The Atom and Elements Democritus 460 370 BC

The Atom and Elements

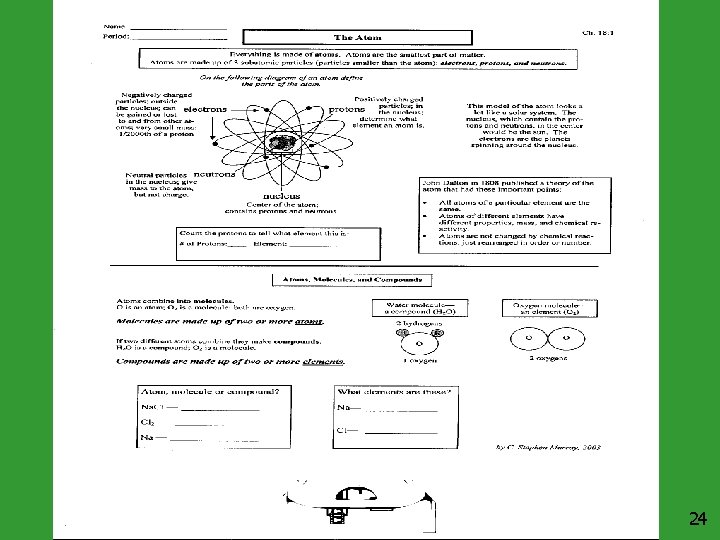
Democritus (460 -370 BC) Proposed that matter was made of small particles he called atoms. In Greek this means indivisible or cannot be divided. He believed different atoms would vary in size and would be in constant motion. Extremely small particles in motion John Dalton (1766 -1844) Published the atomic theory: 1. Elements were composed of atoms. 2. Atoms of a given element are identical. 3. Atoms of different elements have different properties. 4. Atoms don’t change, but can be combined and rearranged with other atoms. 5. Compounds are formed when atoms of more than one element combine. Joseph John Thomson (1856 -1940) Identified the electron, which carries a negative charge. He thought that electrons were embedded in the atom like raisins in raisin bread. Atom as solid object with a positive center and electrons embedded in the atom 2

Ernest Rutherford (1871 -1937) Used high speed lightweight atoms called alpha particles to bombard very thin gold foil. Most of these alpha particles passed through the gold foil. The fact that these particles went through the foil lead to his theory that atoms have mostly empty space. Alpha particles went through spaces. Neils Bohr (1885 -1962) Described the electrons moving around the nucleus in fixed orbits. Each orbit has a set amount of energy. We use this model for a basic understanding of the atom’s structure. However, more recent research has shown that electrons move around the nucleus in waves rather than elliptical orbits. Electrons are better represented as an “electron cloud. ” Electrons move so fast that scientists prefer the atom model where electrons are represented by a cloud. James Chadwick (1891 -1974) Solved the problem of “missing mass” in the atom by discovering the neutron. 3
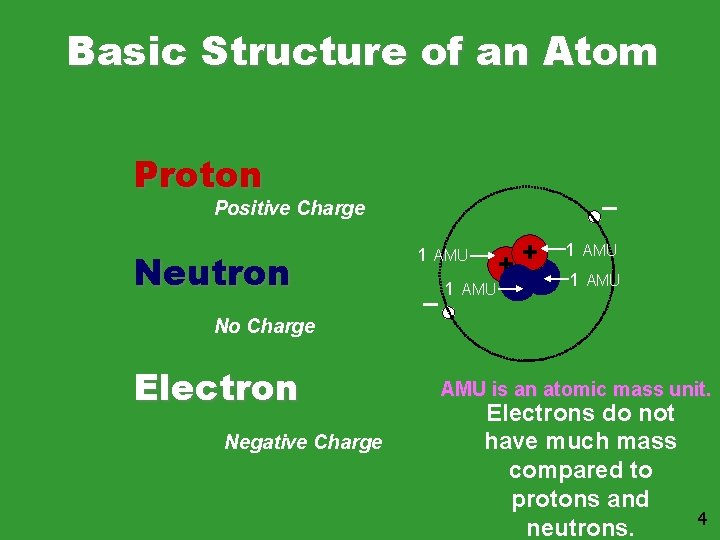
Basic Structure of an Atom Proton Positive Charge Neutron 1 AMU ++ 1 AMU No Charge Electron Negative Charge AMU is an atomic mass unit. Electrons do not have much mass compared to protons and neutrons. 4

5
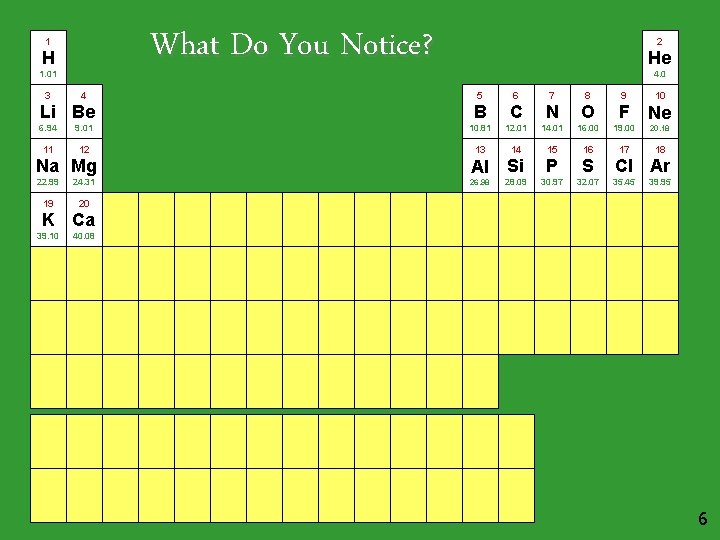
What Do You Notice? 1 H 1. 01 3 4 Li Be 2 He 4. 0 5 6 7 8 9 10 B C N O F Ne 6. 94 9. 01 10. 81 12. 01 14. 01 16. 00 19. 00 20. 18 11 12 13 14 15 16 17 18 Na Mg Al Si P S 22. 99 24. 31 26. 98 28. 09 30. 97 32. 07 19 20 Cl Ar 35. 45 39. 95 K Ca 39. 10 40. 08 6
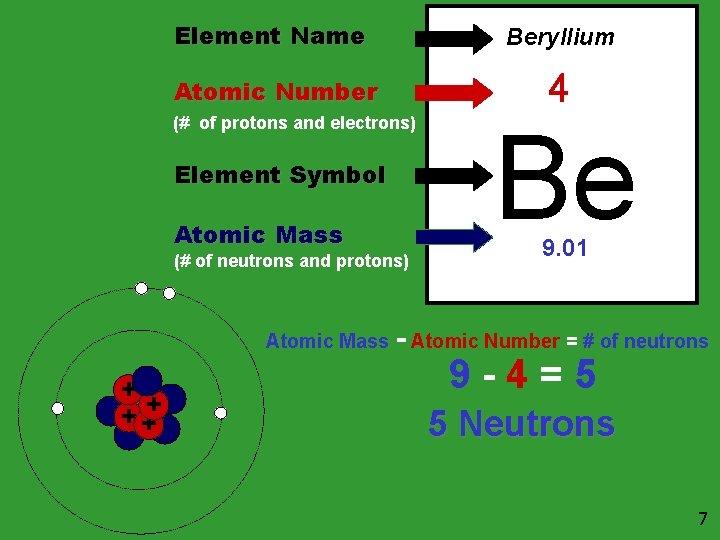
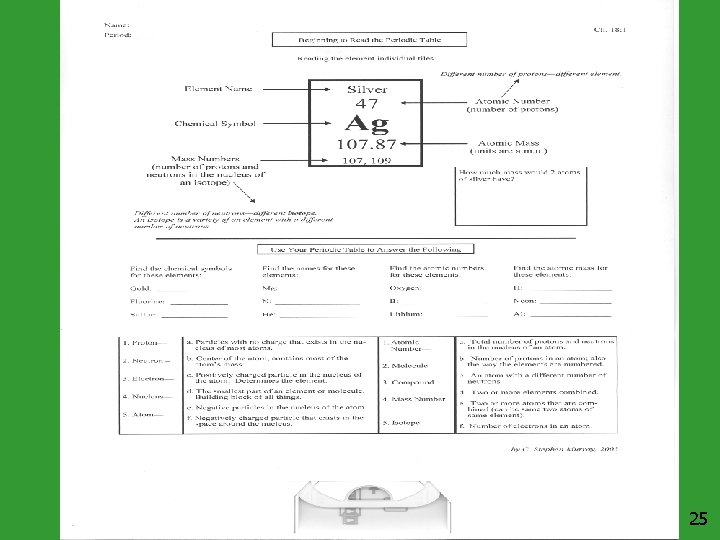
Element Name Beryllium Atomic Number 4 (# of protons and electrons) Element Symbol Atomic Mass (# of neutrons and protons) Atomic Mass + + ++ Be 9. 01 - Atomic Number = # of neutrons 9 -4=5 5 Neutrons 7

Mass Number Mass number is the number of protons and neutrons in the nucleus of an isotope: Mass # = p+ + n 0 p+ n 0 e- Mass # 8 10 8 18 Arsenic - 75 33 42 33 75 Phosphorus - 31 15 16 15 31 Nuclide Oxygen - 18 8

What are the numbers of protons, electrons and neutrons? Nitrogen 7 N P=7 E=7 N =7 14. 01 9

What are the numbers of protons, electrons and neutrons? Fluorine 9 F P=9 E=9 N = 10 19. 00 10
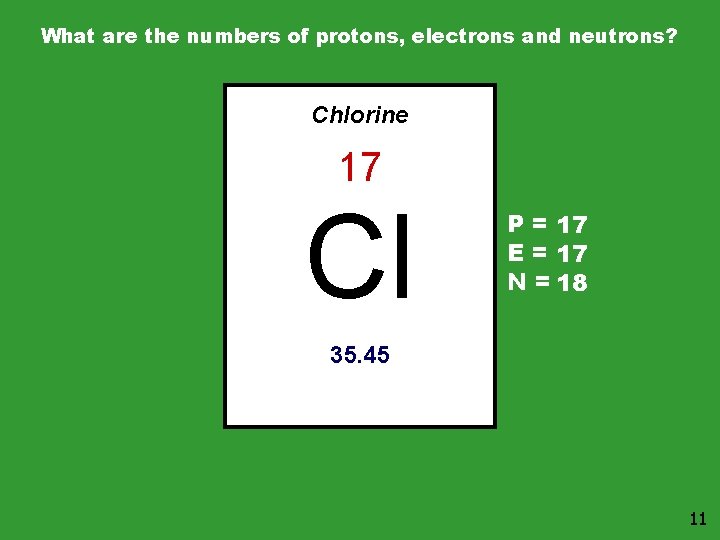
What are the numbers of protons, electrons and neutrons? Chlorine 17 Cl P = 17 E = 17 N = 18 35. 45 11
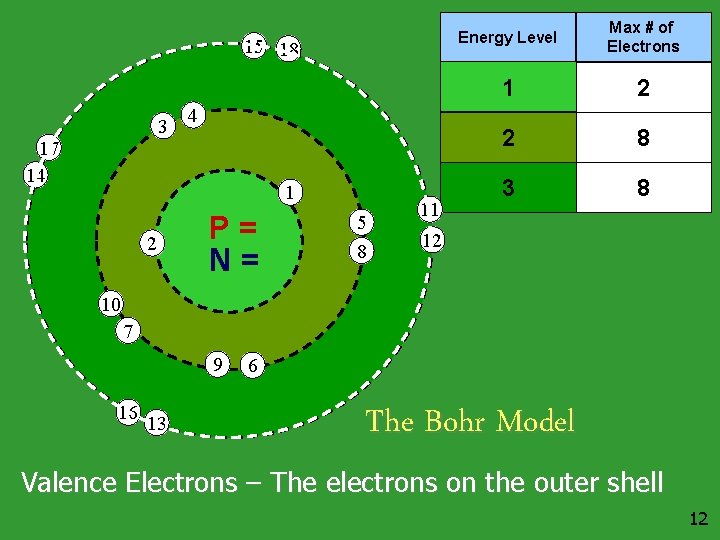
15 18 3 17 14 4 1 2 P= N= 5 8 11 Energy Level Max # of Electrons 1 2 2 8 3 8 12 10 7 9 16 13 6 The Bohr Model Valence Electrons – The electrons on the outer shell 12

Complete Symbols • Contain the symbol of the element, the mass number and the atomic number. Mass Superscript → number Subscript → Atomic number X

Symbols n If an element has 78 electrons and 117 neutrons what is the a) Atomic number b) Mass number c) number of protons d) complete symbol

Symbols n If an element has 91 protons and 140 neutrons what is the a) Atomic number b) Mass number c) number of electrons d) complete symbol

Symbols n If an element has an atomic number of 34 and a mass number of 78, what is the: a) number of protons b) number of neutrons c) number of electrons d) complete symbol

Symbols n Find each of these: a) number of protons b) number of neutrons c) number of electrons d) Atomic number e) Mass Number 80 35 Br
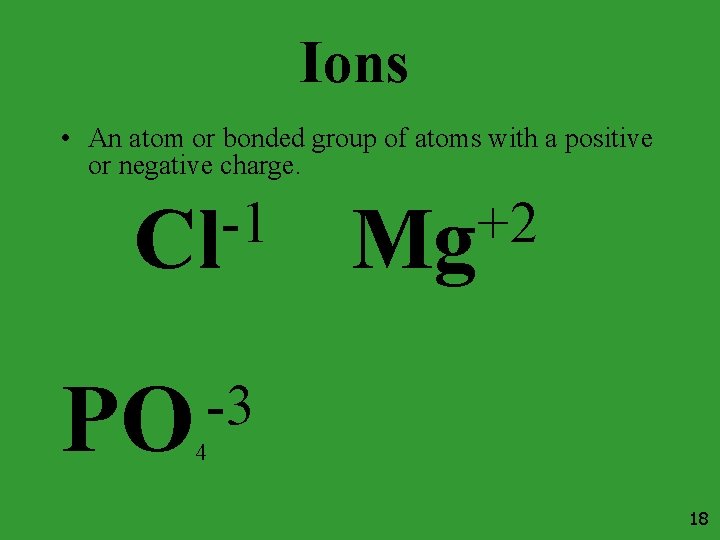
Ions • An atom or bonded group of atoms with a positive or negative charge. -1 Cl PO +2 Mg -3 4 18

Measuring Atomic Mass • Instead of grams, the unit we use is the Atomic Mass Unit (amu) • It is defined as one-twelfth the mass of a carbon-12 atom. – Carbon-12 chosen because of its isotope purity. • Each isotope has its own atomic mass, thus we determine the average from percent abundance.

Elements usually have differing amount of neutrons. These different forms are called isotopes. Let’s take hydrogen as an example: As you can see from the atomic mass (1. 01) the most naturally abundant form of hydrogen does not have a neutron. However, in rare instances isotopes form. Below are the isotopes of hydrogen: 1 Hydrogen H 1. 01 Protium Deuterium Tritium …. are isotopes of hydrogen. 20
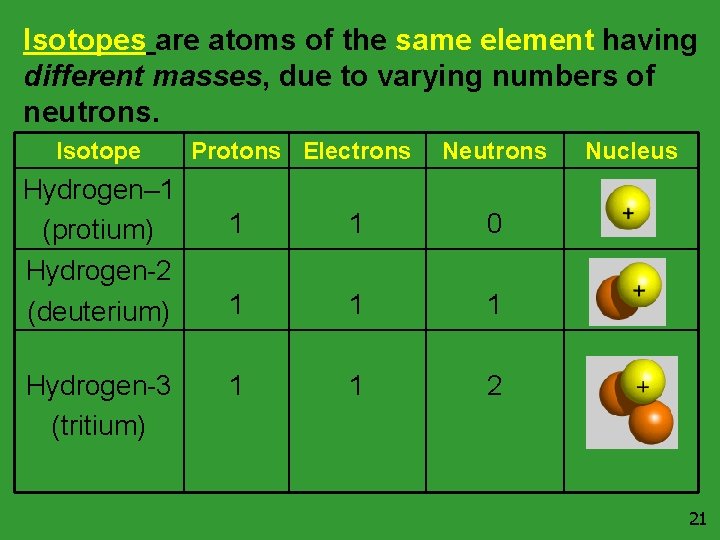
Isotopes are atoms of the same element having different masses, due to varying numbers of neutrons. Isotope Hydrogen– 1 (protium) Hydrogen-2 (deuterium) Hydrogen-3 (tritium) Protons Electrons Neutrons 1 1 0 1 1 1 2 Nucleus 21

- Page 117 Question Knowns and Unknown Solution Answer 22
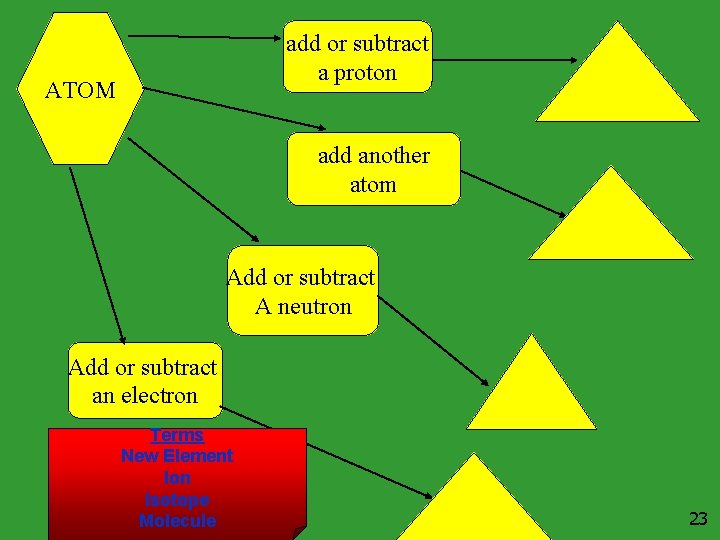
add or subtract a proton ATOM add another atom Add or subtract A neutron Add or subtract an electron Terms New Element Ion Isotope Molecule 23
24
25
- Slides: 25