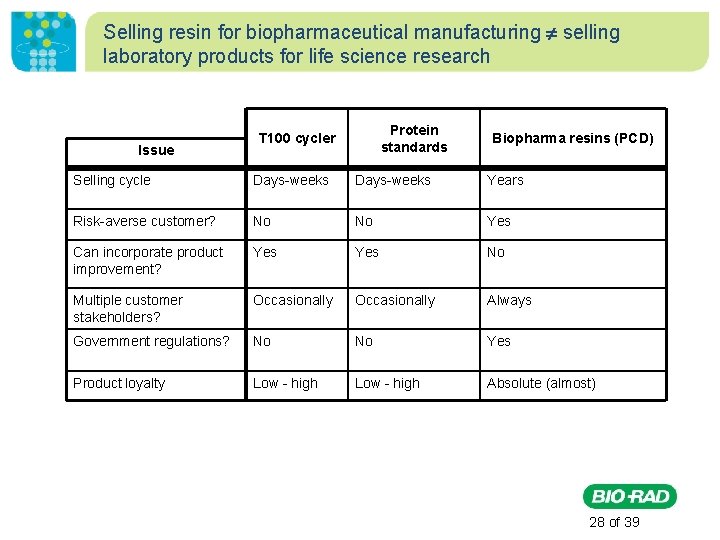
Selling resin for biopharmaceutical manufacturing selling laboratory products for life science research Issue Protein standards T 100 cycler Biopharma resins (PCD) Selling cycle Days-weeks Years Risk-averse customer? No No Yes Can incorporate product improvement? Yes No Multiple customer stakeholders? Occasionally Always Government regulations? No No Yes Product loyalty Low - high Absolute (almost) 28 of 39

5. The license application: control addresses complexity • Typically 100, 000 pages long (about 35 linear shelf feet) • • Legally binding contract License application says, among other things: – Here is exactly how we (the drug company) made 3 -5 lots of this biologic product, at full scale – These lots have clinical safety and efficacy data – We’re pretty confident that all lots made this way will be equivalent – We will keep to narrow specification ranges to ensure future product safety/quality/potency/identity/efficacy, including: • • • What resin we will use Size of the packed bed Number of cycles the resin will be used Length of time a batch resin will be in service Amount of product put over the resin per batch of biologic drug Allows the manufacturer to place a product into interstate commerce (21 CFR 601. 2) http: //www. fiercebiotech. com/topics/fda_approval_process. asp 29 of 39

6. Bio-Rad’s annuity: changes after licensing aren’t easy • Changes to a purification process or equipment are known to have caused detrimental effects – Different host cell protein profile; isoform profile; product stability; etc. • Thus, making a change without compromising safety and efficacy can be a long, costly process – Once a particular resin is in a licensed process, it’s there to stay. PTC for Changes to an Approved Application http: //www. fda. gov/downloads/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/UCM 124805. pdf 30 of 39

But anything, ultimately, is possible • Anything can be changed in drug manufacturing provided that new clinical trials are performed • Sufficiently well-characterized biologics may not require a clinical trial, only a comparability study • In some cases, Quality by Design (Qb. D) gives a more flexible approach to change http: //www. fda. gov/Drugs/Development. Approval. Process/%20 How. Drugsare. Developedand. Approved/Approval. Applications/Therapeutic. Biologic. Applications/ucm 113522. htm http: //www. fda. gov/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/ucm 122879. htm 31 of 39

7. Supply chain management is critical • During initial lab-scale resin screening – Few customers will commit to even try a resin before it’s launched – But no customer can be sure of resin use until drug product is licensed and launched – Delivery time not particularly critical • During clinical trials – Likely to have only one order for each phase, separated by years – No guarantee that a drug will succeed at each stage – Delivery delays of a few weeks might be tolerated • After licensing – – • FDA does not look favorably upon drug shortages (21 U. S. C. 356 c(a)(1) ) Biopharma manufacturing schedules are often tight Ordering is sporadic; an “order” often lasts for years. On-time delivery is critical Severe quality/supply issues could force a resin change – Regardless, delays harm Bio-Rad’s reputation • Most customers expect delivery in 6 - 8 weeks, 12 at the outside 32 of 39

BR supply chain: constrained by regulations • Can you use the current resin until we have an open slot in our Richmond plant? No—we only validated 100 cycles. • Can you use someone else’s resin for awhile? No—we only have clinical data with yours. • Can you make do with 80% of the amount you ordered? No—we validated a particular column bed size and product loading level. • Can we change the part number? Yes, but we’ll have to change a lot of documents. 33 of 39

Supply chain and R&D meet at the customer • Before a lab-scale sample is given: – – – • Is this a launched product? Can I sample three independent manufacturing batches to test? How many liters can you make at one time? Has anyone else used this in a clinical trial or licensed product? Do you have a regulatory support file (RSF)? After the above questions are answered: – Why might this product be better than your competitors for my application? • During process development or clinical trials: – It’s not working the way I ideally need it to. Can you help? • After licensing: – My recent biologic drug lots are different. Is your resin to blame? Have there been any changes to your raw materials or your resin manufacturing/QC processes? 34 of 39

Pharma support isn’t like other divisions • We have no choice but to *always* go the extra mile – Relatively small customer base – Use of our resins in licensed products 35 of 39

Must have at product launch and beyond • • Full technical support during all phases of customer usage—initial application, process development, scale-up, post-launch Fully-validated, auditable manufacturing process at final commercial scale (21 CFR 820. 50) • Assurance that our process will not change over time – Is our equipment / manufacturing process stable? – Is our raw material supply stable? • • • Do we have a second source for raw materials? Robust change control process if we must alter manufacturing Ideally three, but at least one commercial-scale and two pilot-scale lots of material available for testing Regulatory support file (RSF) Full commercial supply availability through entire customer product lifetime 36 of 39

Our products are sold to multiple stakeholders • The BR PCD product is not just a catalog number – resin – support file – manufacturing SOP’s – customer operations – applications support – Interactions with multiple customer stakeholders: PD, QA, QC, RA, mfg. , supply chain, engineering • Our products will not sell if any of these are missing 37 of 39

8. How PCD R&D Applications Group helps our customers through their NPD process • As scientists and (in some cases) former pharma employees, we bring voice of customer and customer contacts to BR – • We develop data giving customers a reason to try our product within a crowded landscape – – • Application notes, technical notes, product information sheets, instruction manuals Posters and presentations at conferences We supply full technical support (site visits, teleconferences, etc. ) during all phases of customer usage—initial application, process development, scale-up, post-launch – – – • We also know the limitations of our customers Targeted presentations at customer sites Training for BR sales staff SME letters We act as a bridge between customer issues and our resin manufacturing facilities – – – XXX CHT “binding capacity” issue XXX Macro-Prep packing Melamine questions XXX study on UNOsphere packing Specification changes for XXX alder seed contamination 38 of 39

Thank you! Questions? 39 of 39

Supplementary slides 40 of 39

Example of an approval letter 41 of 39

One example of FDA enforcement action • • • The U. S. Food and Drug Administration announced today the filing of a consent decree against Medtronic, Inc. , and two of the company’s officers—S. Omar Ishrak and Thomas M. Tefft —for repeatedly failing to correct violations…The consent decree cites violations of the quality system regulation…The legal action requires the company to stop manufacturing, designing and distributing new Synchromed II Implantable Infusion Pump Systems. The consent decree also requires Medtronic to retain a third-party expert to help develop and submit plans to the FDA to correct violations. The consent decree will remain in effect until the FDA has determined that Medtronic has met all the provisions listed in the consent decree. Once Medtronic receives permission from the FDA to resume the design, manufacture and distribution of these products, the company must continue to submit audit reports so the agency can verify the company’s compliance. In addition to these audits, the FDA will monitor the company’s activities through its own inspections. http: //www. fda. gov/News. Events/Newsroom/Press. Announcements/ucm 444690. htm? source=govdelivery&utm_medium=email&utm_source=govdelivery 42 of 39

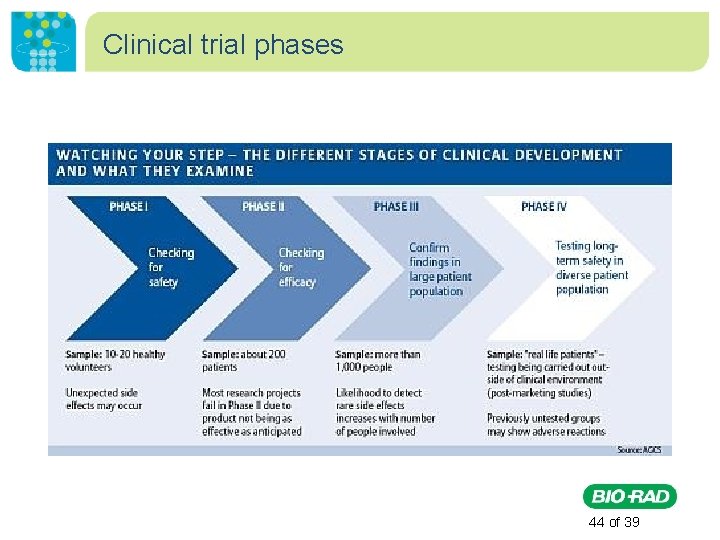
Clinical trial phases • Phase I: Evaluate safety, determine a safe dosage range, and identify side effects. • Phase II: The drug or treatment is given to a larger group of people to see if it is effective and to further evaluate its safety. • Phase III: The drug or treatment is given to large groups of people to confirm its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow the drug or treatment to be used safely. • Phase IV: Studies are done after the drug or treatment has been marketed to gather information on the drug's effect in various populations and any side effects associated with long-term use. http: //www. nlm. nih. gov/services/ctphases. html 43 of 39
Clinical trial phases 44 of 39
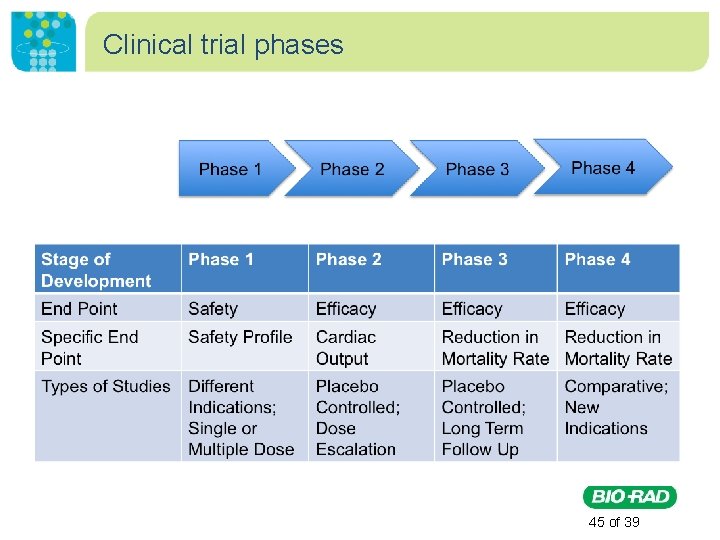
Clinical trial phases 45 of 39
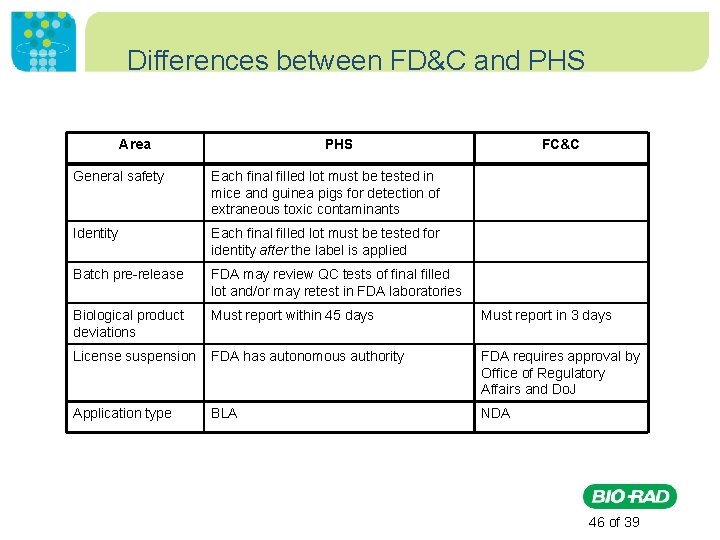
Differences between FD&C and PHS Area PHS FC&C General safety Each final filled lot must be tested in mice and guinea pigs for detection of extraneous toxic contaminants Identity Each final filled lot must be tested for identity after the label is applied Batch pre-release FDA may review QC tests of final filled lot and/or may retest in FDA laboratories Biological product deviations Must report within 45 days Must report in 3 days License suspension FDA has autonomous authority FDA requires approval by Office of Regulatory Affairs and Do. J Application type BLA NDA 46 of 39

Biological products are not like aspirin • “Following initial laboratory and animal testing that show that investigational use in humans is reasonably safe, biological products can be studied in clinical trials in humans. If the data generated by the studies demonstrate that the product is safe and effective for its intended use, the data are submitted as part of a marketing application. ” http: //www. fda. gov/Drugs/Development. Approval. Process/%20 How. Drugsare. Developedand. Approved/Approval. Applications/Therapeutic. Biologic. Applications/ucm 113522. htm 47 of 39

Biological products are not like aspirin • “Issuance of a biologics license is a determination that the product, the manufacturing process, and the manufacturing facilities meet applicable requirements to ensure the continued safety, purity and potency of the product. ” • Regulations regarding therapeutic biological products are codified in 21 CFR parts 210, 211, 225, 226 and 600 – 680, 810. http: //www. fda. gov/Drugs/Development. Approval. Process/%20 How. Drugsare. Developedand. Approved/Approval. Applications/Therapeutic. Biologic. Applications/ucm 113522. htm 48 of 39

Requirements for change • • Sec. 610. 9 Equivalent methods and processes. Modification of any particular test method or manufacturing process or the conditions under which it is conducted as required in this part or in the additional standards for specific biological products in parts 620 through 680 of this chapter shall be permitted only under the following conditions: (a) The applicant presents evidence, in the form of a license application, or a supplement to the application submitted in accordance with 601. 12(b) or (c), demonstrating that the modification will provide assurances of the safety, purity, potency, and effectiveness of the biological product equal to or greater than the assurances provided by the method or process specified in the general standards or additional standards for the biological product; and (b) Approval of the modification is received in writing from the Director, Center for Biologics Evaluation and Research or the Director, Center for Drug Evaluation and Research. 49 of 39

Why Bio-Rad is audited • Sec. 820. 50 Purchasing controls. Each manufacturer shall establish and maintain procedures to ensure that all purchased or otherwise received product and services conform to specified requirements. 50 of 39

What happens if customers don’t audit • http: //www. fda. gov/ICECI/Enforcement. Actions/Warning. Letters/ucm 240617. htm • 4. Failure to follow procedures for evaluating suppliers of critical materials. • For example, your firm failed to audit your suppliers at least once every two years as required by your SOP…Our inspection identified the following facilities that have not been audited within the past two years: • • (b)(4) - Last audit 06/18/2008 (over 25 months prior to the initiation of the FDA inspection) 51 of 39

Changes may be expensive and time-consuming • Please note that several changes you plan to make may result in significant changes to in-process parameters and further downstream manufacturing steps. Your process validation could be affected by these changes and should be evaluated for the need to re-validate based on these changes and modifications. These changes should also be communicated to the license holder so that an evaluation may be made for the need to submit a supplement to the Cinryze BLA. • http: //www. fda. gov/ICECI/Enforcement. Actions/Warning. Letters/2013/ucm 368313. htm 52 of 39

Just because you say so doesn’t make it true • http: //www. fda. gov/ICECI/Enforcement. Actions/Warning. Letters/2011/ucm 291672. htm • In addition, you stated in your response that none of the changes made should have an adverse effect on the approved and licensed (b)(4) API. However, you provided no scientific rationale for this conclusion. In response to this letter, please provide your revised procedure and your scientific rationale to conclude that the changes did not impact drug quality. 53 of 39

Shortages are a very big deal • http: //www. fda. gov/ICECI/Enforcement. Actions/Warning. Letters/2011/ucm 291672. htm • The lack of compliance with CGMPs may lead to production problems and adversely impact the availability of critical medically necessary products that … may lead to … unavailability for those patients who rely on such treatments. To help ensure an ongoing supply, we request that you notify FDA as soon as possible, if you are considering any changes to your production plans that may adversely impact supply. We also request that you develop a proactive plan to ensure a continued supply in the event there is a significant production delay. 54 of 39

From a 2000 Warning Letter 55 of 39

An earlier warning letter 56 of 39

What a license looks like 57 of 39

Another license 58 of 39
- Slides: 58