The Atom Elements and The Periodic Table of

The Atom, Elements and The Periodic Table of Elements Complete the work sheet as we review this Power. Point presentation.

The word “Atom” means…. . “not to be divided”. Definition: An atom is the smallest particle into which an element can be divided and still be the same substance. (+) charge neutron no charge electron (-) charge proton Atomic nucleus
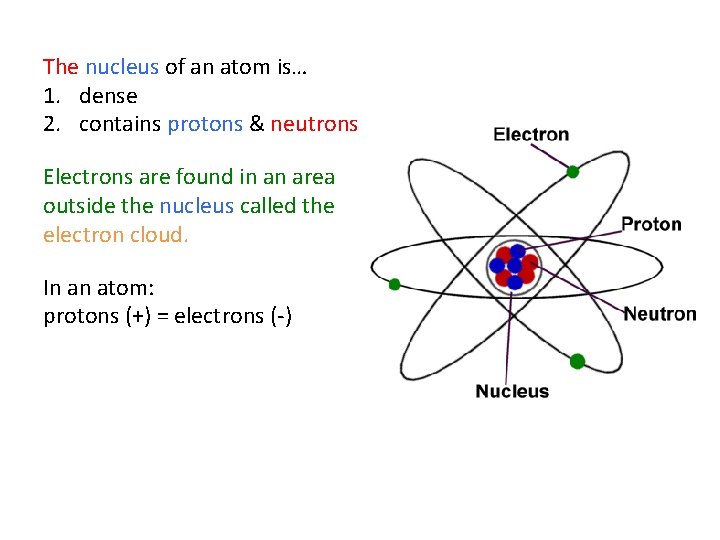
The nucleus of an atom is… 1. dense 2. contains protons & neutrons Electrons are found in an area outside the nucleus called the electron cloud. In an atom: protons (+) = electrons (-)
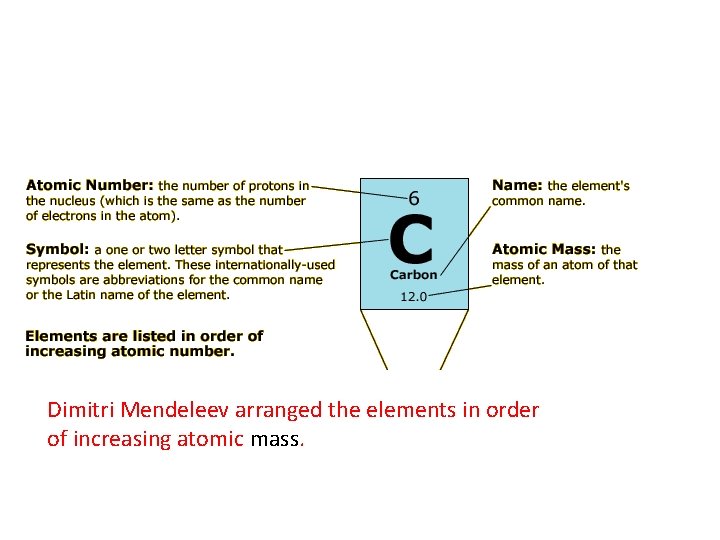
Dimitri Mendeleev arranged the elements in order of increasing atomic mass.
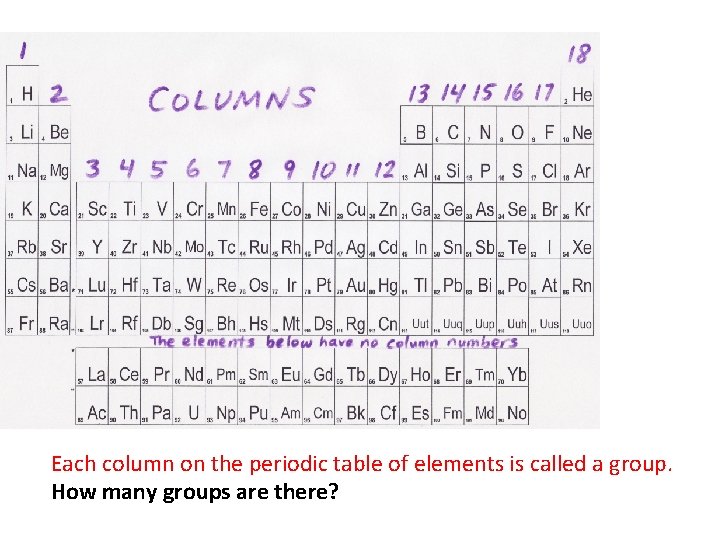
Each column on the periodic table of elements is called a group. How many groups are there?
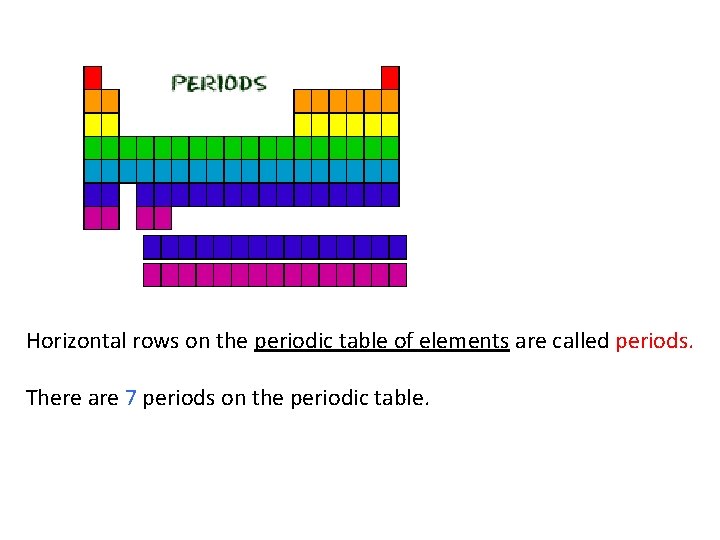
Horizontal rows on the periodic table of elements are called periods. There are 7 periods on the periodic table.
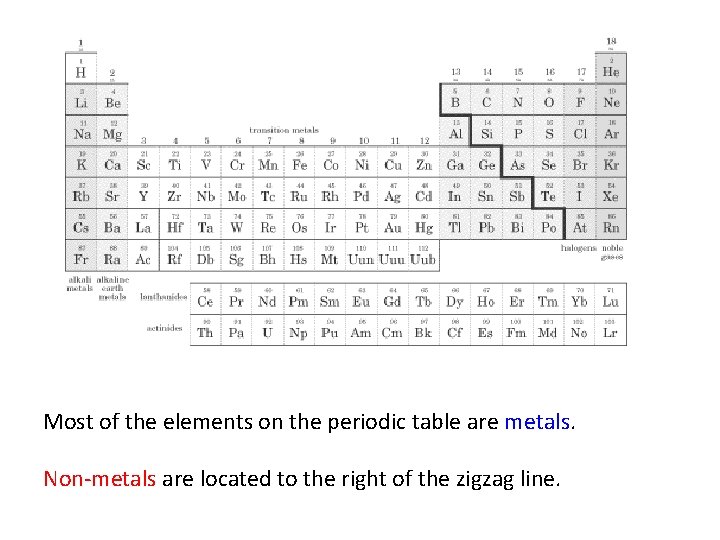
Most of the elements on the periodic table are metals. Non-metals are located to the right of the zigzag line.
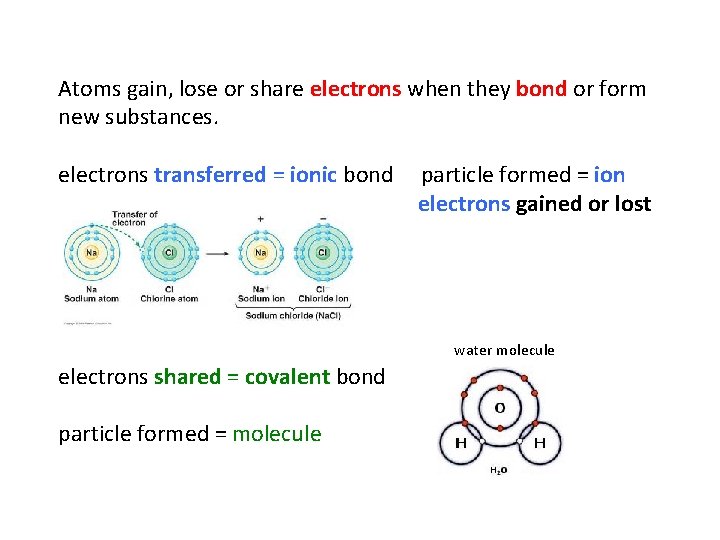
Atoms gain, lose or share electrons when they bond or form new substances. electrons transferred = ionic bond particle formed = ion electrons gained or lost water molecule electrons shared = covalent bond particle formed = molecule
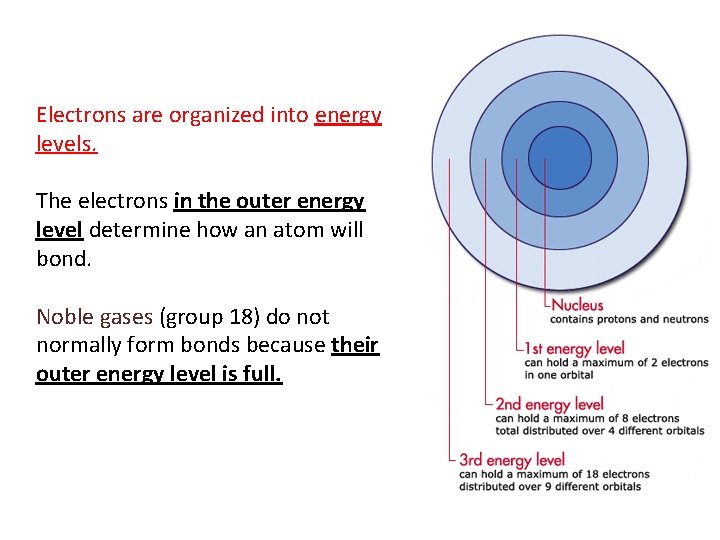
Electrons are organized into energy levels. The electrons in the outer energy level determine how an atom will bond. Noble gases (group 18) do not normally form bonds because their outer energy level is full.
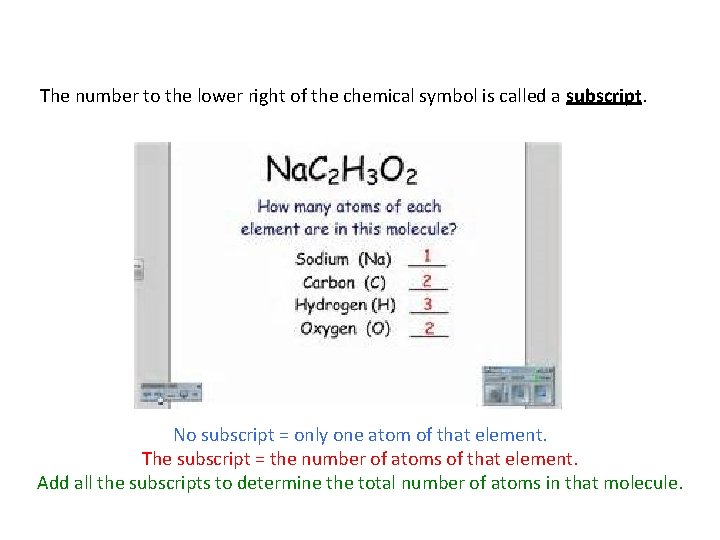
The number to the lower right of the chemical symbol is called a subscript. No subscript = only one atom of that element. The subscript = the number of atoms of that element. Add all the subscripts to determine the total number of atoms in that molecule.

How are elements grouped on the Periodic Table of Elements ? Periodic Table Video
- Slides: 11