The Analysis of Steroid Hormone Drugs Chapter 10






























- Slides: 50

The Analysis of Steroid Hormone Drugs Chapter 10 甾体激素类药物分析

The main contents: Ø 甾体激素类药物的基本结构特点与分类 Ø The structure, characteristics and classification; Ø 甾体激素药物的鉴别试验 Ø The identification tests; Ø 甾体激素药物的特殊杂质检查 Ø The specific impurity tests; Ø 甾体激素药物的含量测定 Ø Assays.

Fundamental requirement: Ø 掌握各类甾体激素药物的基本结构特点、鉴别 实验及含量测定方法; Ø Mastering the general structure characteristics、 identification tests and assays of steroid hormones; Ø 熟悉HPLC在鉴别试验及含量测定中的应用; Ø Familiarizing with the application of HPLC in identification tests and assays; Ø 掌握IR在甾体激素类药物鉴别中的应用。 Ø Mastering the application of IR in identification tests.

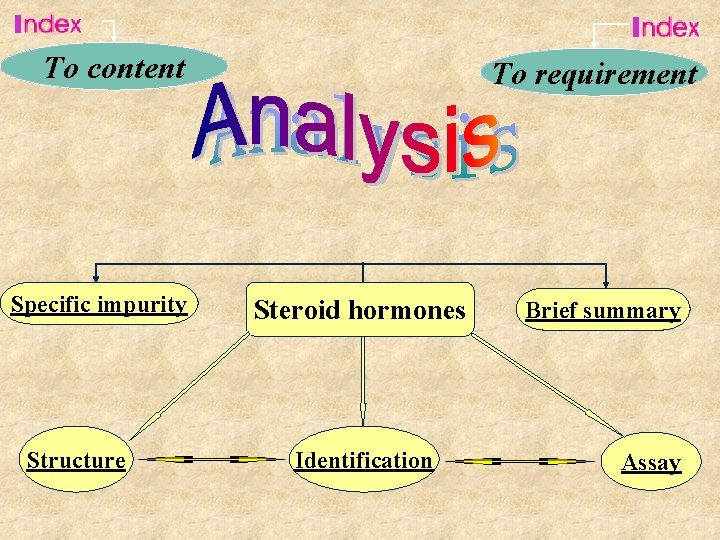
To content Specific impurity Structure To requirement Steroid hormones Identification Brief summary Assay


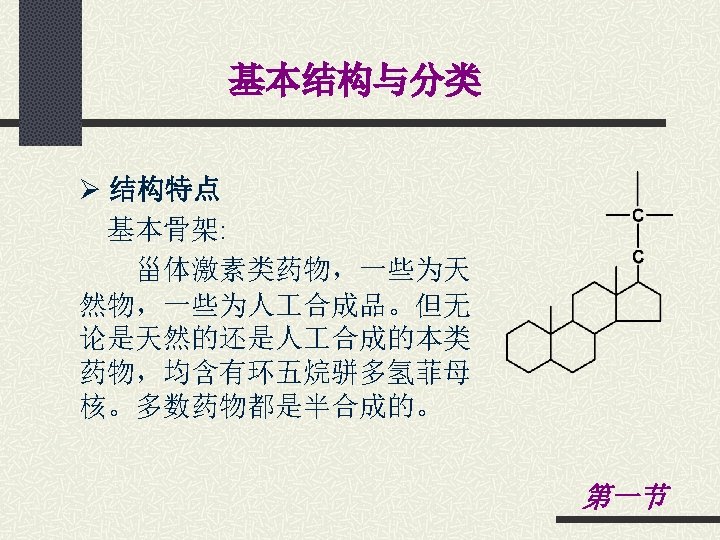
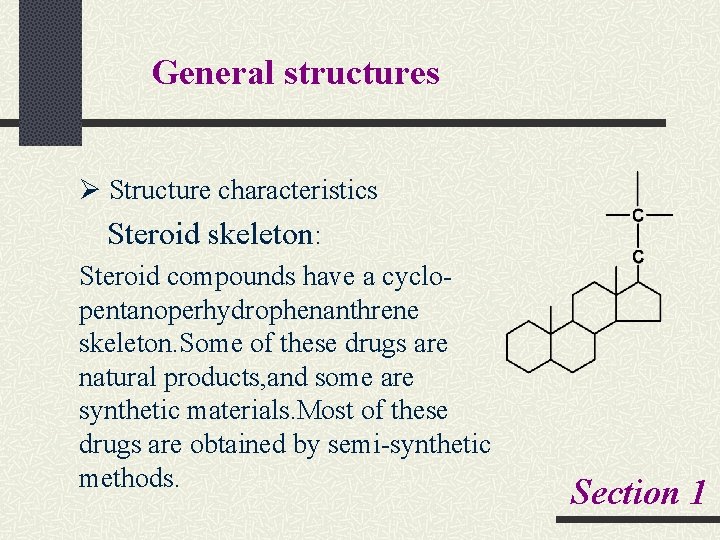
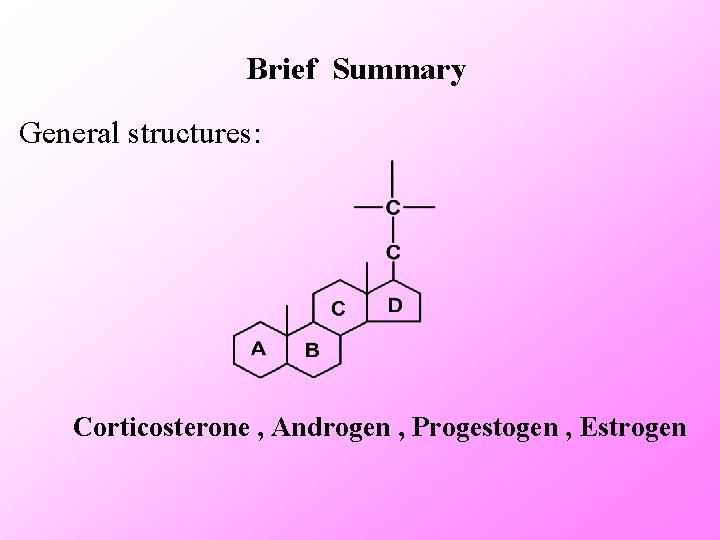
General structures Ø Structure characteristics Steroid skeleton: Steroid compounds have a cyclo- pentanoperhydrophenanthrene skeleton. Some of these drugs are natural products, and some are synthetic materials. Most of these drugs are obtained by semi-synthetic methods. Section 1

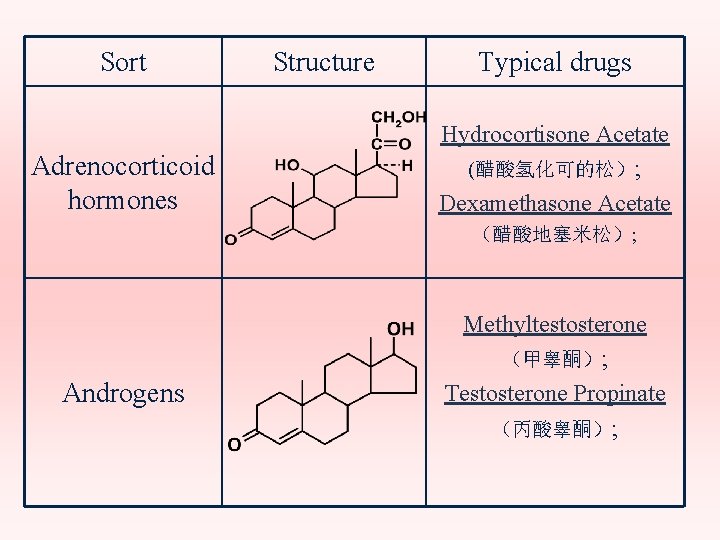
Sort Adrenocorticoid hormones Structure Typical drugs Hydrocortisone Acetate (醋酸氢化可的松); Dexamethasone Acetate (醋酸地塞米松); Androgens Methyltestosterone (甲睾酮); Testosterone Propinate (丙酸睾酮);

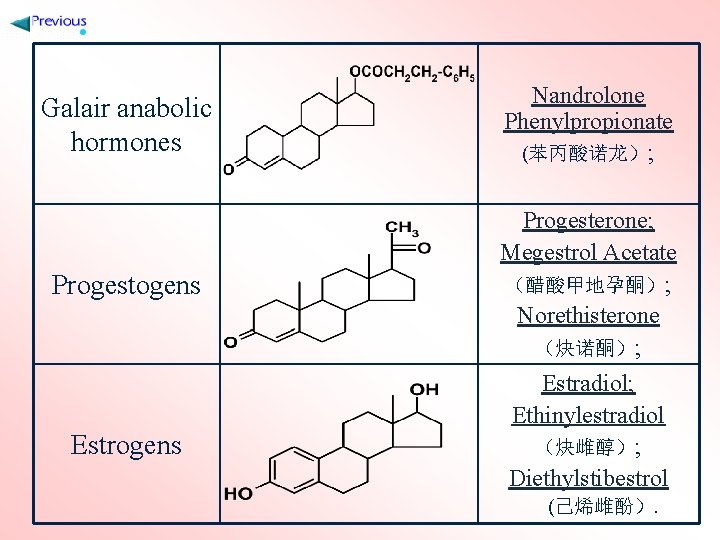
Galair anabolic hormones Progestogens Estrogens Nandrolone Phenylpropionate (苯丙酸诺龙); Progesterone; Megestrol Acetate (醋酸甲地孕酮); Norethisterone (炔诺酮); Estradiol; Ethinylestradiol (炔雌醇); Diethylstibestrol (己烯雌酚).


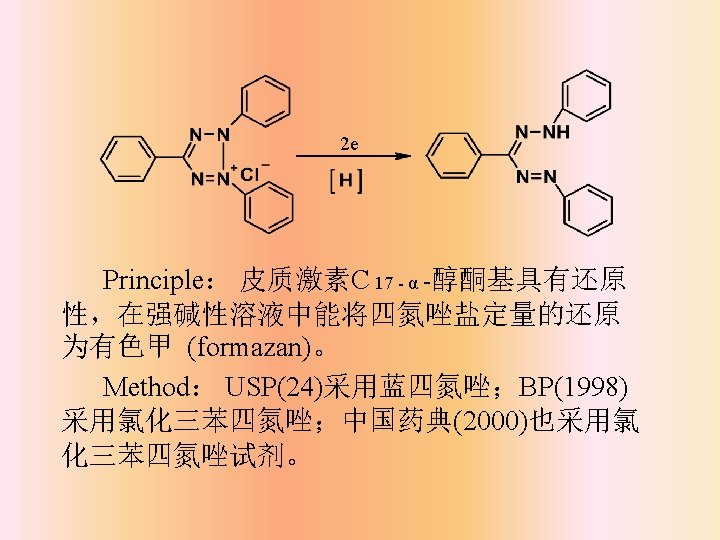
Identification tests Δ 4 -3 -keto — Ultraviolet spectrophotometry Ketone group — Color reaction (with carbonyl reagents) — Melting point determination of derivatives — Reaction with strong acids Functional group — Infrared spectrophotometry α-hydroxyacetone — Color reaction(with tetrazoline salt) — Precipitation reaction Methylketone group — Color reaction(with sodium nitroferricyanide , m-dinitrophenol, aromatic aldehyde ) Section 2


Identification tests Organic fluorine — Color reaction(with alizarin complexone and Cerous nitrate after destroyed by oxygen flask combustion ) Organic chlorine — Precipitation reaction(with nitric acid and silver nitrate after destroyed by oxygen flask combustion) Phenolic hydroxyl — Color reaction (diazobenzenesulfonic acid) — Melting point determination of derivatives Alkynyl group — Precipitation reaction(with silver nitrate) Ester group — Hydrolysis product reaction Section 2

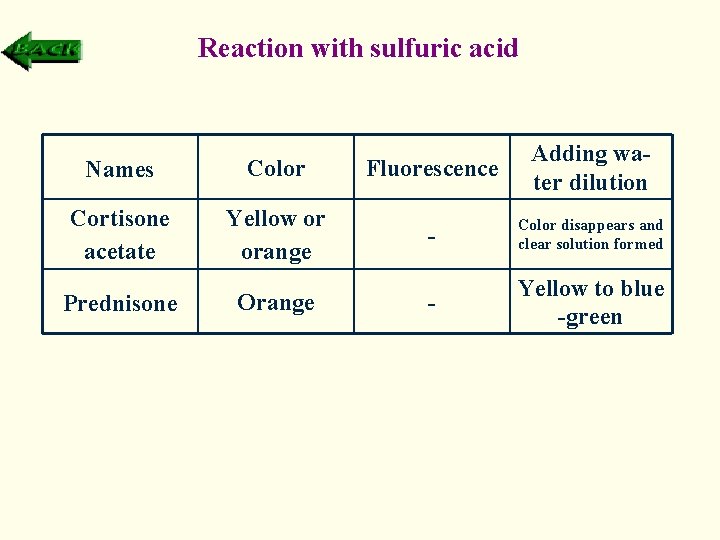
Reaction with sulfuric acid Names Color Fluorescence Adding water dilution Cortisone acetate Yellow or orange - Color disappears and clear solution formed - Yellow to blue -green Prednisone Orange

Ultraviolet spectrophotometry Conjugated bonding systems: 1. α, β-unsaturated ketone group 2. Steroids with aromatic ring A

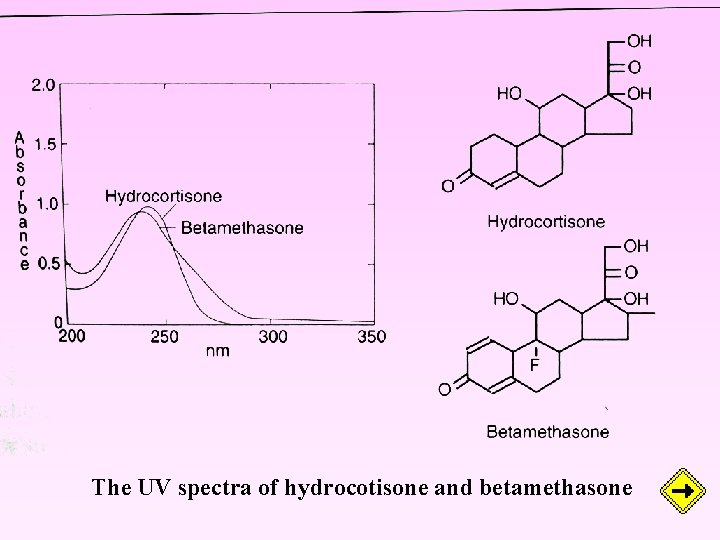
The UV spectra of hydrocotisone and betamethasone

Precipitation reaction Reaction with silver nitrate prepared in ammonia Silver nitrate Silver precipitate Cortical hormones ( black ) Ammonia Reaction with silver nitrate Steroid hormones ( alkynyl ) Silver nitrate Silver salt precipitate ( white )

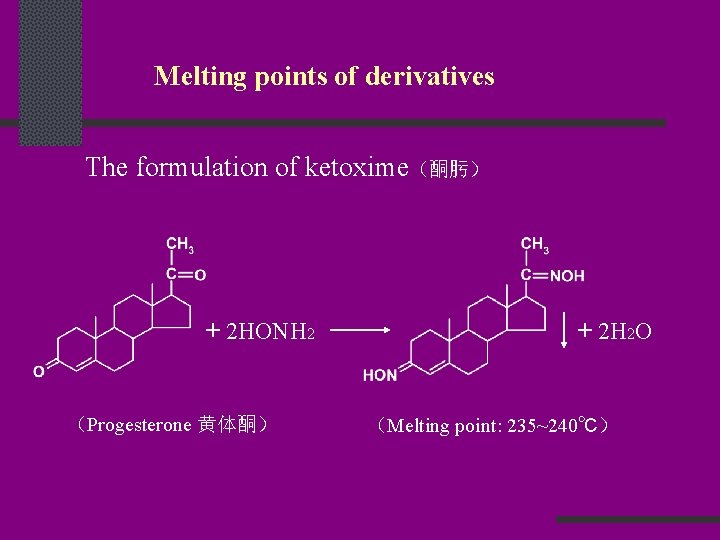
Melting points of derivatives The formulation of ketoxime(酮肟) + 2 HONH 2 (Progesterone 黄体酮) + 2 H 2 O (Melting point: 235~240℃)

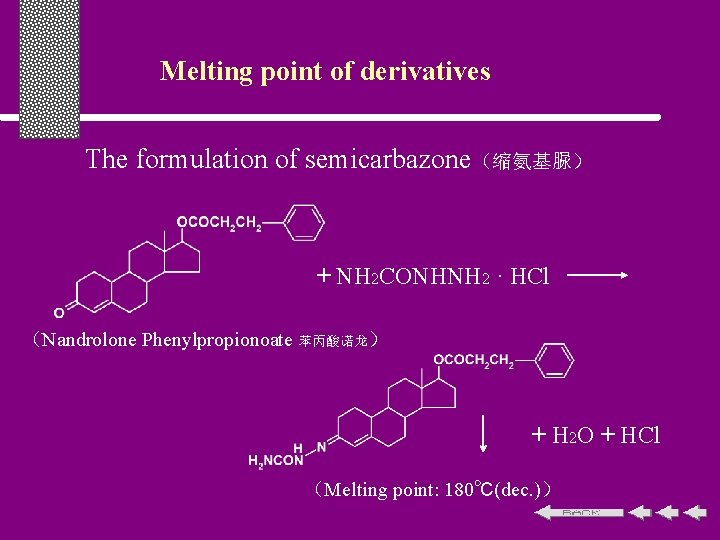
Melting point of derivatives The formulation of semicarbazone(缩氨基脲) + NH 2 CONHNH 2 · HCl (Nandrolone Phenylpropionoate 苯丙酸诺龙) + H 2 O + HCl (Melting point: 180℃(dec. ))

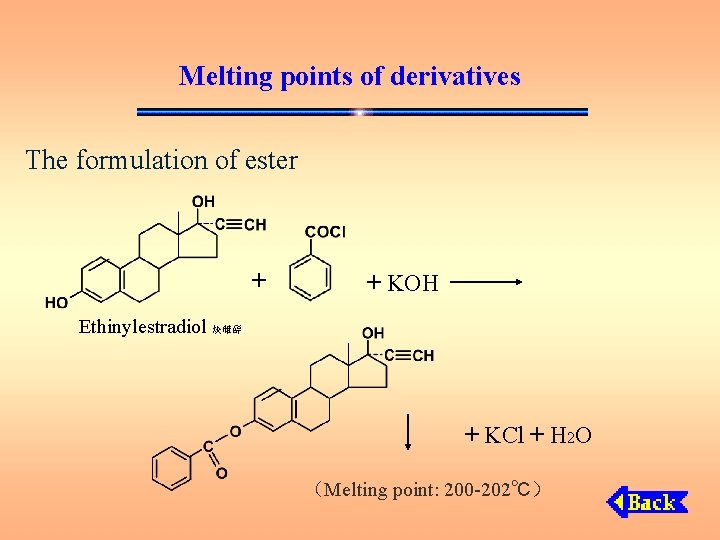
Melting points of derivatives The formulation of ester + + KOH Ethinylestradiol 炔雌醇 + KCl + H 2 O (Melting point: 200 -202℃)

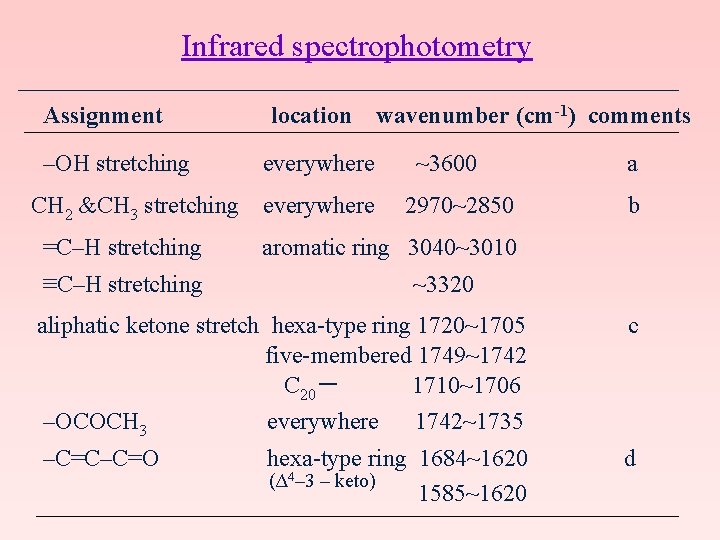
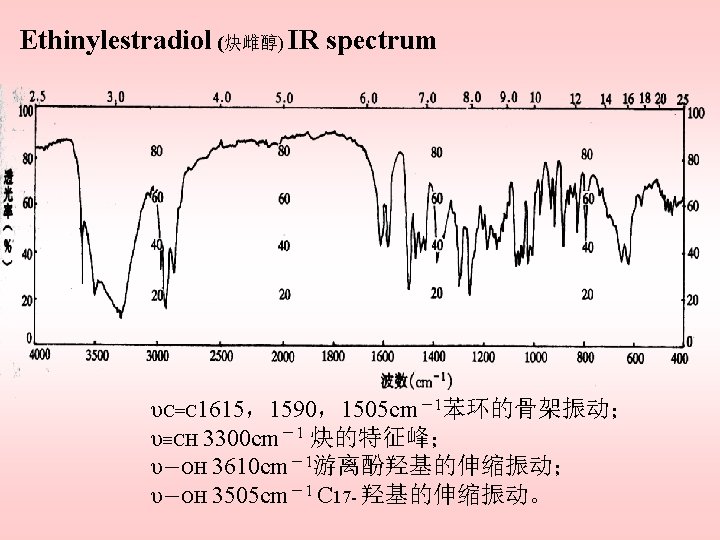
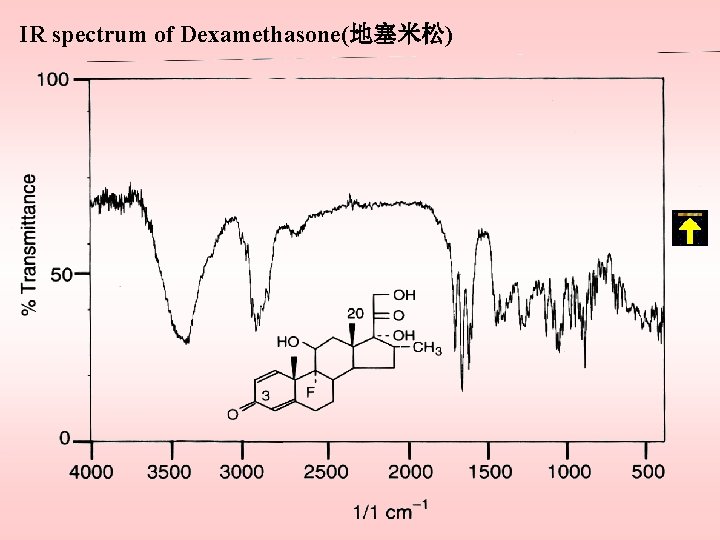
Infrared spectrophotometry Assignment location wavenumber (cm-1) comments –OH stretching everywhere ~3600 a CH 2 &CH 3 stretching everywhere 2970~2850 b =C–H stretching aromatic ring 3040~3010 ≡C–H stretching ~3320 aliphatic ketone stretch hexa-type ring 1720~1705 c five-membered 1749~1742 C 20- 1710~1706 –OCOCH 3 everywhere 1742~1735 –C=C–C=O hexa-type ring 1684~1620 d (Δ 4– 3 – keto) 1585~1620

–C–OH (alcohol ) everywhere 1230~1000 –C–OH (hydroxybenzene) 1300~1200 –OCOR 1200~1000 –C=C–H everywhere 900~650 Comments a: If hydrogen bonds with the hydroxy groups, the band shifts to 3550~3330 cm-1. b: Often appears as a shoulder. c: C=O stretching in CCl 4 occurs at a low wavenumber. d: As a result of hydrogen bonding with the hydroxy groups the νC=O band of the 3 -keto groups appears at relatively low frequency, and thus theνC=O band cannot be observed.


IR spectrum of Dexamethasone(地塞米松)


The Specific Impurity tests Steroid hormone drugs are obtained by modifying other steroid compounds or other structurally relative steroid hormones. Impurities, such as raw materials、intermediate compounds、isomers、degradation products 、 reagents and solvents etc, may be introduced into their final products. Except for general impurities, the limit of other steroids is also an important item in the purity tests. Section 3


Examples Dexamethasone sodium phosphate、hydrocortisone sodium phosphate——free phosphonic acid —— determination of A Dexamethasone sodium phosphate——methanol and acetone——GC Ethinyloestradiol ——estrone ——colorimetry Dexamethasone acetate、fluocinonide acetate—— selenium——OCF-colorimetry Steroid hormones——other steroids——TLC、 HPLC

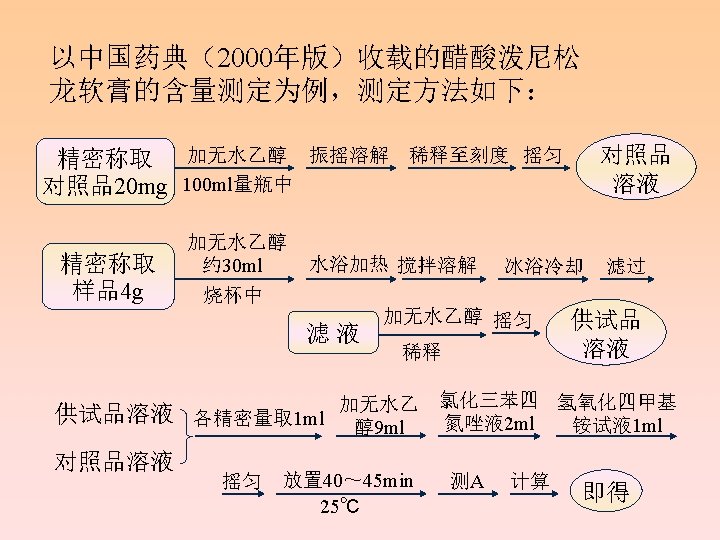
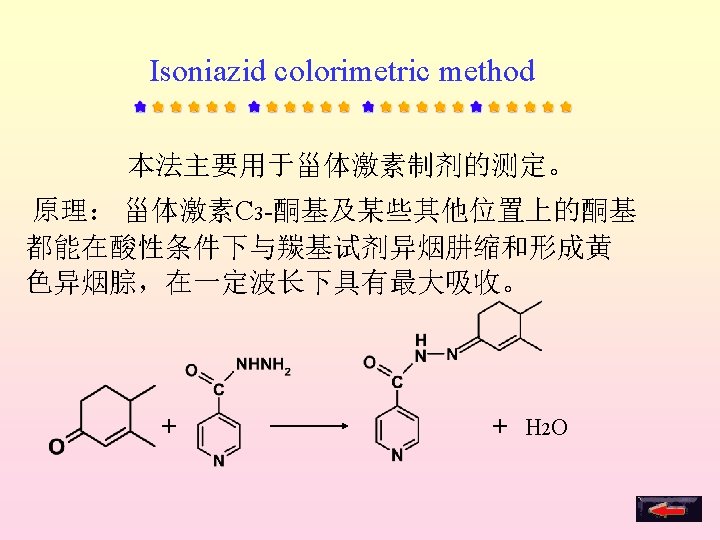
Assays 四氮唑比色法( Tetrazoline colorimetry) 异烟肼比色法(Isoniazide colorimetry) Kober反应比色法(Kober reaction colorimetry) 紫外分光光度法(UV) 高效液相色谱法(HPLC) Section 4


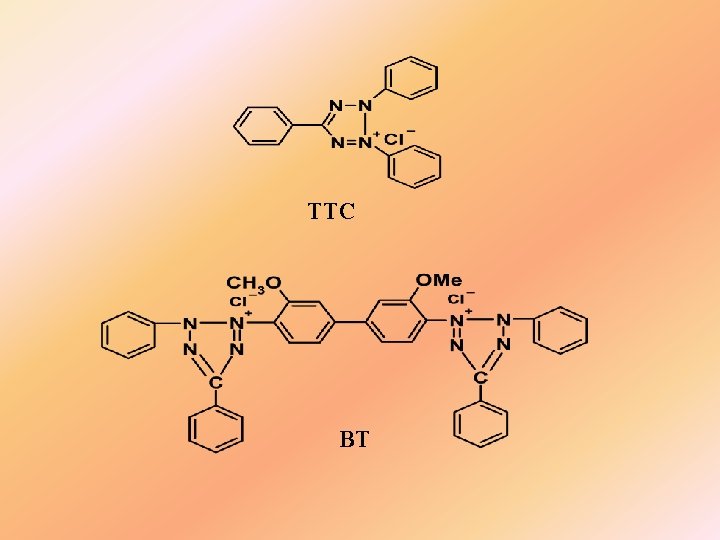
Tetrazoline colorimetry Commonly used tetrazoline salts: Red tetrazoline(RT) : 2, 3, 5 -triphenyltetrazolium chloride (TTC); Blue tetrazoline(BT): {3, 3’-dianisole-bis[4, 4’-(3, 5 -diphenyl) tetrazolium chloride]}

TTC BT





Influence factors: A. Functional groups They often influence the rate of reaction : C 11 -keto>C 11 hydroxy;C 21 -hydroxy>C 21 -hydroxyl ester。 B. Solvent and moisture Water will slow down the rate of color reaction, we use non-aqueous ethanol as the solvent instead of water. As aldehyde will make the absorbance high, we use nonaldehyde ethanol. C. Alkaline reagent D. Tetramethyl ammonium hydroxide is commonly used for a satisfied result. E. Notice: add tetrazoline salt first and then alkaline solution to prohibit the decomposition of corticosteroids.

Influence factors: D. Oxygen and light E. Because reaction products are sensitive to the ray, photophobic container is needed and placed in the dark. Immediate determinationis needed. E. Temperature and time Higher the temperature is , faster the rate of color reaction is. Generally, room temperature or 30℃ constant temperature is the best condition. The result has a better repeatability.


Kober Reaction Colorimetry: Kober reaction is a color reaction of estrogen and sulfate-ethanol when heating. The color will convert to be red when diluting with water or diluted sulfuric acid or heating again. The maximum absorption is at 515 nm. This reaction has two steps:first: estrogen is heated together with sulfuric acid- ethanol to appear yellow color, which has the maximum absorption around 465 nm; secondly: the solution appears pink when diluting with water or diluted sulfuric acid or heating again. The maximum absorption is at 515 nm.




Principle: Many steroid hormones have characteristic UV absorption because of double bonds (aromatic ring A) and their conjugation with keto groups on them. With the structure of Δ 4 -3 -keto corticoid hormones, androgens, progestogens and many per oral contraceptives have the maximum absorption at about 240 nm. With the structure of benzoic ring , estrogens have the maximum absorption at about 280 nm.


HPLC Advantages: Rapid and efficient separation; Small quantities of samples; High sensitivity Conditions: Reverse-phase: ODS silica gels; Mobile phase : methanol and water. In order to improve separation effect, acetic acid buffer or phosphate acid buffer is added to adjust the p. H value of mobile phase. UV detector.

Brief Summary General structures: Corticosterone , Androgen , Progestogen , Estrogen

Characteristics: • Color reaction: steroid ring — reaction with sulfuric acid C 17 -α- hydroxyacetone— reaction with tetrazoline salt Ketone — reaction with 2, 4 -dinitrophenyl hydrazine, isoniazide or phenylhydrazine sulfate Fluorine — reaction with alizarin complexone and cerous nitrato Phenol hydroxyl — reaction with diazobenzene sulfonic acid • Precipitation reaction: C 17 -α- hydroxyacetone — reaction with Fihling reagent — reaction with silver nitrate preparing in ammonia Ethynyl — reaction with silver nitrate Organic combine chlorine — reaction with HNO 3 -Ag. NO 3

• Melting point determination of derivative • Hydrolysis product reaction Ultraviolet spectroscopy Conjugate systems (C=C-C=O, C=C-C=C) • Infrared spectroscopy

Specific impurity tests: Dexamethasone sodium phosphate、hydrocotisone sodium phosphate— free phosphoric acid — determination of A Dexamethasone sodium phosphate—methanol and acetone—GC Ethinyloestradiol — estrone — colorimetry Dexamethasone acetate —selenium Steroid hormones——other steroids——TLC、HPLC Assays: Tetrazoline colorimetry Isoniazide colorimetric method Cober reaction colorimetry HPLC

Editor: Qiang Fu, Hongqin Ke College of Pharmacy , Xi’an Jiaotong university