The Alligation Method De Ana Smalls PBP Program

The Alligation Method De. Ana Smalls PBP Program August 4, 2016

What Are Dilutions and Concentration? Concentration – the amount of a particular substance (solute/active ingredient) present in another substance (solvent/base). Dilution - Process of making a substance weaker or dilute • • • This is usually accomplished by either adding more solvent or a solution that has a weaker concentration of active ingredient • Used to reach desired concentrations

General Understanding – When a mixture/solution is diluted, the amount of active ingredient remains constant – A sample of a mixture/solution has the same concentration as the stock solution. – If a mixture/solution is diluted to double its volume, its concentration is reduced by half – Ex: 5 g in 100 m. L = 5% w/v 5 g in 200 m. L = 2. 5% w/v – If a mixture/solution is concentrated to half its original volume, its concentration is doubled – Ex: 5 g in 100 m. L = 5% w/v 5 g in 50 m. L = 10% w/v

General Understanding –

Quantity and Concentration Example 1: If 500 m. L of a 15% v/v solution of methyl salicylate in alcohol are diluted to 1500 m. L, what will be the percentage strength v/v?

Quantity and Concentration Example 1: If 500 m. L of a 15% v/v solution of methyl salicylate in alcohol are diluted to 1500 m. L, what will be the percentage strength v/v?

Quantity and Concentration Example 2: How many milliliters of water for injection must be added to 10 liters of a 50%(w/v) dextrose injection to reduce the concentration to 30% (w/v)

Quantity and Concentration Example 3: How many milliliters of a 5% (w/v) solution of menthol in alcohol should be used to obtain the amount of menthol needed in preparing the lotion? – – Menthol 0. 1% Hexachlorophene 0. 1% Glycerin 10. 0% Alcohol 70% to make 500 m. L

Need, Have, Add Example 4: How many grams of 20% benzocaine ointment and how many grams of ointment base (diluent) should be used in preparing 5 lb of 2. 5% benzocaine ointment?

Need, Have, Add Example 4: How many grams of 20% benzocaine ointment and how many grams of ointment base (diluent) should be used in preparing 5 lb of 2. 5% benzocaine ointment? Need: 2. 5% benzocaine x = 56. 7 g needed Have: 20% benzocaine = 20 g benzocaine per 100 g of ointment Add: x = 800 g – 100 g = 700 g of base added Final Answer: 20% benzocaine = 100 g ointment base = 700 g

Need, Have, Add Example 5: How many grams of coal tar should be added to 3200 g of 5% coal tar ointment to prepare an ointment containing 20% of coal tar?

Need, Have, Add Example 6: How many grams of opium containing 15% (w/w) of morphine and how many grams of lactose should be used to prepare 150 g of opium containing 10% (w/w) of morphine?

The Box Method Example 7: How many milliliters of water should be added to 100 m. L of a 1: 125 (w/v) solution to make a solution such that 25 m. L diluted to 100 m. L will yield a 1: 4000 dilution?
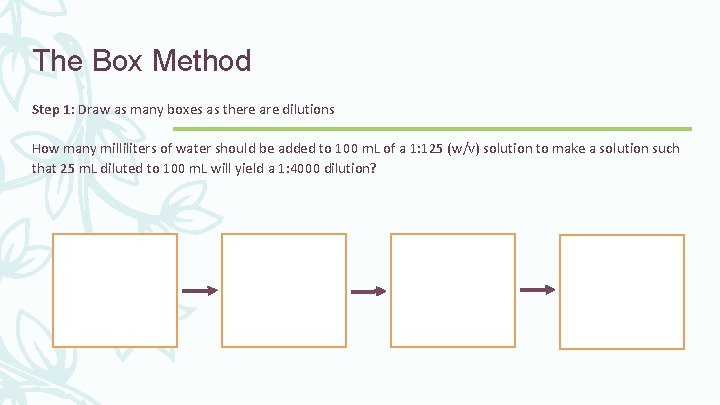
The Box Method Step 1: Draw as many boxes as there are dilutions How many milliliters of water should be added to 100 m. L of a 1: 125 (w/v) solution to make a solution such that 25 m. L diluted to 100 m. L will yield a 1: 4000 dilution?

The Box Method Step 2: Break down the problem. For each dilution there should be a volume and an amount (in grams). Include concentration if applicable. How many milliliters of water should be added to 100 m. L of a 1: 125 (w/v) solution to make a solution such that 25 m. L diluted to 100 m. L will yield a 1: 4000 dilution? Conc: 1: 125 w/v Amount: Volume: 100 + x Amount: Volume: 25 m. L Conc: 1: 4000 Amount: Volume: 100 m. L

The Box Method Step 3: Use the given values to find what is missing. Conc: 1: 125 w/v Amount: Volume: 100 + x Amount: 0. 025 g Volume: 25 m. L Conc: 1: 4000 Amount: 0. 025 g Volume: 100 m. L
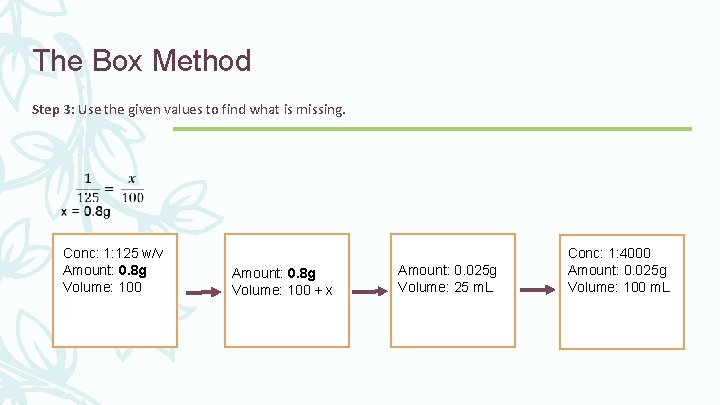
The Box Method Step 3: Use the given values to find what is missing. Conc: 1: 125 w/v Amount: 0. 8 g Volume: 100 + x Amount: 0. 025 g Volume: 25 m. L Conc: 1: 4000 Amount: 0. 025 g Volume: 100 m. L
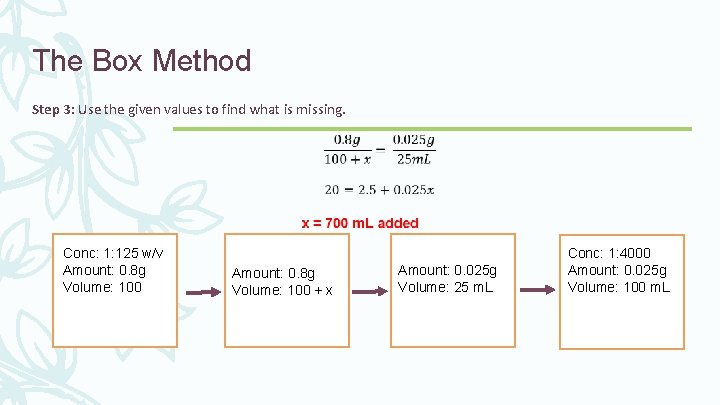
The Box Method Step 3: Use the given values to find what is missing. Conc: 1: 125 w/v Amount: 0. 8 g Volume: 100 + x Amount: 0. 025 g Volume: 25 m. L Conc: 1: 4000 Amount: 0. 025 g Volume: 100 m. L

The Box Method Example 8: A protein solution is prepared by dissolving 40 mg of protein in 10 m. L of water. 0. 5 m. L of this solution is diluted to a volume of 20 m. L. How many mg of protein will be in a 2 m. L sample of the diluted solution?

The Box Method Example 9: What weight of ingredient is required to produce 1000 m. L of a solution such that, when 2. 5 m. L of it is diluted to 50 m. L, it gives a 0. 25% w/v solution?

What Are Alligations? – A shortcut method of solving mathematical problems involving mixtures – Two types: – Alligation medial: Finds the quantity of a mixture based off the given amount of ingredients – Alligation alternate: Find the quantity of each ingredient needed to make a given amount of mixture

Intro to A. Medial: A Step by Step Guide What is the percentage (v/v) of alcohol in a mixture of 3000 m. L of 40% alcohol, 1000 m. L of 60% alcohol, and 1000 m. L of 70% alcohol? (assume no contraction of volume) This problem involves the use of alligation medial

Intro to A. Medial: A Step by Step Guide Step 1: List the ingredients and their corresponding percentages What is the percentage (v/v) of alcohol in a mixture of: – 3000 m. L of 40% alcohol – 1000 m. L of 60% alcohol – 1000 m. L of 70% alcohol

Intro to A. Medial: A Step by Step Guide Step 2: use the percentages to find the amount of alcohol present in each ingredient What is the percentage (v/v) of alcohol in a mixture of: – 3000 m. L of 40% alcohol: (3000 x 0. 4)= 1200 m. L alcohol – 1000 m. L of 60% alcohol: (1000 x 0. 6)= 600 m. L alcohol – 1000 m. L of 70% alcohol: (1000 x 0. 7)= 700 m. L alcohol

Intro to A. Medial: A Step by Step Guide Step 3: add the calculated amounts alcohol present and the total volume of the mixture What is the percentage (v/v) of alcohol in a mixture of: – 3000 m. L of 40% alcohol: (3000 x 0. 4)= 1200 m. L alcohol – 1000 m. L of 60% alcohol: (1000 x 0. 6)= 600 m. L alcohol – +1000 m. L of 70% alcohol: (1000 x 0. 7)= + 700 m. L alcohol 5000 m. L total 2500 m. L alcohol

Intro to A. Medial: A Step by Step Guide Step 4: find the percentage (v/v) of alcohol based on the calculations What is the percentage (v/v) of alcohol in a mixture of: – 3000 m. L of 40% alcohol: (3000 x 0. 4)= 1200 m. L alcohol – 1000 m. L of 60% alcohol: (1000 x 0. 6)= 600 m. L alcohol – +1000 m. L of 70% alcohol: (1000 x 0. 7)= + 700 m. L alcohol 5000 m. L total 2500 m. L alcohol 2500 x 100 = 50% alcohol (v/v) 5000
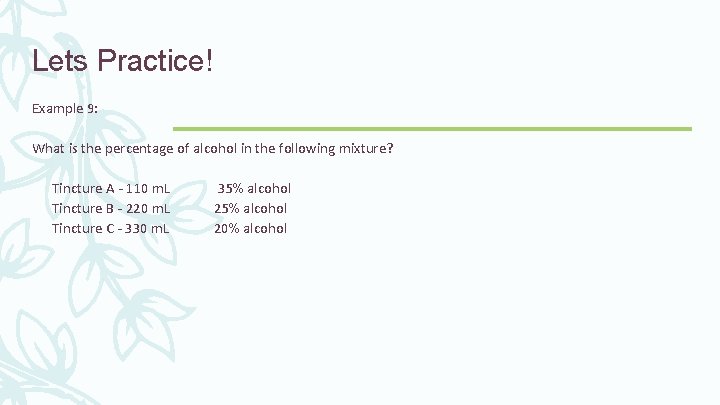
Lets Practice! Example 9: What is the percentage of alcohol in the following mixture? Tincture A - 110 m. L 35% alcohol Tincture B - 220 m. L 25% alcohol Tincture C - 330 m. L 20% alcohol

Lets Practice! Example 10: What is the percentage strength of alcohol in a mixture of 500 m. L of a solution containing 40% v/v alcohol, 400 m. L of a second solution containing 21% v/v alcohol, and a sufficient quantity of a nonalcoholic third solution to make a total of 1000 m. L?

Intro to A. Alternate: A Step by Step Guide How many milliliters of 2. 5 w/v chlorpromazine hydrochloride injection and how many milliliters of 0. 9% w/v Na. Cl injection should be used to prepare 500 m. L of a 0. 3% w/v chlorpromazine hydrochloride injection? This problem involves the use of alligation alternate

Intro to A. Alternate: A Step by Step Guide Step 1: separate what you have from what you want to get How many milliliters of 2. 5% w/v chlorpromazine hydrochloride injection and how many milliliters of 0. 9% w/v Na. Cl injection should be used to prepare 500 m. L of a 0. 3% w/v chlorpromazine hydrochloride injection? Have: To get: 2. 5% CPZ 0. 3% CPZ O. 9% Na. Cl
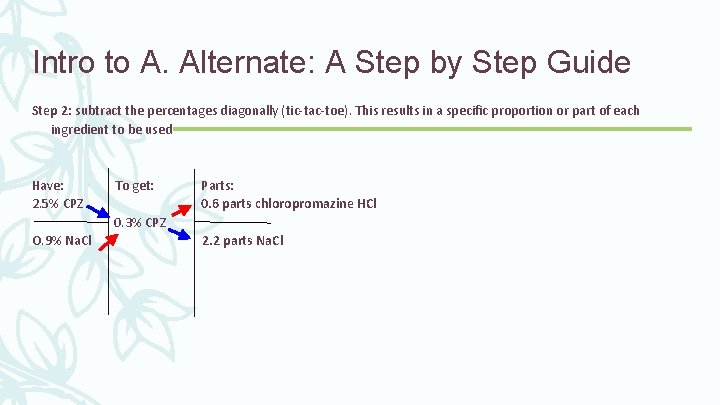
Intro to A. Alternate: A Step by Step Guide Step 2: subtract the percentages diagonally (tic-tac-toe). This results in a specific proportion or part of each ingredient to be used Have: To get: Parts: 2. 5% CPZ 0. 6 parts chloropromazine HCl 0. 3% CPZ O. 9% Na. Cl 2. 2 parts Na. Cl

Intro to A. Alternate: A Step by Step Guide Step 3: calculate the total parts. This is results in the total proportion of the mixture Have: To get: Parts: 2. 5% CPZ 0. 6 parts chloropromazine HCl 0. 3% CPZ O. 9% Na. Cl + 2. 2 parts Na. Cl 2. 8 total parts

Intro to A. Alternate: A Step by Step Guide Step 4: set up a proportion to find the quantity of each ingredient needed 0. 6 parts chloropromazine HCl 2. 2 parts Na. Cl 2. 8 Total Parts (500 m. L) 2. 2/2. 8 = x/500 m. L 2. 8 x =1100 x = 392. 857 m. L of sodium chloride injection 0. 6/2. 8 = x/500 m. L 2. 8 x = 300 x = 107. 143 m. L of chloropromazine HCL injection

Lets Practice! Example 11: In what proportion should 20% benzocaine ointment be mixed with an ointment base to produce a 2. 5% benzocaine ointment?

Lets Practice! Example 12: How many milliliters of each of two liquids with specific gravities of 0. 950 and 0. 875 should be used to prepare 1500 m. L of a liquid having a specific gravity of 0. 925?

Lets Practice! Example 13: Rx Castor Oil Euresol Alcohol 85% 5. 0 m. L ad 15. 0 m. L 240. 0 m. L How many milliliters each of 95% (v/v) alcohol and water should be used in preparing the prescription?

Lets Practice! Example 14: The formula for a buffer solution contains 1. 24% (w/v) of boric acid. How many milliliters of a 5% (w/v) boric acid solution should be used to obtain the boric acid needed in preparing 1 liter of the buffer solution?

Lets Practice! Example 15: You have in stock a 30 gram tube of 1% hydrocortisone cream and a 45 gram tube of 2. 5% hydrocortisone cream and you need to prepare a 30 gram tube of 2% hydrocortisone. How much of each tube do you use to create this new concentration?

Lets Practice! Example 16: You need a 200 ml stock solution of 10 mg/ml aminophylline to prepare several unit-dose oral syringes. You carry the standard 25 mg/ml aminophylline solution. How much water and standard aminophylline (25 mg/ml) do you combine to create this new solution?

Lets Practice! Example 17: How much water should you mix with 70% isopropyl alcohol to make 500 ml of 20% alcohol?

Intro to A. Alternate: A Step by Step Guide A hospital pharmacist wants to use three lots of ichthammol ointment containing, respectively, 50%, 20%, and 5% ichthammol. In what proportion should they be mixed to prepare a 10% ichthammol ointment?

Intro to A. Alternate: A Step by Step Guide Step 1: separate what you have from what you want to get A hospital pharmacist wants to use three lots of ichthammol ointment containing, respectively, 50%, 20%, and 5% ichthammol. In what proportion should they be mixed to prepare a 10% ichthammol ointment? Have: 50% To Get: 20% 10% 5%

Intro to A. Alternate: A Step by Step Guide Step 2: arrage the haves in such a way that your to get falls in between their ranges numerically. Have: 50% To Get: 10% 5% 20% 10% 5% It’s essentially combining two alligations in one
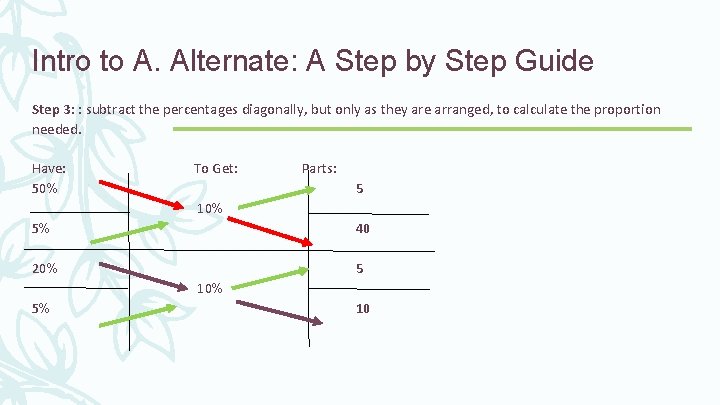
Intro to A. Alternate: A Step by Step Guide Step 3: : subtract the percentages diagonally, but only as they are arranged, to calculate the proportion needed. Have: 50% To Get: Parts: 5 10% 5% 40 20% 5 10% 5% 10

Intro to A. Alternate: A Step by Step Guide Step 4: combine the parts that share the same percentage Have: 50% Parts: 5 20% 5 5% 10 + 40 = 50 Ratio = 5: 5: 50 or 1: 1: 10
- Slides: 45