The Australian Pathology Units and Terminology Standardisation Project





























- Slides: 37

The Australian Pathology Units and Terminology Standardisation Project Michael Legg Ph. D FFSc(RCPA) FAICD FAIM FACHI 1

2

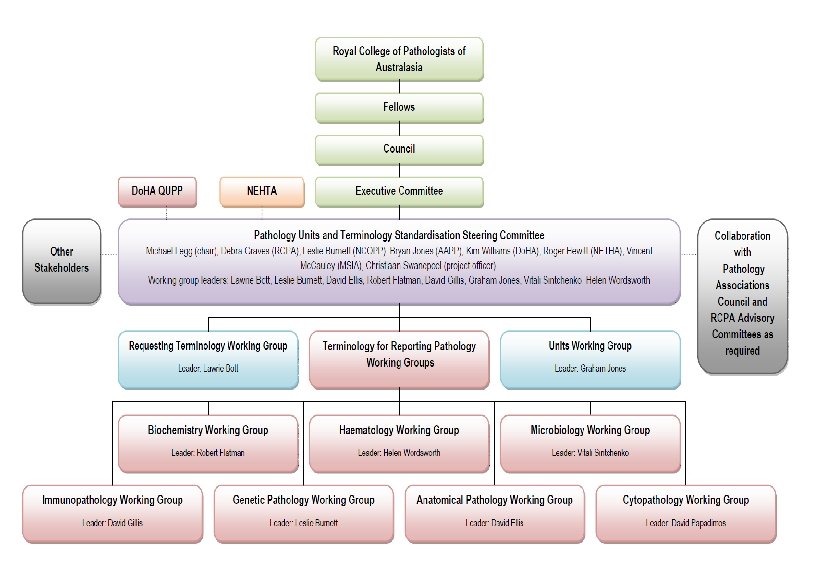
Affiliations • Chair, Pathology Units and Terminology Standardisation Project Steering Committee for the RCPA • Chair, RCPA Informatics Advisory Committee • Chair, Standards Australia IT-14 -6 -5 - Pathology Messaging • Professorial Fellow, University of Wollongong • Principal, Michael Legg & Associates 3

Today • What is the PUTS Project? • What has it done? • What does that mean? • SNOMED • LOINC • UCUM 4

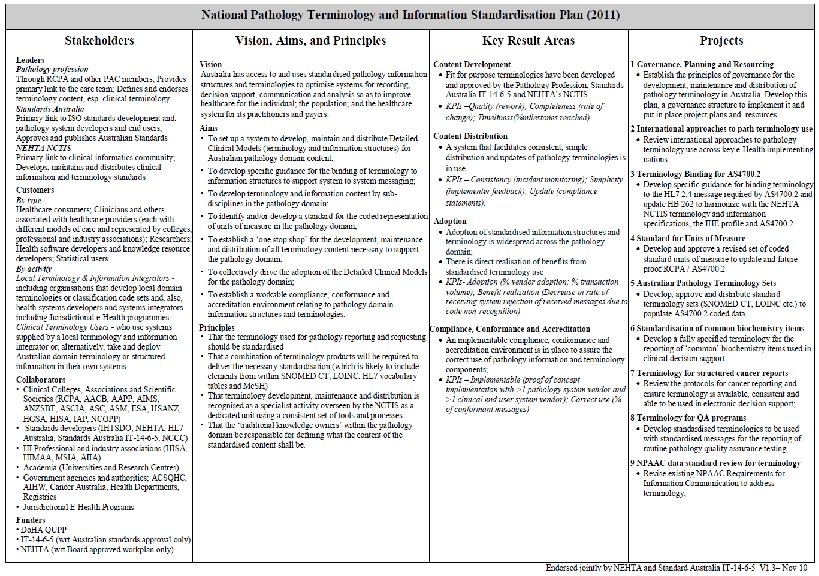
Background • A change in reporting practice • discipline-based departments reporting in isolation • episode based with results from the whole laboratory • multi-institution reporting with synthesis of results and opinions • Has led to exposure of variation and increased the risk of error • This is driving the requirement for standardisation of information structures and terminology • and units of measure • A national plan was developed 5

6

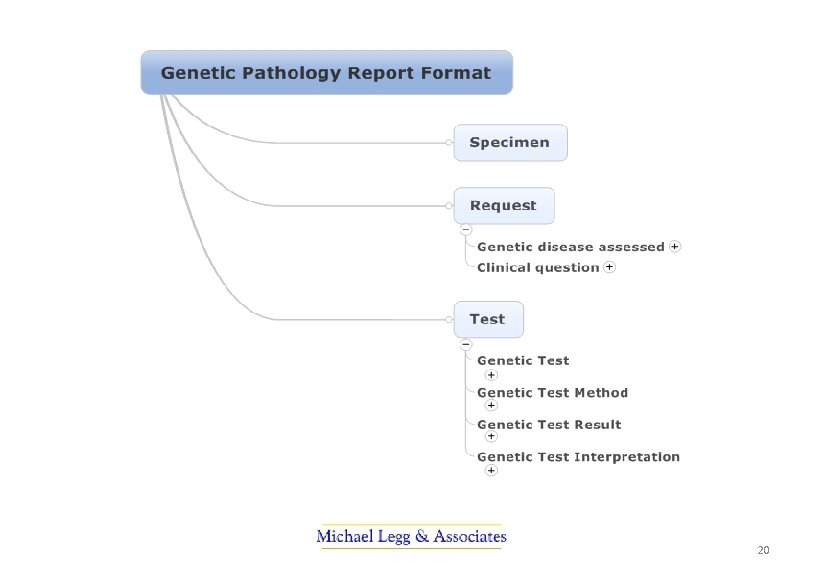
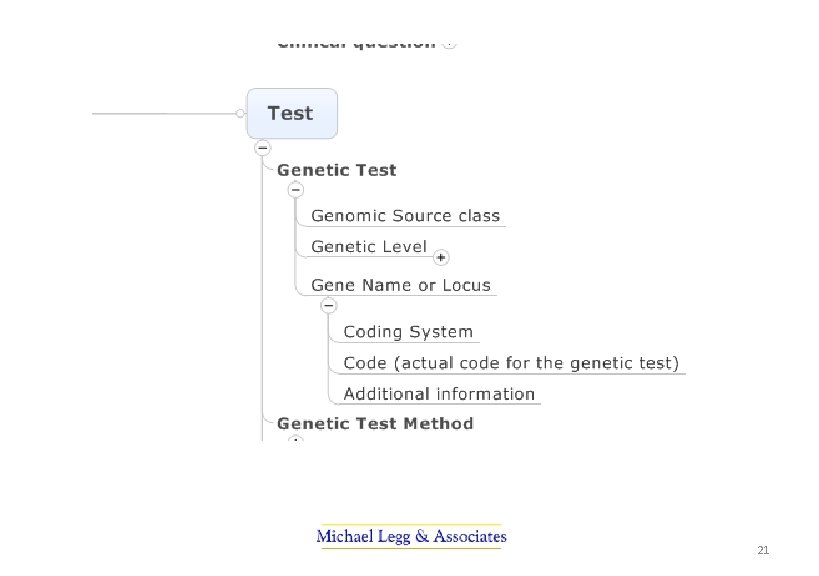
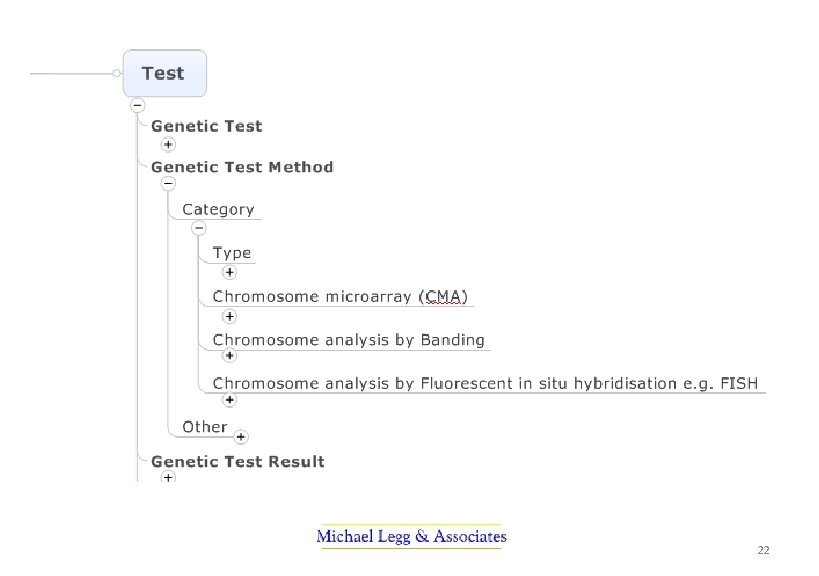
PUTS Project The profession-led projects became the Pathology Units and Standardisation Project. They were: • A revised standard for the use of units in pathology indicating preferred units for display and a mechanism for their representation in electronic messaging • Terminology sub-sets (or reference sets) of pathology terminology for requesting and reporting pathology by discipline • Standardisation of report terminology for common tests used in decision support – to improve safety in interpretation when results are combined • Terminology for structured cancer reporting ensuring terminology is available, consistent and ultimately able to be used in electronic decision support 7

8

9

10

11

12

HL 7 Result Message (ORU) MSH|^~&|||||19981105131523||ORU^R 01<cr> PID|||100928782^9^M 11||Smith^John^J<cr> OBR||||Z 0063 -0^^LN<cr> OBX||XCN|Z 0063 -0^^LN||2093467^Smits^J^<cr> OBX||Z 0092 -0^^LN||203 BE 0004 Y^^X 12 PTX<cr> Segment Data Field Component From an HL 7 Tutorial by Dr Stan Huff, Intermountain Healthcare

A code that identifies the datatype of OBX-5 Other data fields include: date of observation, identity of provider giving observation, normal ranges, abnormal flags OBX-5: Data Status OBX||NM|11289 -6^^LN||38|C^^ISO+|||||F A code that identifies the data in OBX-5 (Temp Reading) A code that identifies the units of numerical data in OBX-5 From an HL 7 Tutorial by Dr Stan Huff, Intermountain Healthcare

A real one MSH|^~&|ULTRA|Mayne Health Laverty Pathology|200312161332||ORU^R 01|243012|D|2. 3|||NE|AL|AU PID|||||XYZ^ADRIAN^J||19870410|F|||HERE^^LEICHHARDT^NSW^2040||^^^^^^N/A||||||2466093262 PV 1|||NOR|||||SEAN^JACKSON^SEAN|7 SEAN^JACKSON^SEAN^ ^^DR ORC|RE||03 -123 -CHM-0||R OBR|1||03 -123 -CHM-0|CHM^CHLAMYDIA PCR||200312161129|200305011000|||NOR|||testing line 1~and line 2|200312161129||SEAN^JACKSON^SEAN^ ^^DR||||||200312161332||SR|F||^^^200312161129|COMP^COMPUTER DEPARTMENT^TESTING^ ^^DR NTE|1|L|testing line 1 NTE|2|L|. brCurrent recommendations for the treatment of uncomplicated gonococal infections are a single intramuscular dose of ceftriaxone. For alternative treatments please discuss with our clinical microbiologists as required. A full screeen for other sexually transmitted diseases and treatment to eradicate any coexisting chlamydia infection should also be initiated. . brAs discussed . brDr Vitali Sintchenko Clinical Microbiologist (02 9005 7290 or vitali. sintchenko@maynegroup. com) OBX|1|ST|8100 -0^SPECIMEN SITE MICRO^LN||URINE||||||F|||200312161311|LEI|adrian OBX|2|ST|6357 -8^Chlamydia PCR^LN||Not detected||||||F|||200312161311|LEI|adrian OBX|3|ST|24111 -7^N GONORRHOEAE PCR^LN||DETECTED|||A|||F|||200312161311|LEI|adrian OBX|4|ST|15412 -0^ Comment: ^LN||""||||||F|||200312161311|LEI|adrian OBX|5|FT|^^PIT||001 WESTERN DIAGNOSTIC PATHOLOGY - PATHOLOGY REPORTS 05. br�02 . br�03 Report Run Number: 1686 Created: 16/12/2003 at 13: 32. br�04 Surgery: Reports: 01/05/2003 10: 00 to 01/05/2003 10: 00. br�09 --------------------------------------. br�10 DR. SEAN JACKSON 7 SEAN 1234567 X. br�19 --------------------------------------. br�20 Your Ref. Patient Name Lab Ref. Test Code. br�21 XYZ, ADRIAN J 03 -123 CHM-0 LEI. br�29 --------------------------------------. br100 Start Patient : XYZ, ADRIAN J. br101 HERE, LEICHHARDT, NSW 2040. br104 Birthdate: 10/04/1987 Age: Y 16 Sex: F. br109 . br110 Your Reference : . br111 Lab Reference : 03 -123. br112 Medicare Number: 2466093262. br115 Phone Enquiries: . br119 . br121 Referred By : DR. SEAN JACKSON. br122 Copy To : DR. TESTING COMPUTER DEPARTMENT. br123 Addressee : DR. SEAN JACKSON 1234567 X. br129 . br200 Start of Result: . br201 Specimen: . br203 Requested: 01/05/2003. br204 Collection: 01/05/2003 10: 00. br205 Name of Test: Chlamydia Pcr. br206 Reported: 16/12/2003 13: 32. br209 . br301 POLYMERASE CHAIN REACTION (PCR) . br301 Site URINE. br301 Chlamydia trachomatis DNA Not detected. br301 Neisseria gonorrhoeae DNA DETECTED . br301 Current recommendations for the treatment of uncomplicated gonococal. br301 infections are a single intramuscular dose of ceftriaxone. For alternative. br301 treatments please discuss with our clinical microbiologists as required. A. br301 full screeen for other sexually transmitted diseases and treatment to. br301 eradicate any coexisting chlamydia infection should also be initiated. . br301 As discussed. br301 Dr Vitali Sintchenko Clinical Microbiologist (02 9005 7290 or. br301 vitali. sintchenko@maynegroup. com). br301 Requested Tests: HEP, CHM, . br309 . br390 End of Report: . br399 --------------------------------------. br999 END OF LISTING - Run Number: 1686 16/12/2003 13: 32. br||||||F

So we are all using HL 7, what is the problem? • Site 1: OBX|1|CE|ABO^ABO GROUP||O^Type O| • Site 2: OBX|1|CE|BLDTYP^ABO GROUP||TYPEO^Type O| • Site 3: OBX|1|CE|ABOTYPE^ABO GROUP||OPOS^Type O| You and I may know that these are similar results, but our computers will not. From an HL 7 Tutorial by Dr Stan Huff, Intermountain Healthcare

The goal • Site 1: OBX|1|CE|883 -9^ABO GROUP||F-D 1250^Group O| • Site 2: OBX|1|CE|883 -9^ABO GROUP||F-D 1250^Group O| • Site 3: OBX|1|CE|883 -9^ABO GROUP||F-D 1250^Group O| Agree on a universal coding system From an HL 7 Tutorial by Dr Stan Huff, Intermountain Healthcare

Terminology • The standardisation of pathology terminology and units in Australia is desirable and achievable • No single existing terminology will be sufficient • Having well-developed subsets of terms will improve conformance, compliance and efficiency • A high level of knowledge and familiarity with the practice of pathology is required to develop and maintain these subsets • The terms used in Australia should reflect common usage but be consistent and safe. • The terms should also be practical and capable of ready implementation • All pathology terminology and associated units should be available in one place 18

19

20

21

22

23

Pre and post coordination • There are two ways of dealing with the development of terms that cover multiple concepts such as is needed for identifying most tests in pathology • Have a grammar that allows for the fundamental terms to be put together to make a compound statement – postcoordination eg sodium observationt+substance conc+point in time+serum+quant+ISE • Have a different term for each unique set of combinations of characteristics (concepts) – pre-coordination 24

SNOMED • SNOMED (Systematized Nomenclature of Medicine) is a systematically organised, computer processable collection of medical terms providing codes, terms, synonyms and definitions covering diseases, findings, procedures, microorganisms, substances, etc. • The International Health Terminology Standards Development Organisation administers SNOMED http: //www. ihtsdo. org/ NEHTA is the Australian Release Centre http: //www. nehta. gov. au/connectingaustralia/terminology-and-information/clinicalterminology/snomed-ct-au 25

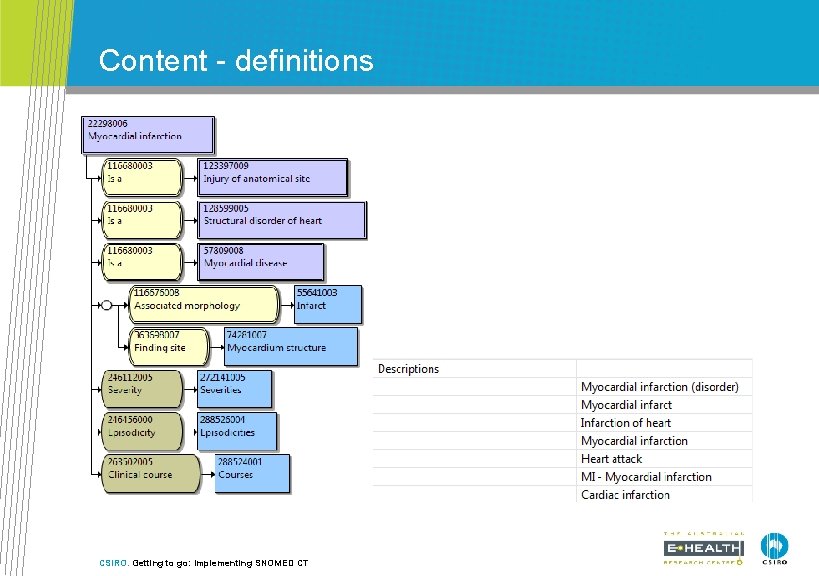
Content - definitions CSIRO. Getting to go: Implementing SNOMED CT

LOINC • Logical Observation Identifiers Names and Codes (LOINC) is a database of terms and standard for identifying medical laboratory observations. • It was developed and is maintained by the Regenstrief Institute - http: //loinc. org/ 27

LOINC • LOINC has six axes to its pre-co-ordination. For each code (eg 2951 -2) they are: • Component (analyte) – Sodium • Property measured – Substance concentration • Timing – A point in time • System – Serum (or plasma) • Scale – Quantitative (mmol/L) • Method used – ISE (but this is only used where different methods give clinically significant different results) 28

PUTS Terminology • SNOMED is to be used as the preferred terminology for requesting pathology • LOINC is to be used as the preferred terminology for the highest level test name in reporting pathology 29

PUTS Reporting • A rendering of the pathology report as the issuing laboratory intends it to be read must be sent by the laboratory in all electronic messages and receiving systems should be able to conveniently display this to the reader for review if it is not used as the primary display form • Combining data from what appears to be the same test in time series for a subject such as in cumulative reports or graphs carries with it significant clinical risk and should only be done after that risk has been properly assessed • This same variation in results means caution is required when grouping results from different laboratories, methods or times for research or other statistical purposes 30

Units - Background • RCPA Standard since 1973 but significant variation • The units system – somewhere between Europe and the Americas yielding a hybrid of common practices eg substance concentration (eg mmol/L) and mass units (mg/L) and sometimes its own conventions • Its rendering – eg Creatinine Clearance: m. L/min, m. L/min/1. 7 m 2, m. L/sec • Whether it is computable – eg 24 hour urine potassium: mmol/day, mmol/d, mmol/24 hrs, mmol/24 hr, mmol/24 h, mmol/24 hour • While unlikely to be misinterpreted by human readers this is a significant challenge to a computer • To address this issue, the Unified Code for Units of Measure (UCUM) was developed 31

UCUM • The Unified Code for Units of Measure is a code system intended to include all units of measures with the purpose of facilitating unambiguous electronic communication of quantities together with their units • The focus is on electronic communication, as opposed to communication between humans, but the coded unit is easily read eg L/(24. h), m. L/min/{1. 73_m 2}, mmol/L • UCUM is administered by the Regenstrief Institute http: //unitsofmeasure. org/ 32

PUTS Units • The standardisation of units used for reporting pathology in Australia is desirable and achievable • A single, test-specific, standardised unit of measure is preferred for use in reports from pathology laboratories • Units should be represented in electronic messages in such a way that receiving systems can readily convert units under the clinical governance of the receivers • The Unified Code for Units of Measure (UCUM) is to be used as the logical representation of units of measure in electronic messages • Numeric results should always have the appropriate units associated with them and they should never be displayed without them 33

34

35

36

37