The Basics of Multidrug Resistant TB NAME OF
![The Basics of Multi-drug Resistant TB [NAME OF PRESENTER] [TITLE OF PRESENTER] STREAM Clinical The Basics of Multi-drug Resistant TB [NAME OF PRESENTER] [TITLE OF PRESENTER] STREAM Clinical](https://slidetodoc.com/presentation_image_h2/0e0c1d7ff3bcefe13f9982c8817c3e3e/image-1.jpg)
The Basics of Multi-drug Resistant TB [NAME OF PRESENTER] [TITLE OF PRESENTER] STREAM Clinical Trial

Overview of the presentation

Outline • Burden of disease • TB treatments • Conclusions

Status of MDR TB in the world

Status of MDR/RR-TB worldwide • In 2017, there were 558, 000 new MDR/RR‐ TB cases • Of those cases, 139, 114 (~ 1 in 4) were started on treatment Source: Global Tuberculosis Report 2018
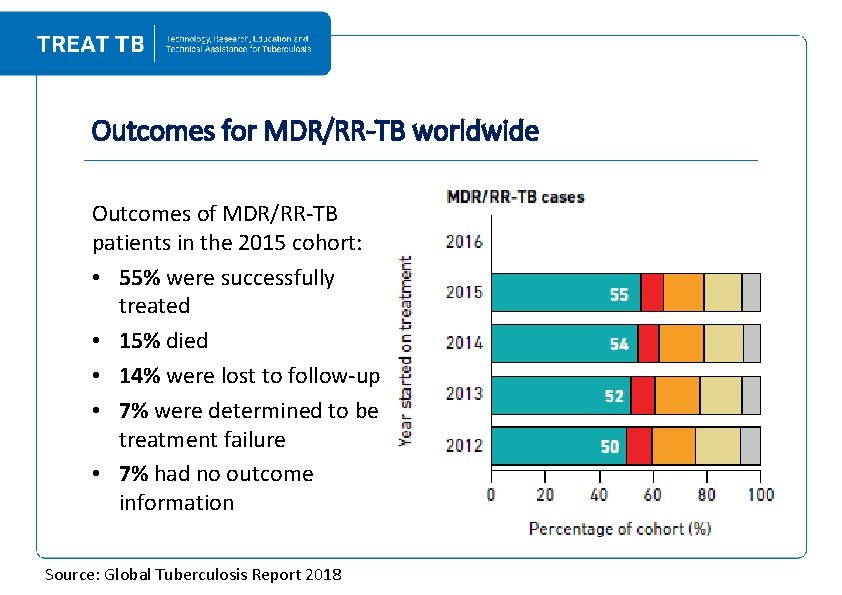
Outcomes for MDR/RR-TB worldwide Outcomes of MDR/RR‐TB patients in the 2015 cohort: • 55% were successfully treated • 15% died • 14% were lost to follow‐up • 7% were determined to be treatment failure • 7% had no outcome information Source: Global Tuberculosis Report 2018

Where does MDR-TB occur?

Treatment for TB

Treatment of drug sensitive TB • Two main drugs for treatment of DS TB • Isoniazid • Rifampicin • Treatment taken 6 months by mouth • Treatment very successful • >90% cure for patients who take their medication • If resistance to rifampicin occurs treatment • takes much longer • more side effects • does not work as well

How does resistance to TB medicines happen? Resistance is acquired when patients with drug sensitive TB can’t or don’t take medications as required • Miss doses or not given the correct doses to take • Pharmacy runs out of medicines • Suffer side effects and stop treatment Resistance is transmitted when patients are infected with resistant TB • TB organism that infects the patient is resistant to medication from the start of treatment

What if TB resistance exists? • Rifampicin is the best currently available TB medicine • If resistance to Rifampicin exists, more medications have to be given: • • not as strong worse side effects • Patients have to have injections for 6 months • Treatment less effective • Only ~55% of patients are cured on average

Earlier WHO recommendations: longer MDR-TB treatments

Longer treatments for MDR-TB Usually between 20‐ 24 months, with 2 phases Intensive phase • Quinolone (moxifloxacin) • Injections (amikacin, kanamycin or capreomycin) • PZA • Ethionamide • Terizidone/cycloserine • Ethambutol (depends on local prevalence or resistance) Continuation phase • Injections (amikacin, kanamycin or capreomycin) • PZA • Ethionamide • Terizidone/cycloserine • Ethambutol (depends on local prevalence or resistance)

Earlier WHO recommendations: Shorter MDR-TB treatments From 2016: For most patients with MDR‐TB, a shorter regimen (9‐ 11 months) can be used instead of the longer, conventional regimen But: WHO stressed the need for additional research regarding the shorter treatments (effectiveness and safety) http: //apps. who. int/iris/bitstream/handle/10665/250125/9789241549639‐eng. pdf? sequence=1
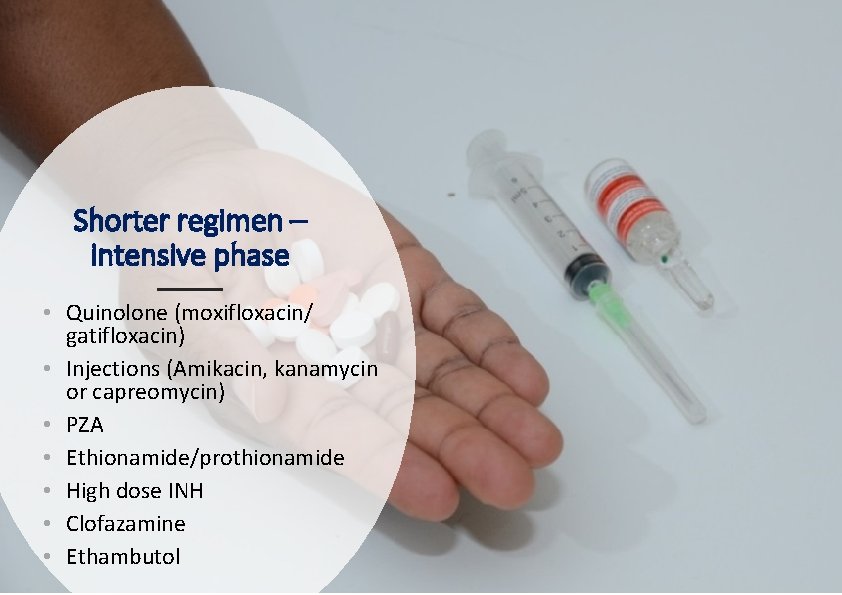
Shorter regimen – intensive phase • Quinolone (moxifloxacin/ gatifloxacin) • Injections (Amikacin, kanamycin or capreomycin) • PZA • Ethionamide/prothionamide • High dose INH • Clofazamine • Ethambutol
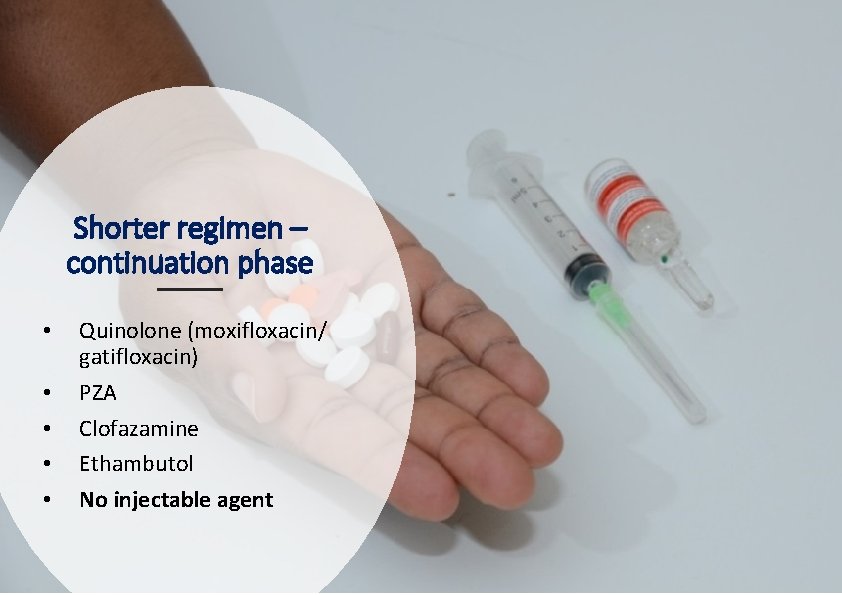
Shorter regimen – continuation phase • • • Quinolone (moxifloxacin/ gatifloxacin) PZA Clofazamine Ethambutol No injectable agent

Most recent WHO recommendations: From 2018 • An all‐oral, 18‐ 20 month regimen that contains bedaquiline is recommended in most MDR/RR‐TB cases • In some cases a shorter, 9‐ month regimen that contains an injection is recommended https: //www. who. int/tb/publications/2018/WHO. 2018. MDR‐TB. Rx. Guidelines. prefinal. text. pdf? ua=1

Conclusions

Conclusions • Each year, ~560, 000 new cases of drug‐resistant TB worldwide • On average, just over half of those who are treated for drug resistant TB are successfully treated • Based on recent research, promising new treatment regimens for MDR/RR‐TB have emerged: • Better treatment outcomes • Shorter • Some without injectable agents • There continues to be an urgent need for research to further improve MDR/RR‐TB regimens • Elimination of injectable agents in shorter regimens

Acknowledgement This presentation is made possible by the generous support of the American people through the United States Agency for International Development (USAID) through the TREAT TB Cooperative Agreement No. GHN‐A‐ 00‐ 08‐ 00004‐ 00. The contents are the responsibility of St. Peter’s Tuberculosis Hospital and do not necessarily reflect the views of USAID or the United States Government.

Thank you!
- Slides: 21