The ABGs or Alpha Beta Gamma of Radioactivity

The ABG's (or Alpha, Beta, Gamma) of Radioactivity

Ch. 6 Notes Day 1

Objectives • SWBAT state what radioactivity is, where these rays come from, what each ray is made of and state why they are dangerous. • SWBAT identify 4 pioneer scientists who made important contributions to understanding radioactivity • SWABT to explain the meaning of “half-life”. • Student will be asked to find any sources of Radioactivity in his/her environment Science Park HS -- Honors Chemistry

Early Pioneers in Radioactivity Rutherford: Roentgen: Discoverer Alpha and Beta rays 1897 Discoverer of X-rays 1895 The Curies: Discoverers of Radium and Polonium 19001908 Becquerel: Discoverer of Radioactivity 1896

What do we mean by Radioactivity? Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. There are numerous types of radioactive decay. The general idea: An unstable nucleus releases energy to become more stable

Kinds of Radioactivity The three main decays are Alpha, Beta and Gamma

Radiation • Radiation comes from the nucleus of an atom. • Unstable nucleus emits a particle or energy alpha beta gamma 7
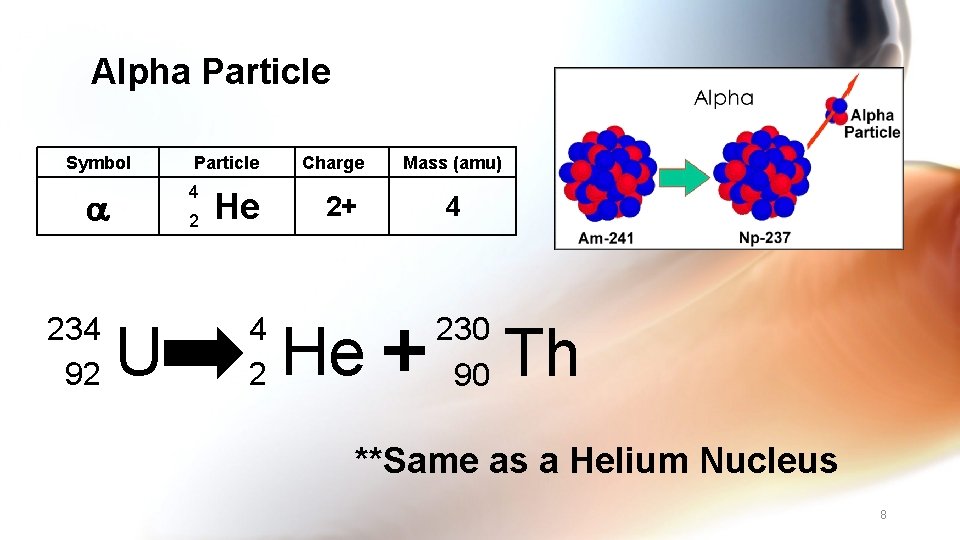
Alpha Particle Symbol 4 234 92 Particle 2 U He 4 2 Charge 2+ He Mass (amu) 4 230 90 Th **Same as a Helium Nucleus 8
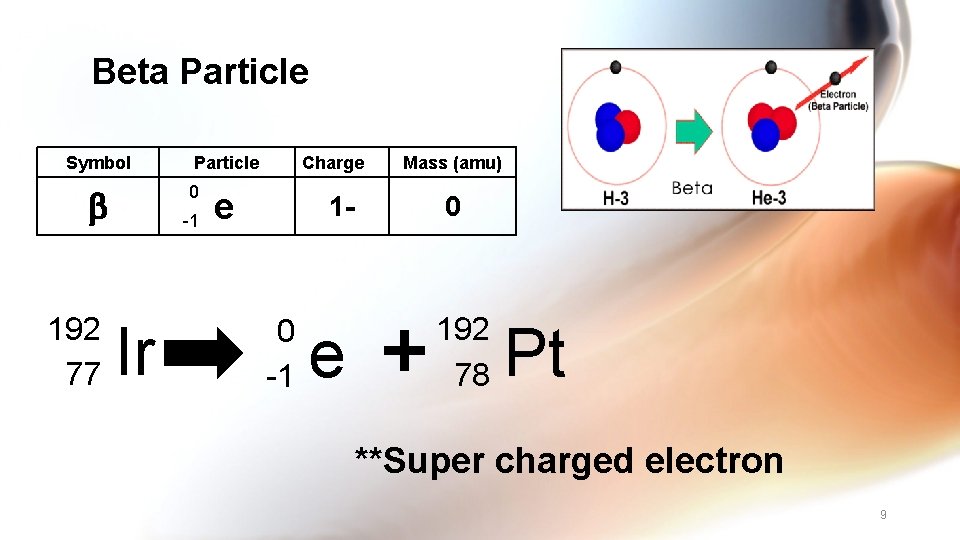
Beta Particle Symbol 0 192 77 Particle -1 Ir Charge e 1 - 0 -1 e Mass (amu) 0 192 78 Pt **Super charged electron 9
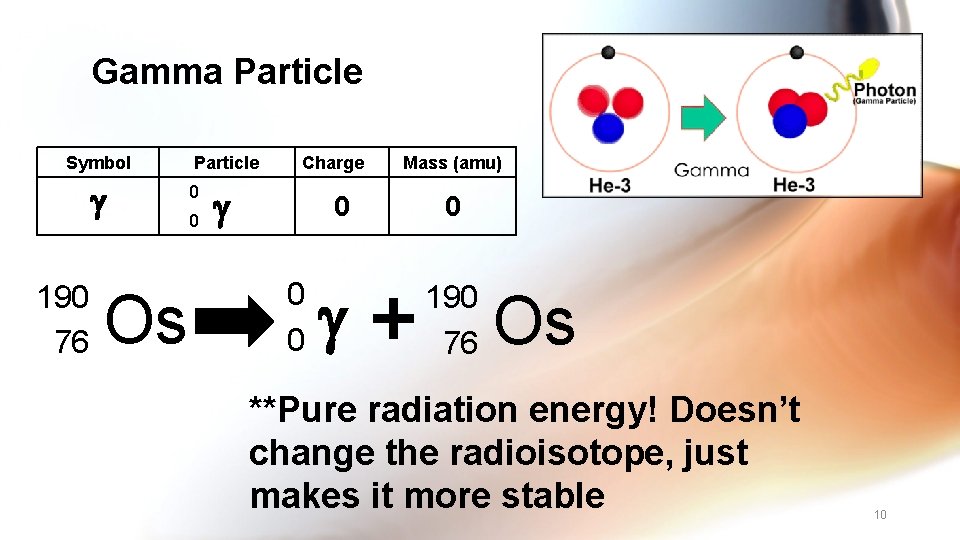
Gamma Particle Symbol 190 76 Os Particle 0 0 Charge 0 0 0 Mass (amu) 0 190 76 Os **Pure radiation energy! Doesn’t change the radioisotope, just makes it more stable 10

Radiation Protection • Shielding alpha – paper, clothing beta – lab coat, gloves, aluminum foil gamma- 6 inches of lead, 12 feet of concrete • Limit time exposed • Keep distance from source 11
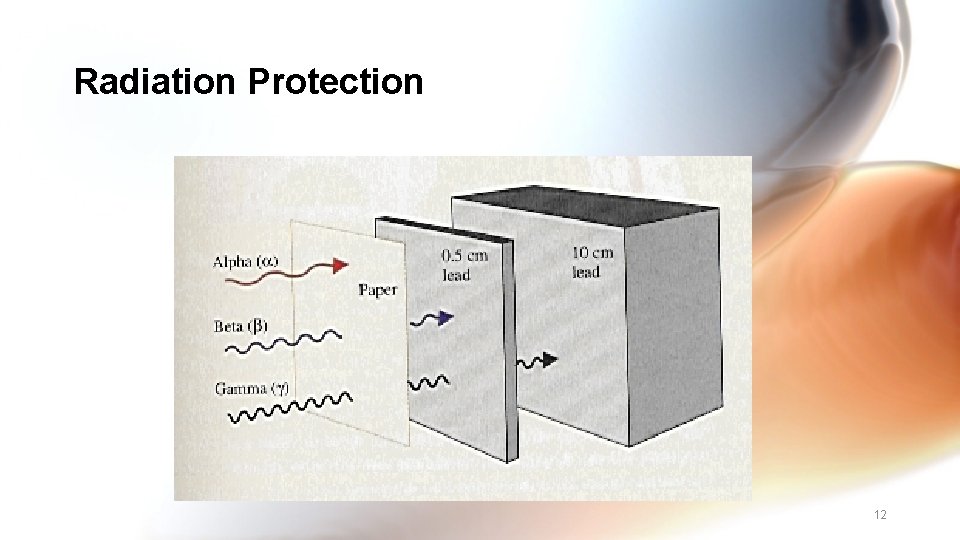
Radiation Protection 12

Learning Check Write the nuclear equation for the beta emitter Co-60. 13

Solution Write the nuclear equation for the Beta emitter Co-60. 60 Co 60 Ni 27 28 + 0 e -1 14

Producing Radioactive Isotopes Bombardment of atoms produces radioisotopes = 60 59 Co = 60 + 1 n 27 0 = 27 cobalt atom 56 Mn + 25 4 H e 2 = 27 neutron manganese alpha radioisotope particle 15

Learning Check What radioactive isotope is produced in the following bombardment of boron? 10 B 5 + 4 He 2 ? + 1 n 0 16

Solution What radioactive isotope is produced in the following bombardment of boron? 10 B 5 + 4 He 2 13 N 7 + 1 n 0 nitrogen radioisotope 17

Sources of Radioactivity • Primordial - from before the creation of the Earth • Cosmogenic - formed as a result of cosmic ray interactions • Human produced - enhanced or formed due to human actions (minor amounts compared to natural)

Where are the Sources of Radioactivity? • Naturally Occurring Sources: • Radon from the decay of Uranium and Thorium • Potassium -40 – found in minerals and in plants • Carbon 14 – Found in Plants and Animal tissue • Manmade Sources: • Medical use of Radioactive Isotopes • Certain Consumer products –(eg Smoke detectors) • Fallout from nuclear testing • Emissions from Nuclear Power plants

Radioactivity – Is it a Health Problem? • The Alpha, Beta and Gamma particles all add energy to the body’s tissues. The effect is called the Ionizing Energy. It can alter DNA. • Even though Alpha particles are not very penetrative if the decaying atom is already in the body (inhalation, ingestion) they can cause trouble. • The Time, Distance and Shielding principle Science Park HS -- Honors Chemistry
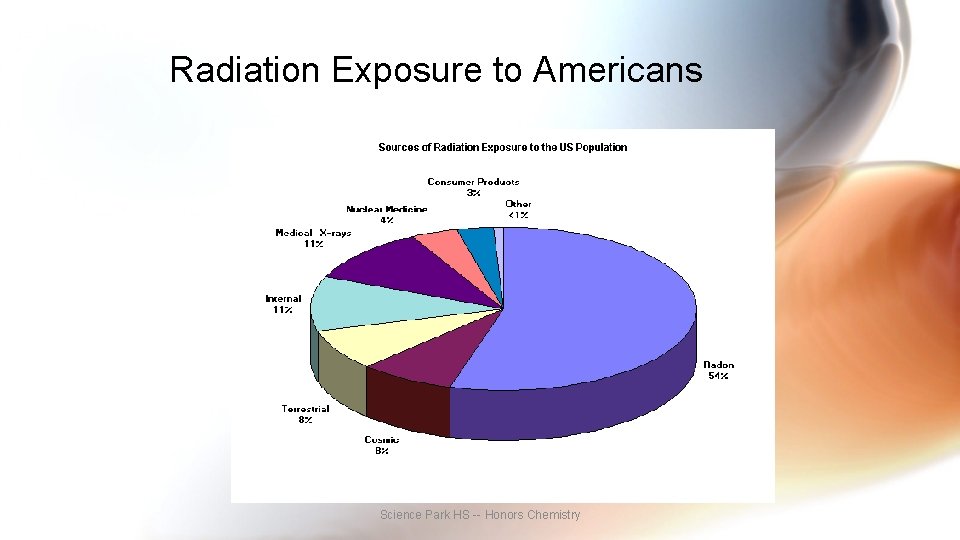
Radiation Exposure to Americans Science Park HS -- Honors Chemistry

Ch. 6 Notes Day 2

Objectives • SWBAT identify and explain the purpose of the three main components of a nuclear power plant • SWBAT explain how humans control fission chain reactions to generate power inside the nuclear core • SWBAT explain some of the measures nuclear power plants take to ensure safety

Parts of a Nuclear Power Plant

Main Parts of a Nuclear Power Plant - Containment Building seals in nuclear reactor - Radioactive fuel rods made of Uranium or Plutonium heat water with radiation **makes water radioactive - Hot radioactive water turns water inside heat exchanger to steam - Steam from heat exchanger spins turbines - Turbines spin rod inside generator, to produce electricity - Steam turned to water in condenser, then returned to heat exchanger - Cold air in cooling tower or cold river water used to cool down the condenser - Cooling towers only make steam!!! The water in each of the three sections, is contained in those section to prevent contamination!!!
Reactor and Fission Chain Reaction
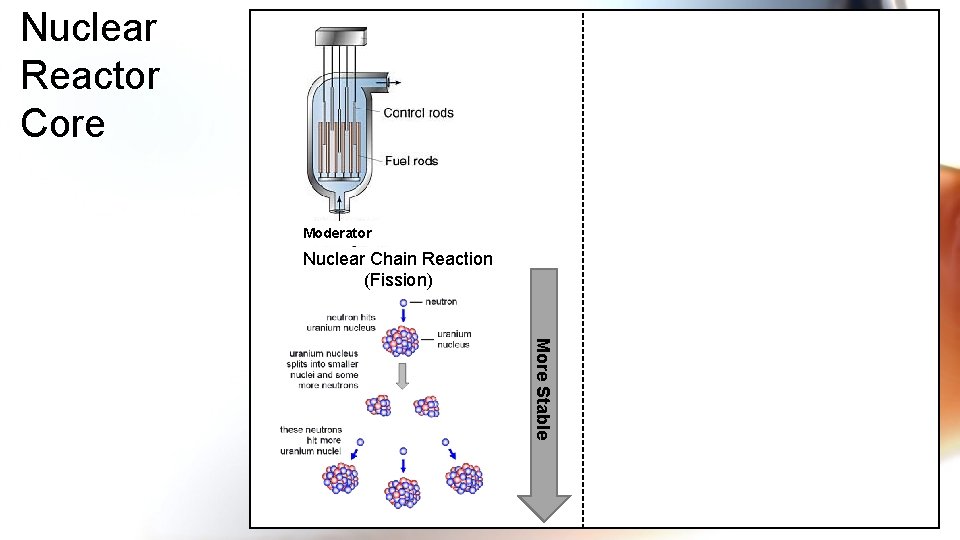
Nuclear Reactor Core Moderator Nuclear Chain Reaction (Fission) More Stable
Nuclear Power Pros & Cons

Ch. 6 Notes Day 3

Objectives • SWBAT define radioactivity • SWBAT explain the concept of half-life through experimentation in the lab

Vocabulary • Radioactivity – process where an unstable atomic nucleus emits charged particles and energy to become more stable. • Nuclear Radiation- charged particles emitted from radioactive nuclei called radioisotopes. • Alpha Decay – emits an alpha particle. Alpha particles can be stopped with paper • Beta Decay – emits a beta particle. Beta particles can be stopped with aluminum foil • Gamma Decay – emits gamma rays. Gamma rays are pure energy, and can only be stopped with 12 or more feet of concrete. (This is what made the Hulk…)

Half Life • Half-Life – time required for half of a radioisotope to decay. • Used for dating old materials like rocks • Radon-222 half-life of 3. 82 days • Uranium-238 half-life of 4. 47 billion years # of Half-lives 0 1 2 3 4 5 6 Amount of Radio Isotope Left 1 ½ 1/4 1/8 1/16 1/32 1/64 100% 50% 25% 12. 5% 6. 25% 3. 125% 1. 56% Percentage Left

Ch. 6 Notes Day 4

Objectives • SWBAT use the half-life equation to determine the amount of an radioisotope that remains after a certain amount of time • SWBAT to determine how many half-lives have elapsed, given the starting and ending amounts of a substance.

Half-Life of a Radioisotope The time for the radiation level to fall (decay) to one-half its initial value decay curve initial 1 half-life 8 mg 4 mg 2 2 mg 3 1 mg 35

Examples of Half-Life Isotope C-15 Ra-224 Ra-223 I-125 C-14 U-235 Half life 2. 4 sec 3. 6 days 12 days 60 days 5700 years 710 000 years 36

Half-Life Equation # of half-lives (n) = • N(t) - Amount of radioisotope remaining • No – Amount of radioisotope to start (at time = 0) • t - time elapsed • t 1/2 – half- life

Learning Check The half life of I-123 is 13 hr. How much of a 64 mg sample of I-123 is left after 26 hours? 38

Solution t 1/2 26 hours Amount initial = = 13 hrs 2 x t 1/2 = 64 mg Amount remaining = 64 mg x ½ = 16 mg 39
- Slides: 39