The allergy umbrella World Allergy Organ J 2013

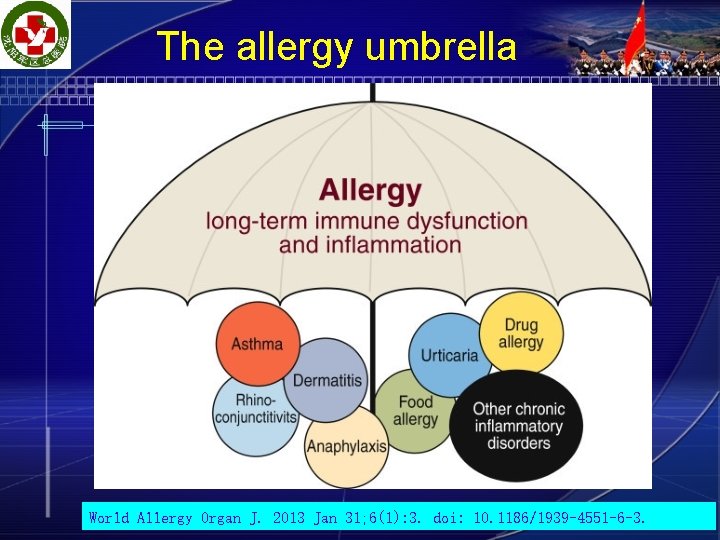
The allergy umbrella World Allergy Organ J. 2013 Jan 31; 6(1): 3. doi: 10. 1186/1939 -4551 -6 -3.

Self-amplification mechanisms of mast cell activation: a new look in allergy. • The current definition of allergy, a group of Ig. E mediated diseases appears difficult to cover allergic reactions • Since even Ig. E dependent allergic reactions are carried out through activation of mast cells and basophils, and allergens mentioned above can activate these cells • we hypothesize that allergic reactions are mast cell and basophil mediated inflammatory process as it is the activated mast cells and basophils that initiate the pathological process of the immediate allergic reactions • whereas Ig. E only serves as one of the activators of these cells. He S, Zhang H, Zeng X, Yang P. Curr Mol Med. 2012 Dec; 12(10): 1329 -39.




2013

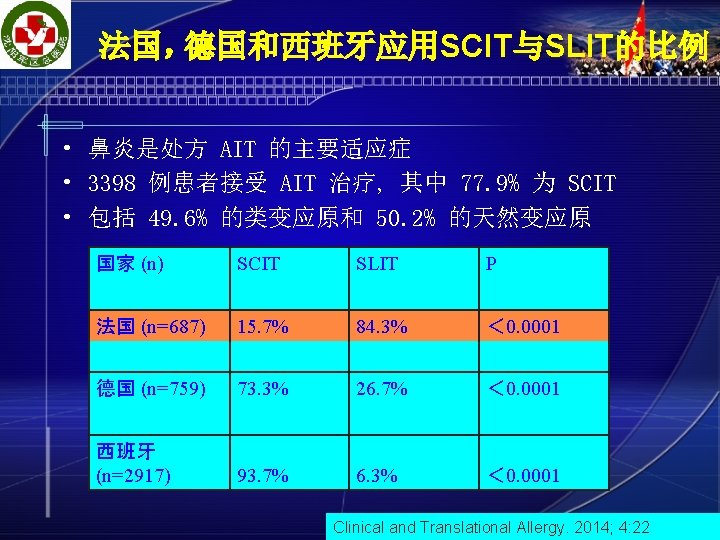
关于AIT安全性 Clinical and Translational Allergy. 2014; 4: 22


Risk factors • Risk factors for fatal and nonfatal reactions to subcutaneous immunotherapy: National surveillance study on allergen immunotherapy (2008 -2013) • 28. 9 million injection visits, including 344, 480 patients Ann Allergy Asthma Immunol. 2016 Mar 3. doi: 10. 1016/j. anai. 2016. 02. 001 [Epub ahead of print]

BACKGROUND and CONCLUSIONS • In 2008, an annual surveillance study of systemic reactions (SRs) from subcutaneous immunotherapy (SCIT) injections was initiated in North America. • CONCLUSIONS: – SCIT-related fatality rates may be decreasing – Vigilance regarding modifiable risk factors • Careful patient selection • Dose adjustment during pollen seasons for highly sensitive patients • Potential risk for SRs from off-label SLIT exists Ann Allergy Asthma Immunol. 2016 Mar 3.

RESULTS • The rate of SRs from SCIT remained stable, occurring in 1. 9% of patients, with 0. 08% and 0. 02% experiencing grade 3 and 4 SRs. • SRs occurred in 1. 4% of patients receiving off-label SLIT, including 0. 03% with grade 3 SRs. • There were no SLIT-related grade 4 SRs or fatalities. • Practices that never administered SCIT in patients with uncontrolled asthma (Asthma Control Test score <20) had significantly fewer grade 3 and 4 SRs • Lowering doses during pollen seasons for patients with highly positive skin tests reduced SRs of all severity grades (P <. 05). Ann Allergy Asthma Immunol. 2016 Mar 3.

The relationship between autoimmunity and specific immunotherapy for allergic diseases. • • The aim of this study was to perform a 20 -year post-specific immunotherapy (SIT) observational evaluation for an assessment of any manifestations of autoimmune disease or the appearance of autoantibodies in serum. MATERIALS AND METHODS: – In total, 1, 888 patients (902 women and 986 men) were observed. The mean age of the patients was 34. 1 ± 12. 4 years at the start of the prospective observation after finishing SIT. – New incidences of autoimmune disease and/or the presence of autoantibodies in serum were monitored. – The SIT group was compared with control groups consisting of allergic patients who had very received SIT and with non-allergic subjects. Hum Vaccin Immunother. 2015 Dec 2; 11(12): 2764 -8

The relationship between autoimmunity and specific immunotherapy for allergic diseases. • RESULTS: – There were no significant differences in the autoimmune disease prevalence between the allergic patients with or without SIT. – However, significantly higher prevalence of four different autoimmune diseases (AID) were observed in the non-allergic patients during the same period. – Additionally, the incidence of 8 different autoantibodies was significantly higher in non-allergic patients than in control subjects. – Hashimoto disease was the most common autoimmune disease observed. • CONCLUSION: – The results of this long-term observational study indicated a lack of a significant prevalence of new instances of autoimmune disease during 20 years of observation post-SIT and at a rate lower than that of non-allergic control subjects – suggesting that SIT is safe in this regard in the long term. Hum Vaccin Immunother. 2015 Dec 2; 11(12): 2764 -8



成本和效果经济学 • 成本效果分析(cost effectiveness analysis, CEA) • 成本效用分析(cost utility analysis , CUA) • 质量调整生命年(quality adjusted life years, QALY) A systematic review and economic evaluation of subcutaneous and sublingual allergen immunotherapy in adults and children with seasonal allergic rhinitis. Health Technol Assess. 2013 Jul; 17(27): 1 -322

RESULTS • Economic modelling suggested that, when compared with symptomatic treatment (ST) • Both SCIT and SLIT may become cost-effective at a threshold of £ 20, 000 -30, 000 per quality-adjusted life-year (QALY) from around 6 years, or 5 years for SCIT compared with SLIT Health Technol Assess. 2013 Jul; 17(27): 1 -322

Allergy immunotherapy for allergic rhinitis effectively prevents asthma: Results from a large retrospective cohort study. • • Identified a consecutive cohort of 118, 754 patients with AR but without asthma who had not received AIT in 2005 In a total of 2431 (2. 0%) patients, AIT was started in 2006. Asthma was newly diagnosed from 2007 -2012 in 1646 (1. 4%) patients. The risk of incident asthma was significantly lower in patients exposed to AIT (RR, 0. 60; 95% CI, 0. 42 -0. 84) compared with patients receiving no AIT in 2006. Sensitivity analyses suggested significant preventive effects of subcutaneous immunotherapy (RR, 0. 54; 95% CI, 0. 38 -0. 84) and AIT including native (nonallergoid) allergens (RR, 0. 22; 95% CI, 0. 02 -0. 68). AIT for 3 or more years tended to have stronger preventive effects than AIT for less than 3 years. AIT effectively prevents asthma in patients with AR in a real-world setting. J Allergy Clin Immunol. 2015 Dec; 136(6): 1511 -6



International consensus on allergy immunotherapy • • Allergen immunotherapy (AIT) has been used to treat allergic disease since the early 1900 s. Despite numerous clinical trials and meta-analyses proving AIT efficacious, it remains underused and is estimated to be used in less than 10% of patients with allergic rhinitis or asthma worldwide. In addition, there are large differences between regions, which are not only due to socioeconomic status. There is practically no controversy about the use of AIT in the treatment of allergic rhinitis and allergic asthma, but for atopic dermatitis or food allergy, the indications for AIT are not well defined. The elaboration of a wider consensus is of utmost importance because AIT is the only treatment that can change the course of allergic disease by preventing the development of asthma and new allergen sensitizations and by inducing allergen-specific immune tolerance. Safer and more effective AIT strategies are being continuously developed both through elaboration of new allergen preparations and adjuvants and alternate routes of administration. J Allergy Clin Immunol. 2015 Sep; 136(3): 556 -68.






关于asthma AIT的途径 • SCIT or SLIT ? • AIT新途径-ILIT • 其它

Selection of patients for sublingual versus subcutaneous immunotherapy • Subjects – have a clear medical history of allergic disease, with exacerbation of symptoms on exposure to one or more allergens and a corresponding positive skin or in vitro test • The route of administration should be based on – published evidence of clinical and immunologic efficacy (which varies per allergic disease and per allergen) – mono- or multi-allergen immunotherapy • SLIT multi-allergen immunotherapy was not effective – safety • adverse events with SLIT are more frequent, but less severe – costs and patient preferences, closely related to adherence issues Immunotherapy. 2014; 6(7): 871 -84.

Selection of patients for sublingual immunotherapy versus subcutaneous immunotherapy • Both SLIT and SCIT – shown to be beneficial for therapy of seasonal allergic rhinoconjunctivitis. – Each therapeutic measure has its associated benefits • SLIT – A better safety profile with less systemic reactions and to date, no reported fatal reactions • SCIT – A slightly better efficacy profile and readily allows for treatment of polyallergic patients Allergy Asthma Proc. 2015 Mar-Apr; 36(2): 100 -4.

Adherence to Sublingual Immunotherapy • The recent data from manufacturers revealed that only 13 % of patients treated with SLIT reach the recommended 3 -year duration. • The success of technology-based tools, including online platforms, social media, e-mail, and a short message service by phone, is currently considered to improve the adherence • Complete at least a 3 -year course of treatment. Curr Allergy Asthma Rep. 2016 Jan; 16(2): 12

Sublingual Immunotherapy for Asthmatic Children Sensitized to House Dust Mite: A Meta-Analysis. • • There is good evidence that house dust mite subcutaneous immunotherapy is efficacious and has long-term benefit in children Medical Literature Analysis and Retrieval System Online, databases until February 2014 were searched. We found that – SLIT significantly decreased asthma symptom score (P = 0. 007) and increased s. Ig. G 4 levels (P = 0. 011) greater than control in children (<18 years of age) with asthma. – There was no difference between SLIT and control groups in specific D pteronyssinus Ig. E levels (P = 0. 076) and medication score (P = 0. 408). – The safety profile was similar between groups. Our findings are not enough to support the use of dust mite SLIT in children with asthma. Medicine (Baltimore). 2015 Jun; 94(24): e 701.

The Future of Sublingual Immunotherapy in the United States • • FDA in the USA has approved three SLIT tablets for the treatment of (AR) and allergic rhinoconjunctivitis (ARC) in relation to pollen Although these approvals provide support for physicians to prescribe SLIT, barriers to prescribing SLIT still remain such as FDA approval for additional formulations, a standard dose and dosing schedule, and cost/insurance coverage. In order to further support the use of SLIT, research is currently being conducted to expand the indication for SLIT to other common comorbidities to AR/ARC. For example, allergic asthma, food allergies, and atopic dermatitis are other diseases which are being explored. The future of SLIT in the USA is unknown; however, education will be necessary for both providers and patients Curr Allergy Asthma Rep. 2015 Aug; 15(8): 44.

Patient selection for subcutaneous versus sublingual immunotherapy. • RECENT FINDINGS – Enhance AIT simplicity • by changing the route to home-administrated SL-AIT, • by shortening the subcutaneous-allergen-specific immunotherapy (SC-AIT) build-up or maintenance phase. The latter was safe with several hypoallergenic extracts. – As for SL-AIT • double blind placebo-controlled large trials in patients with allergic rhinitis and asthma have shown the efficacy and safety of ragweed pollen and house dust mite • A large trial with SLIT in 3 -year-old children was effective. • SUMMARY: – With the improvement of SL-AIT efficacy, the selection of SC-versus-SL-AIT will probably increasingly be based not on efficacy, but on practical aspects, without losing sight of which SL-AIT products have proven efficacy Curr Opin Allergy Clin Immunol. 2015 Dec; 15(6): 588 -595.

New developments in AIT • • • SCIT: – remains a disease modifying treatment for allergic rhinoconjunctivitis and asthma with rare complications of therapy. – may be effective in select cases of atopic dermatitis, particularly for patients with dust mite sensitivity. SLIT tablets : – are now commercially available for grass and ragweed allergy and appear to have a superior safety profile to SCIT with similar long-term effectiveness – are administered daily at home (after initial supervised dosing) and may be used shortly before and during the target pollen seasons in a precoseasonal fashion (instead of perennial dosing). OIT: – Research continues into experimental approaches using oral food allergen immunotherapy (OIT) to modify the natural history of food allergies – Research continues into novel food allergy treatments. Curr Opin Pediatr. 2015 Oct; 27(5): 649 -55.

House dust mite sublingual immunotherapy is safe in patients with mild-to-moderate, persistent asthma: a clinical trial • • METHODS: Asthmatic adults were randomized 2: 1 to twelve months of daily treatment with a sublingual solution of Dermatophagoides pteronyssinus and Dermatophagoides farinae extracts or a placebo. RESULTS: Overall, the patients in the safety analysis set (n=484; active treatment: n=322; placebo: n=162) had mostly well-controlled, persistent asthma (mild in 290 patients (59. 9%), moderate in 183 (37. 8%) and severe in 11 (2. 3%)). – No treatment-related serious AEs were reported. – Respectively 87. 0% and 75. 9% of the patients in the active and placebo groups experienced at least one AE (mostly mild), and 78. 9% and 48. 1% experienced an ADR (mostly mild or moderate oral reactions). – The incidence of asthma exacerbations (symptoms requiring a short-course of oral corticosteroids) during the study was similar in the active treatment group (3. 7%) and the placebo group (4. 3%). – There were no significant intergroup differences or intragroup changes over time in respiratory AEs, lung function or asthma-related quality of life. • CONCLUSIONS: HDM sublingual AIT was safe and well tolerated in adult patients with mild-to-moderate, persistent asthma Allergy. 2016 Feb; 71(2): 249 -57.

Update on immunotherapy for the treatment of asthma • SUMMARY: For AIT – it will become increasingly important to use products designed properly, standardized and with a well documented effectiveness in clinical studies – Overcome the disputes of subcutaneous immunotherapy versus sublingual immunotherapy – Arrive at the concept of personalized medicine regarding AIT, framing in different phenotypes of asthma patients to use the optimal preparation for each particular patient Curr Opin Pulm Med. 2016 Jan; 22(1): 18 -24.

关于asthma AIT的途径 • SCIT or SLIT ? • AIT新途径-ILIT • 其它
ILIT:intralymphatic immunotherapy Juillard, GJF and Bubbers, JE, "Experimental Intralymphatic Immunotherapy (ILI) of Canine Allergic Disease". Federation Proceedings, Vol. 42, No. 3, March 1, 1983.

ILIT-intralymphatic immunotherapy • Intralymphatic allergen-specific immunotherapy: an effective and safe alternative treatment route for pollen-induced allergic rhinitis – J Allergy Clin Immunol. 2013 Feb; 131(2): 412 -20 • Is intralymphatic immunotherapy ready for clinical use in patients with grass pollen allergy? – J Allergy Clin Immunol. 2013 Nov; 132(5): 12481252. e 5.

ILIT-intralymphatic immunotherapy • Intralymphatic immunotherapy. – World Allergy Organ J. 2015 Mar 7; 8(1): 9. • Intralymphatic immunotherapy of pollen-induced rhinoconjunctivitis: a double-blind placebo-controlled trial. – Respir Res. 2016 Jan 27; 17(1): 10 • Three-injection intralymphatic immunotherapy in adolescents and young adults with grass pollen rhinoconjunctivitis. – Ann Allergy Asthma Immunol. 2016 Feb; 116(2): 168 -70.

关于asthma AIT的途径 • SCIT or SLIT ? • AIT新途径-ILIT • 其它



关于 asthma AIT 治疗进展 omalizumab and AIT Toll-like receptors
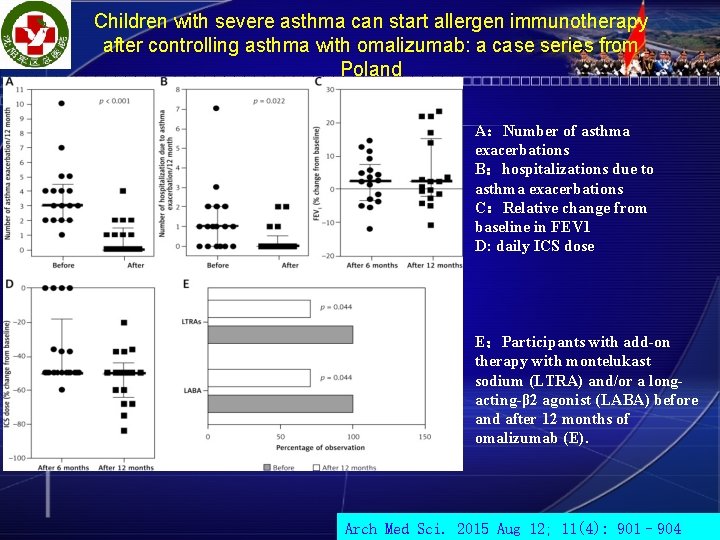
Children with severe asthma can start allergen immunotherapy after controlling asthma with omalizumab: a case series from Poland A:Number of asthma exacerbations B:hospitalizations due to asthma exacerbations C:Relative change from baseline in FEV 1 D: daily ICS dose E:Participants with add-on therapy with montelukast sodium (LTRA) and/or a longacting-β 2 agonist (LABA) before and after 12 months of omalizumab (E). Arch Med Sci. 2015 Aug 12; 11(4): 901– 904

Result • • All 17 patients who received omalizumab for at least 52 weeks were included in the analysis. All patients tolerated omalizumab well. After 12 months of therapy, all patients were treated with lower ICS doses; the median ICS dose reduction was – 50% (quartile range: from – 50% to – 60%), and the lowest reduction was 20%. In 5 patients, LABA or LTRA therapy was discontinued after 12 months. During the study, SIT was started in 12 (70. 6%) patients – in 4 (33%) patients after 3 months, – in 5 (42%) patients after 6 months, – in 3 (25%) patients after 9 months of omalizumab treatment – – and was successfully continued according to the SIT scheme. – In 5 children who had not tolerated immunotherapy before omalizumab treatment, SIT was introduced after 6 months of treatment and has been successfully continued. – Logistic regression analysis did not show any independent effect of SIT on the ICS dose reduction or the number of exacerbations; – this subgroup analysis did not have sufficient power. Arch Med Sci. 2015 Aug 12; 11(4): 901– 904

CONCLUSIONS • This real-life study showed that pretreatment with omalizumab should be specifically considered in children with severe allergic asthma to prevent asthma deterioration/exacerbation. • This approach allows for the safe and effective initiation of SIT and changes the natural course of severe allergic asthma in children. Children with severe asthma can start allergen immunotherapy after controlling asthma with omalizumab Arch Med Sci. 2015 Aug 12; 11(4): 901– 904

关于 asthma AIT 治疗进展 omalizumab and AIT Toll-like receptors

Toll-like receptors as targets for AIT • • Toll-like receptors (TLRs) are novel and promising targets for AIT TLR agonists reduce Th 2 responses and ameliorate airway hyper-responsiveness Curr Opin Allergy Clin Immunol. 2015 Dec; 15(6): 568 -74.

Toll-like receptors as targets for AIT • RECENT FINDINGS: – To date, 2 allergy vaccine-containing TLR agonists have been investigated in clinical trials, The former contains monophosphoryl lipid, a TLR 4 agonist and the latter contains, Cp. G motifs activating the TLR 9 cascade. – Preseasonal subcutaneous injection of both of these allergy vaccines has been safe and efficacious in control of nasal symptoms of patients with allergic rhinitis. – CRX-675 (a TLR 4 agonist), AZD 8848 (a TLR 7 agonist), VTX-1463 (a TLR 8 agonist) and 1018 ISS and Qb. G 10 (TLR 9 agonists) are currently in clinical development for allergic rhinitis and asthma. • SUMMARY: – TLR agonists herald promising results for allergen immunotherapy of patients with allergic rhinitis and asthma. – Future research should be directed at utilizing these agents for immunotherapy of food allergy as well. Curr Opin Allergy Clin Immunol. 2015 Dec; 15(6): 568 -574.


Allergen immunotherapy: an update on protocols of administration • The efficacy of only preseasonal subcutaneous AIT was demonstrated for tree and grass pollen. • Sublingual AIT was shown to be effective and well tolerated in allergic rhinitis and asthma with tablets and with highly concentrated liquid formulations (ragweed, house dust mite), but not cockroach. • For food allergy, oral immunotherapy is promising, but close attention should be paid to the exact administration schedule, maintenance dose, and the definition of efficacy • The practicing physician should be watchful for advances in the field of aeroallergen AIT and food oral immunotherapy, analyzing the presented information in detail and interpreting conclusions product specifically, without generalizing. Curr Opin Allergy Clin Immunol. 2015,15(6): 556 -567.


Allergy-asthma (OAT) 最佳过敏症治疗方案 (OAT) Management



- Slides: 60