The Addition of Evolocumab AMG 145 Allows the

The Addition of Evolocumab (AMG 145) Allows the Majority of Heterozygous Familial Hypercholesterolemic Patients to Achieve Low-density Lipoprotein Cholesterol Goals Results from the Phase 3 Randomized, Double-blind, Placebo-controlled Study Frederick Raal, 1 Robert Dufour, 2 Traci Turner, 3 Fernando Civeira, 4 Lesley Burgess, 5 Gisle Langslet, 6 Russell Scott, 7 Anders G. Olsson, 8 David Sullivan, 9 Gerard K. Hovingh, 10 Bertrand Cariou, 11 Ioanna Gouni-Berthold, 12 Ransi Somaratne, 13 Ian Bridges, 14 Rob Scott, 13 Scott M. Wasserman, 13 and Daniel Gaudet 15 for the RUTHERFORD-2 Investigators 1 Carbohydrate & Lipid Metabolism Research Unit, University of Witwatersrand, Johannesburg, South Africa; 2 Institut de Recherches Cliniques de Montreal, Universite de Montreal, Quebec, Canada; 3 Metabolic and Atherosclerosis Research Center, Cincinnati, OH, USA; 4 Hospital Universitario Miguel Servet, Zaragoza, Spain; 5 TREAD Research, Cardiology Unit, Department of Internal Medicine, University of Stellenbosch, Parow, South Africa; 6 Lipid Clinic, Oslo University Hospital, Oslo, Norway; 7 Lipid and Diabetes Research Group, Christchurch, New Zealand; 8 Linkoping University and Stockholm Heart Center, Stockholm, Sweden; 9 Department of Clinical Biochemistry, Royal Prince Alfred Hospital, Camperdown, Australia; 10 Academisch Medisch Centrum, Vascular Medicine, Amsterdam, The Netherlands; 11 Institut du Thorax, Nantes University Hospital, Nantes, France; 12 Center for Endocrinology, Diabetes and Preventive Medicine, University of Cologne, Germany; 13 Amgen Inc. , Thousand Oaks, CA, USA; 14 Amgen Ltd, Uxbridge, United Kingdom; 15 ECOGENE-21, Dyslipidemia, Diabetes and Atherosclerosis Research Group, Department of Medicine, Université de Montréal, Chicoutimi, Québec, Canada March 29, 2014, Featured Clinical Research Session 400 American College of Cardiology, Washington DC

Background: Heterozygous Familial Hypercholesterolemia (He. FH) Ø He. FH is characterized by markedly elevated low-density lipoprotein cholesterol (LDL-C), and if untreated, is associated with significant premature cardiovascular morbidity and mortality. 1 Ø He. FH is most commonly caused by loss-of-function mutations in the LDLR gene. 2 Ø Although current treatments (e. g. , statins +/- ezetimibe, bile acid sequestrants and/or niacin) can produce reductions in LDL-C of 50% – 65%, many He. FH patients are still unable to achieve recommended LDL-C targets. 3 1. Eur Heart J 2013; 34: 3478 -90 2. Ann Human Genet. 2008; 72: 485 -98. 3. J Clin Lipidol. 2007; 1: 280 -6. 2

The RUTHERFORD-2 Study Ø Reduction of LDL-C with PCSK 9 Inhibition in Heterozygous Familial Hypercholesterolemia Disorder (NCT 20110117) Ø Design: A 12 -week, randomized, double-blind, placebo-controlled, multicenter phase 3 study Ø Objective: To evaluate the efficacy and safety of evolocumab (AMG 145) 140 mg Q 2 W and 420 mg QM administered subcutaneously in a large cohort of He. FH patients unable to achieve an LDL-C < 100 mg/d. L despite statin therapy with or without ezetimibe 3
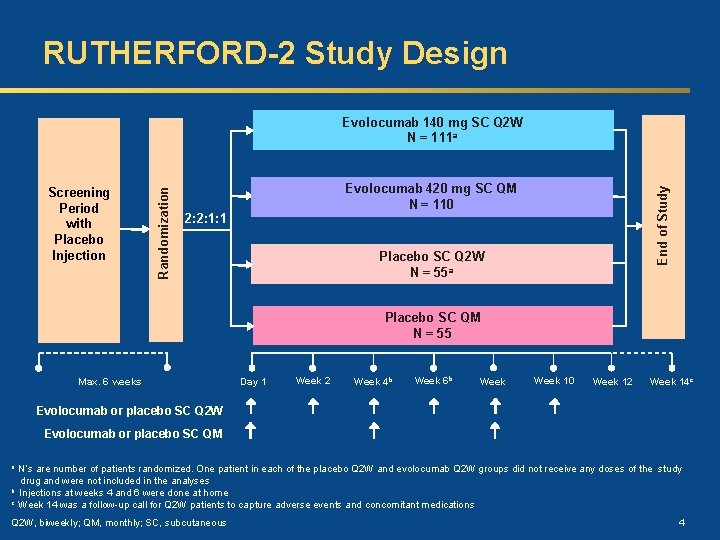
RUTHERFORD-2 Study Design Evolocumab 420 mg SC QM N = 110 2: 2: 1: 1 End of Study Screening Period with Placebo Injection Randomization Evolocumab 140 mg SC Q 2 W N = 111 a Placebo SC Q 2 W N = 55 a Placebo SC QM N = 55 Max. 6 weeks Day 1 Week 2 Week 4 b Week 6 b Week 10 Week 12 Week 14 c Evolocumab or placebo SC Q 2 W Evolocumab or placebo SC QM a b c N’s are number of patients randomized. One patient in each of the placebo Q 2 W and evolocumab Q 2 W groups did not receive any doses of the study drug and were not included in the analyses Injections at weeks 4 and 6 were done at home Week 14 was a follow-up call for Q 2 W patients to capture adverse events and concomitant medications Q 2 W, biweekly; QM, monthly; SC, subcutaneous 4

RUTHERFORD-2: Baseline Characteristics Placebo Q 2 W (N = 54) Evolocumab 140 mg Q 2 W (N = 110) Placebo QM (N = 55) Evolocumab 420 mg QM (N = 110) 51 (14) 53 (12) 47 (12) 52 (12) Female, % 46 40 44 42 Race: white, % 93 90 89 89 431 (124) 458 (145) 441 (146) 436 (139) Definite 83 77 78 76 Probable 17 23 22 24 Statin use, % 100 100 Ezetimibe use, % 61 61 66 62 Characteristic Age (years), mean (SD) PCSK 9 (ng/m. L), mean (SD) He. FH classificationa, % Based on Simon Broome criteria He. FH, heterozygous familial hypercholesterolemia; Q 2 W, biweekly; QM, monthly; SD, standard deviation a 5
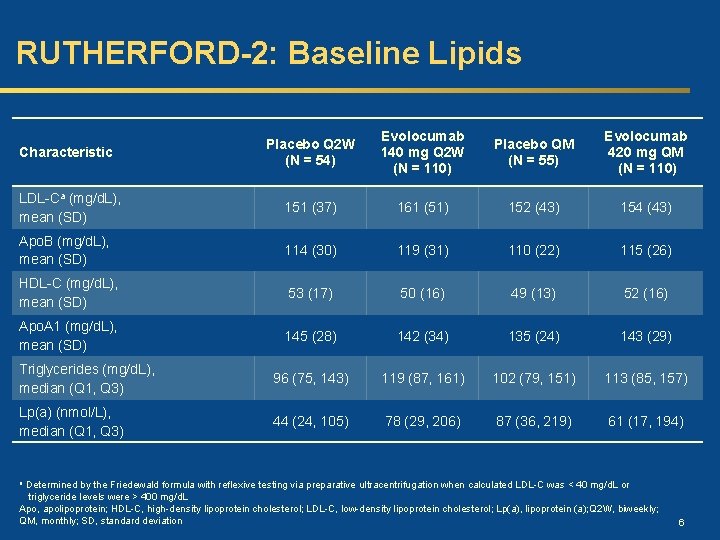
RUTHERFORD-2: Baseline Lipids Characteristic Placebo Q 2 W (N = 54) Evolocumab 140 mg Q 2 W (N = 110) Placebo QM (N = 55) Evolocumab 420 mg QM (N = 110) LDL-Ca (mg/d. L), mean (SD) 151 (37) 161 (51) 152 (43) 154 (43) Apo. B (mg/d. L), mean (SD) 114 (30) 119 (31) 110 (22) 115 (26) HDL-C (mg/d. L), mean (SD) 53 (17) 50 (16) 49 (13) 52 (16) Apo. A 1 (mg/d. L), mean (SD) 145 (28) 142 (34) 135 (24) 143 (29) Triglycerides (mg/d. L), median (Q 1, Q 3) 96 (75, 143) 119 (87, 161) 102 (79, 151) 113 (85, 157) Lp(a) (nmol/L), median (Q 1, Q 3) 44 (24, 105) 78 (29, 206) 87 (36, 219) 61 (17, 194) Determined by the Friedewald formula with reflexive testing via preparative ultracentrifugation when calculated LDL-C was < 40 mg/d. L or triglyceride levels were > 400 mg/d. L Apo, apolipoprotein; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; Lp(a), lipoprotein (a); Q 2 W, biweekly; QM, monthly; SD, standard deviation a 6
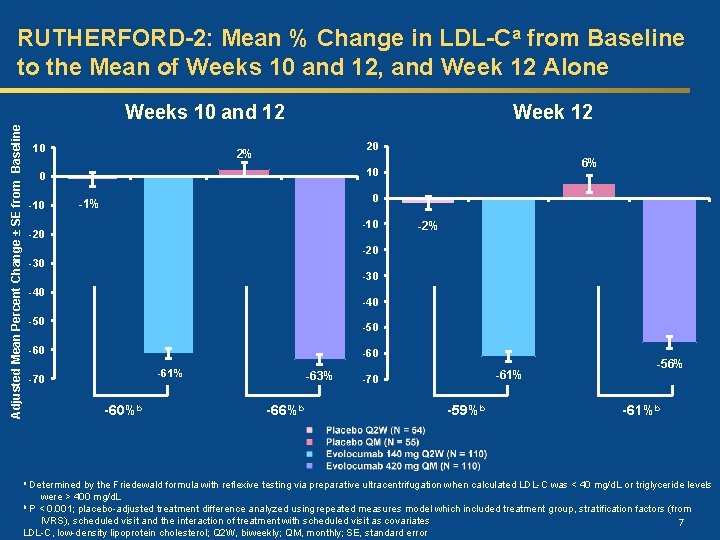
RUTHERFORD-2: Mean % Change in LDL-Ca from Baseline to the Mean of Weeks 10 and 12, and Week 12 Alone Adjusted Mean Percent Change ± SE from Baseline Weeks 10 and 12 10 Week 12 20 2% 0 -10 6% 10 0 -1% -10 -2% -20 -30 -40 -50 -60 -61% -70 -60%b -63% -66%b -61% -70 -59%b -56% -61%b Determined by the Friedewald formula with reflexive testing via preparative ultracentrifugation when calculated LDL-C was < 40 mg/d. L or triglyceride levels were > 400 mg/d. L b P < 0. 001; placebo-adjusted treatment difference analyzed using repeated measures model which included treatment group, stratification factors (from IVRS), scheduled visit and the interaction of treatment with scheduled visit as covariates 7 LDL-C, low-density lipoprotein cholesterol; Q 2 W, biweekly; QM, monthly; SE, standard error a
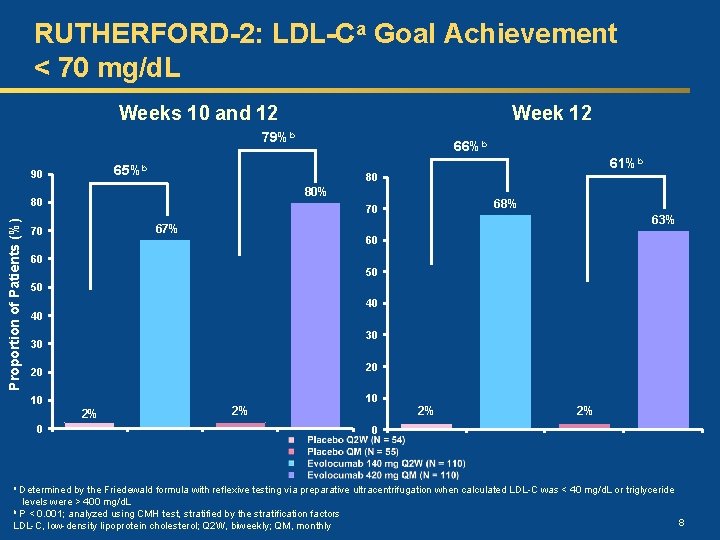
RUTHERFORD-2: LDL-Ca Goal Achievement < 70 mg/d. L Weeks 10 and 12 Week 12 79%b 65%b 90 61%b 80 80% 80 Proportion of Patients (%) 66%b 68% 70 67% 70 63% 60 60 50 50 40 40 30 30 20 20 10 2% 2% 0 Determined by the Friedewald formula with reflexive testing via preparative ultracentrifugation when calculated LDL-C was < 40 mg/d. L or triglyceride levels were > 400 mg/d. L b P < 0. 001; analyzed using CMH test, stratified by the stratification factors 8 LDL-C, low-density lipoprotein cholesterol; Q 2 W, biweekly; QM, monthly a
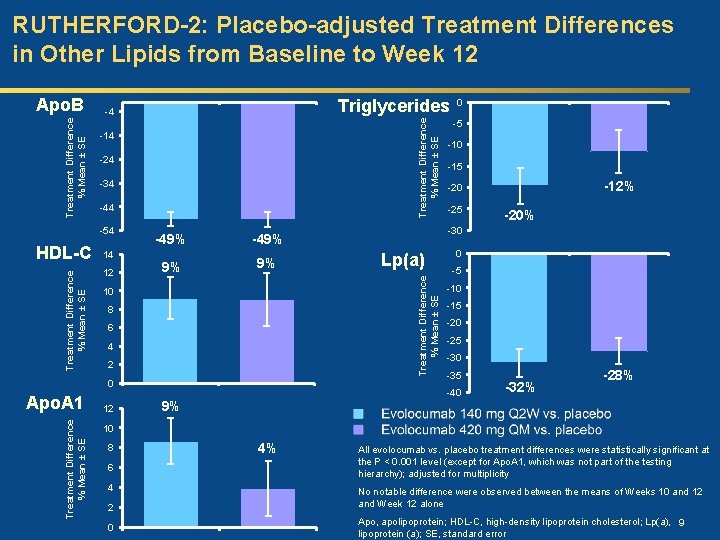
RUTHERFORD-2: Placebo-adjusted Treatment Differences in Other Lipids from Baseline to Week 12 Treatment Difference % Mean ± SE -14 -24 -34 -44 -54 HDL-C Triglycerides -4 14 12 -49% 9% 9% 10 8 6 4 2 0 Treatment Difference % Mean ± SE Apo. A 1 12 0 -5 -10 -15 -12% -20 -25 -20% -30 Lp(a) Treatment Difference % Mean ± SE Apo. B 0 -5 -10 -15 -20 -25 -30 -35 -40 -32% -28% 9% 10 8 6 4 2 0 4% All evolocumab vs. placebo treatment differences were statistically significant at the P < 0. 001 level (except for Apo. A 1, which was not part of the testing hierarchy); adjusted for multiplicity No notable difference were observed between the means of Weeks 10 and 12 and Week 12 alone Apo, apolipoprotein; HDL-C, high-density lipoprotein cholesterol; Lp(a), 9 lipoprotein (a); SE, standard error
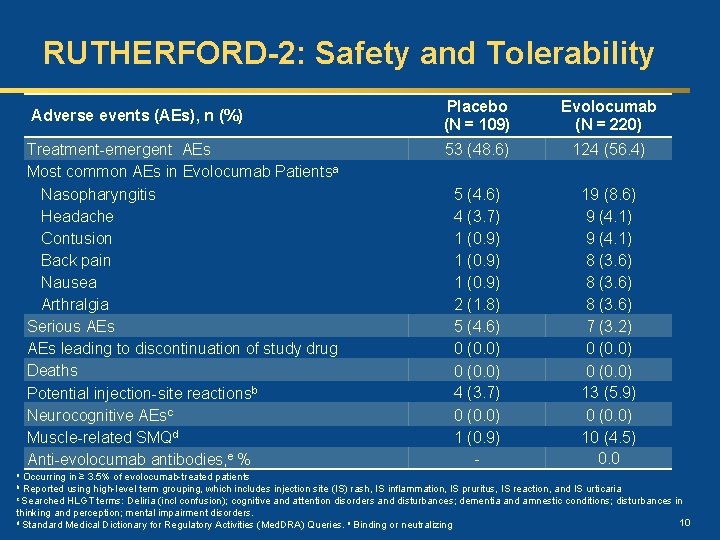
RUTHERFORD-2: Safety and Tolerability Adverse events (AEs), n (%) Treatment-emergent AEs Most common AEs in Evolocumab Patientsa Nasopharyngitis Headache Contusion Back pain Nausea Arthralgia Serious AEs leading to discontinuation of study drug Deaths Potential injection-site reactionsb Neurocognitive AEsc Muscle-related SMQd Anti-evolocumab antibodies, e % Placebo (N = 109) Evolocumab (N = 220) 53 (48. 6) 124 (56. 4) 5 (4. 6) 4 (3. 7) 1 (0. 9) 2 (1. 8) 5 (4. 6) 0 (0. 0) 4 (3. 7) 0 (0. 0) 1 (0. 9) - 19 (8. 6) 9 (4. 1) 8 (3. 6) 7 (3. 2) 0 (0. 0) 13 (5. 9) 0 (0. 0) 10 (4. 5) 0. 0 Occurring in ≥ 3. 5% of evolocumab-treated patients Reported using high-level term grouping, which includes injection site (IS) rash, IS inflammation, IS pruritus, IS reaction, and IS urticaria c Searched HLGT terms: Deliria (incl confusion); cognitive and attention disorders and disturbances; dementia and amnestic conditions; disturbances in thinking and perception; mental impairment disorders. d Standard Medical Dictionary for Regulatory Activities (Med. DRA) Queries. e Binding or neutralizing 10 a b
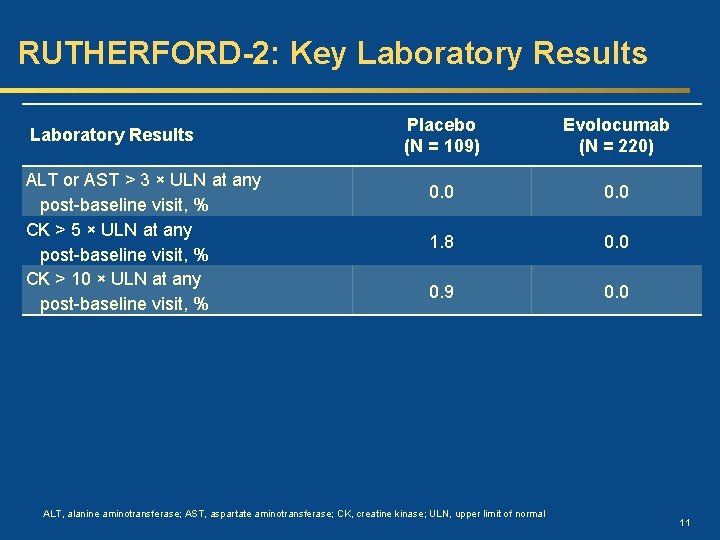
RUTHERFORD-2: Key Laboratory Results ALT or AST > 3 × ULN at any post-baseline visit, % CK > 5 × ULN at any post-baseline visit, % CK > 10 × ULN at any post-baseline visit, % Placebo (N = 109) Evolocumab (N = 220) 0. 0 1. 8 0. 0 0. 9 0. 0 ALT, alanine aminotransferase; AST, aspartate aminotransferase; CK, creatine kinase; ULN, upper limit of normal 11

RUTHERFORD-2: Conclusions Ø Evolocumab administered either biweekly or monthly yielded significant reductions in LDL-C in He. FH patients on statins with or without ezetimibe. § The mean reduction of LDL-C at Week 12 was 61% in the 140 mg Q 2 W and 56% in the 420 mg QM evolocumab dose groups, respectively. § The mean reduction of LDL-C at the mean of Weeks 10 and 12 was 61% in the 140 mg Q 2 W and 63% in the 420 mg QM evolocumab dose groups, respectively. Ø Evolocumab 140 mg biweekly and 420 mg monthly dosing regimens were clinically equivalent. Ø The majority of patients achieved LDL-C targets. Ø Evolocumab treatment resulted in favorable changes in other 12 lipoproteins.

RUTHERFORD-2: Conclusions Ø Evolocumab was well tolerated, with no notable difference in the AE profile compared with placebo. § The rate of nasopharyngitis and muscle-related adverse events (AEs) was higher in the evolocumab group. • The imbalance in the overall set of muscle-related AEs was not due to significant imbalances in any individual muscle-related event (i. e. , creatine kinase). Ø Evolocumab may offer a new and effective treatment option to further reduce LDL-C in He. FH patients. 13
- Slides: 13