The BIG question Why are balanced equations useful

The BIG question Why are balanced equations useful What are we learning: Use a balanced chemical equation to calculate the mass of a reactant or product Work out the mass of a given chemical Describe how the mass is conserved ?

The BIG question Why are balanced equations useful REACTING MASS CALCULATIONS 1. Circle the 2 important substances in the equation. 2. Rewrite those 2 substances on your paper. 3. Work out the mass of each of the substances. 4. Scale up or scale down. See next slide for worked example ?

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 1) Step 1 What mass of magnesium oxide will be produced from 12 g of magnesium? 2 Mg + O 2 2 Mg. O So the balanced equation tells me that: 2 moles of magnesium (Mg) will produce 2 moles of magnesium oxide (Mg. O) Step 2 Step 3 Convert to masses 2 Mg. O 48 g 80 g So if I had 48 g Mg I would theoretically make 80 g Mg. O BUT the question says I only have 12 g magnesium so……… To get from 48 to 12 Step 4 divide by 4 2 Mg. O 48 g 80 g 12 g 20 g Therefore you must divide 80 by 4 too!!

The BIG question Why are balanced equations useful REACTING MASS CALCULATIONS See next slide for worked example ?

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 1) What mass of magnesium oxide will be produced from 12 g of magnesium? ? 2 Mg + O 2 2 Mg. O Moles Mg = mass Mr = 12 24 = 0. 5 Moles Mg. O = 1: 1 ratio therefore 0. 5 Mass Mg. O = Mr x moles = 40 x 0. 5 = 20 g

The BIG question Why are balanced equations useful • Hopefully you can see that you get the same answer no matter which method you use. • Have a go at the calculations on the following slides. • You can use either method as long as you show ALL of your working. • Use whichever makes sense to you! • The slides are colour coded: Pink/red = proportion method. Green = formula method. ?
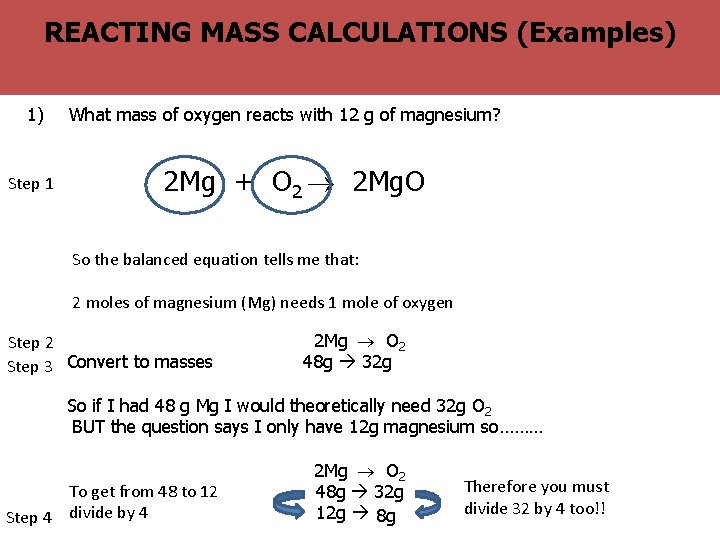
? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 1) Step 1 What mass of oxygen reacts with 12 g of magnesium? 2 Mg + O 2 2 Mg. O So the balanced equation tells me that: 2 moles of magnesium (Mg) needs 1 mole of oxygen Step 2 Step 3 Convert to masses 2 Mg O 2 48 g 32 g So if I had 48 g Mg I would theoretically need 32 g O 2 BUT the question says I only have 12 g magnesium so……… To get from 48 to 12 Step 4 divide by 4 2 Mg O 2 48 g 32 g 12 g 8 g Therefore you must divide 32 by 4 too!!
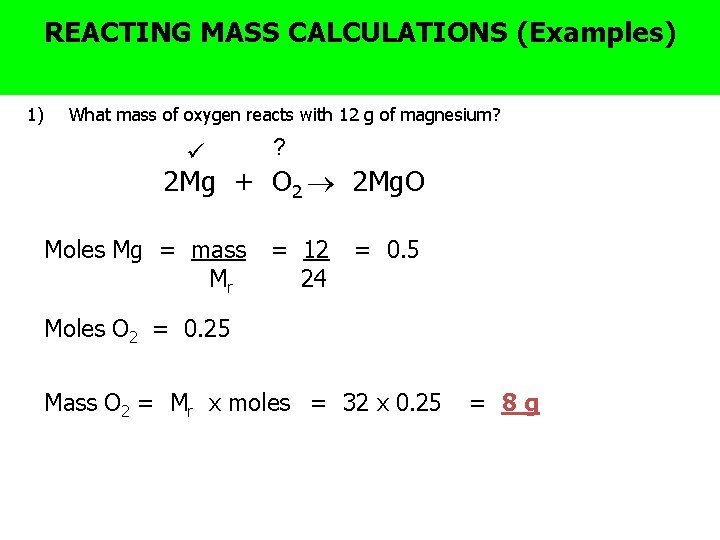
? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 1) What mass of oxygen reacts with 12 g of magnesium? ? 2 Mg + O 2 2 Mg. O Moles Mg = mass Mr = 12 24 = 0. 5 Moles O 2 = 0. 25 Mass O 2 = Mr x moles = 32 x 0. 25 = 8 g

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 2) Step 1 What mass of calcium hydroxide is made from 14 kg of calcium oxide? Ca. O + H 2 O Ca(OH)2 So the balanced equation tells me that: 1 mole of calcium oxide (Ca. O) makes 1 mole of calcium hydroxide Ca(OH)2 Step 3 Convert to masses Ca. O Ca(OH)2 56 g 74 g So if I had 56 g Ca. O I would theoretically make 74 g Ca(OH)2 BUT the question says I have 14 kg Ca. O so……… To get from 56 to 14000 Step 4 multiply by 250 Ca. O Ca(OH)2 56 g 74 g 14000 g 18500 g Therefore you must multiply 74 by 250 too!!

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 2) What mass of calcium hydroxide is made from 14 kg of calcium oxide? ? Ca. O + H 2 O Ca(OH)2 Moles Ca. O = mass Mr = 14000 56 = 250 Moles Ca(OH)2 = 250 Mass Ca(OH)2 = Mr x moles = 74 x 250 = 18500 g

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 3) Step 1 What mass of aluminium is needed to react with 640 g of iron oxide? Fe 2 O 3 + 2 Al 2 Fe + Al 2 O 3 Step 2 Fe 2 O 3 2 Al Step 3 160 g 54 g Step 4 To get from 160 to 640 multiply by 4 640 g 216 g Therefore you must multiply 54 by 4 too!!

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 3) What mass of aluminium is needed to react with 640 g of iron oxide? ? Fe 2 O 3 + 2 Al 2 Fe + Al 2 O 3 Moles Fe 2 O 3 = mass = 640 Mr 160 = 4 Moles Al = 8 Mass Al = Mr x moles = 27 x 8 = 216 g
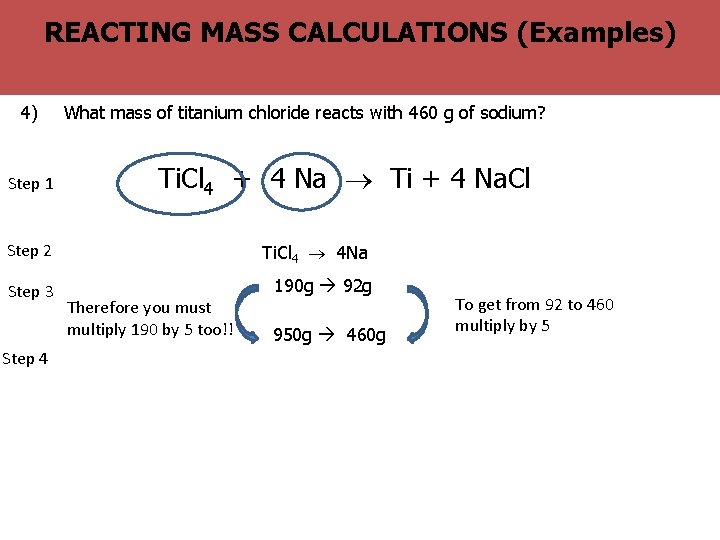
? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 4) Step 1 What mass of titanium chloride reacts with 460 g of sodium? Ti. Cl 4 + 4 Na Ti + 4 Na. Cl Step 2 Ti. Cl 4 4 Na Step 3 190 g 92 g Step 4 Therefore you must multiply 190 by 5 too!! 950 g 460 g To get from 92 to 460 multiply by 5

? The BIG Why MASS are balanced equations useful (Examples) REACTING CALCULATIONS question 4) What mass of titanium chloride reacts with 460 g of sodium? ? Ti. Cl 4 + 4 Na Ti + 4 Na. Cl Moles Na = mass Mr = 460 23 = 20 Moles Ti. Cl 4 = 5 Mass Ti. Cl 4 = Mr x moles = 190 x 5 = 950 g
- Slides: 14