The BIG question How to complete a titration
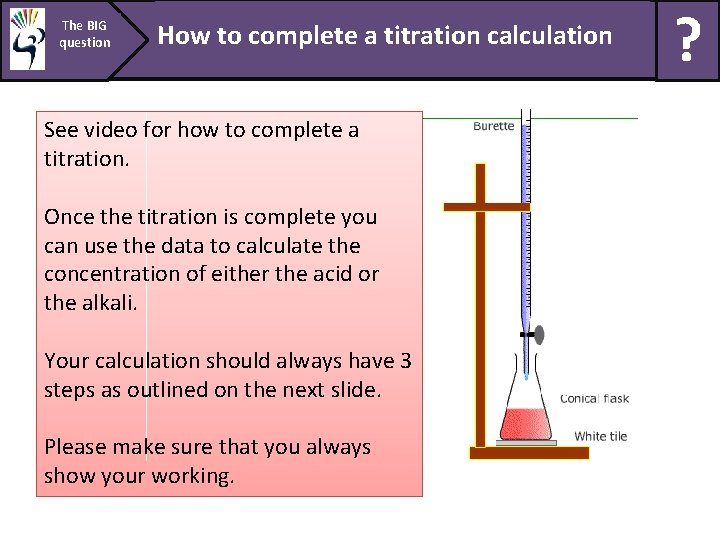
The BIG question How to complete a titration calculation See video for how to complete a titration. Once the titration is complete you can use the data to calculate the concentration of either the acid or the alkali. Your calculation should always have 3 steps as outlined on the next slide. Please make sure that you always show your working. ?
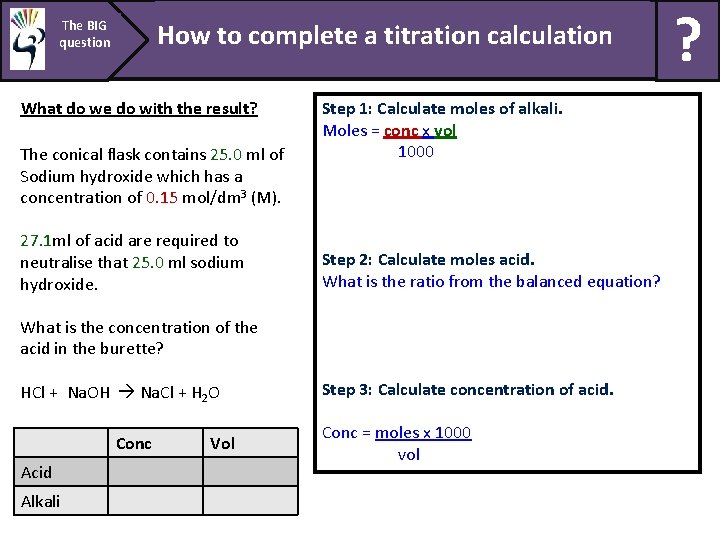
The BIG question How to complete a titration calculation What do we do with the result? The conical flask contains 25. 0 ml of Sodium hydroxide which has a concentration of 0. 15 mol/dm 3 (M). 27. 1 ml of acid are required to neutralise that 25. 0 ml sodium hydroxide. Step 1: Calculate moles of alkali. Moles = conc x vol 1000 Step 2: Calculate moles acid. What is the ratio from the balanced equation? What is the concentration of the acid in the burette? HCl + Na. OH Na. Cl + H 2 O Conc Acid Alkali Vol Step 3: Calculate concentration of acid. Conc = moles x 1000 vol ?

The BIG question How to complete a titration calculation What do we do with the result? The conical flask contains 25. 0 ml of Sodium hydroxide which has a concentration of 0. 15 mol/dm 3 (M). 27. 1 ml of acid are required to neutralise that 25. 0 ml sodium hydroxide. What is the concentration of the acid in the burette? HCl + Na. OH Na. Cl + H 2 O Conc Acid Alkali Vol ? 27. 1 0. 15 25. 0 Step 1: Calculate moles of alkali. Moles = conc x vol 1000 = 0. 15 x 25 = 0. 00375 mol 1000 Step 2: Calculate moles acid. What is the ratio from the balanced equation? 1: 1 ratio, so the moles of acid = moles alkali = 0. 00375 mol Put this value in the next equation. Step 3: Calculate concentration of acid. Conc = moles x 1000 vol = 0. 00375 x 1000 = 0. 138 mol/dm 3 27. 1 ?

The BIG question How to complete a titration calculation ? 1. In a titration, 20 cm 3 of 0. 15 M HCl reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? HCl + Na. OH Na. Cl + H 2 O 2. In a titration, 20. 0 cm 3 of 1. 0 M nitric acid, HNO 3, reacted with 25 cm 3 of Na. OH. What Increasing Difficulty was the concentration of the sodium hydroxide? HNO 3 + Na. OH Na. NO 3 + H 2 O 3. In a titration, 30. 0 cm 3 of 1. 0 M ethanoic acid, CH 3 COOH, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? CH 3 COOH + Na. OH CH 3 COONa + H 2 O 4. In a titration, 18. 2 cm 3 of 1. 0 M sulfuric acid, H 2 SO 4, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? ___ Na. OH + H 2 SO 4 Na 2 SO 4 + ___ H 2 O 5. In a titration, 20. 0 cm 3 of 1. 0 M phosphoric acid, H 3 PO 4, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide?

The BIG question 1. How to complete a titration calculation ? In a titration, 20 cm 3 of 0. 15 M HCl reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? HCl + Na. OH Na. Cl + H 2 O 1: 1 ratio Moles acid = (conc x vol)/1000 = (0. 15 x 20)/1000 = 0. 003 moles So 0. 003 moles alkali Conc alkali = (moles x 1000)/vol = (0. 003 x 1000)/25 = 0. 12 moldm-3 2. In a titration, 20. 0 cm 3 of 1. 0 M nitric acid, HNO 3, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? HNO 3 + Na. OH Na. NO 3 + H 2 O 1: 1 ratio Moles acid = (conc x vol)/1000 = (1 x 20)/1000 = 0. 02 moles So 0. 02 moles alkali Conc alkali = (moles x 1000)/vol = (0. 02 x 1000)/25 = 0. 8 moldm-3

The BIG question How to complete a titration calculation ? 3. In a titration, 30. 0 cm 3 of 1. 0 M ethanoic acid, CH 3 COOH, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? CH 3 COOH + Na. OH CH 3 COONa + H 2 O 1: 1 ratio Moles acid = (conc x vol)/1000 = (1 x 30)/1000 = 0. 03 moles So 0. 03 moles alkali Conc alkali = (moles x 1000)/vol = (0. 03 x 1000)/25 = 1. 2 moldm-3 4. In a titration, 18. 2 cm 3 of 1. 0 M sulfuric acid, H 2 SO 4, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O 1: 2 ratio Moles acid = (conc x vol)/1000 = (1 x 18. 2)/1000 = 0. 0182 moles So 0. 0182 x 2 = 0. 0364 moles alkali Conc alkali = (moles x 1000)/vol = (0. 0364 x 1000)/25 = 1. 456 moldm -3

The BIG question How to complete a titration calculation ? 5. In a titration, 20. 0 cm 3 of 1. 0 M phosphoric acid, H 3 PO 4, reacted with 25 cm 3 of Na. OH. What was the concentration of the sodium hydroxide? H 3 PO 4 + 3 Na. OH Na 3 PO 4 + 3 H 2 O 1: 3 ratio Moles acid = (conc x vol)/1000 = (1 x 20)/1000 = 0. 02 moles So 0. 02 x 3 = 0. 06 moles alkali Conc alkali = (moles x 1000)/vol = (0. 06 x 1000)/25 = 2. 4 moldm-3
- Slides: 7