The Abbott PostApproval Study 1 Mitra Clip Registry

The Abbott Post-Approval Study 1 Mitra. Clip Registry: 1 -year Results of the First 2, 000 Patients in the Transcatheter Valve Therapy Registry James Hermiller MD, Ramon Quesada MD, Mark Reisman MD, Vasilis Babaliaros MD, D. Scott Lim MD, Jason Rogers MD, Robert Kipperman MD, Andrew Wang MD, Paul Sorajja MD, Vivek Rajagopal MD, and Saibal Kar MD

Disclosures James Hermiller, MD: q I do not have any potential conflict of interest I have the following potential conflicts of interest to report: Honorarium: Abbott Vascular Institutional grant/research support: Abbott Vascular Consultant: Abbott Vascular
Mitra. Clip® Therapy Worldwide Clinical Experience • Received CE mark in 2008. • Received FDA approval in 2013 for significant symptomatic degenerative mitral regurgitation (DMR) in patients at prohibitive risk (PR) for MV surgery. • >60, 000 patients treated worldwide. All drawings are artist's representations only and should not be considered as an engineering drawing or photograph.

Background • As part of the Mitra. Clip approval by the FDA, Abbott committed to a registry-based post-approval study (Mitra. Clip PAS-1) to study the long term safety and effectiveness in the real-world commercial use of the Mitra. Clip device. • Recently, Sorajja, et. al. (2017)1 reported 30 -day and 1 -year results from records linked to CMS administrative claims. Sorajja P, et al. J Am Coll Cardiol. 2017; 70(19): 2315 -27. 1

Purpose • To present results from the first 2, 000 patients enrolled in the Mitra. Clip Post-Approval Study 1 (PAS-1).

Study Design • Prospective, single-arm, multi-center, observational study • Data extracted from first 2, 000 Mitra. Clip patients consecutively entered into the TVT Registry PAS-1 is the first and largest report of site-reported echocardiographic and clinical (functional and quality of life) outcomes from commercial US experience as captured in the TVT registry through 1 -year.

Methods • 2, 000 Mitra. Clip commercial patients entered into the TVT Registry at 125 sites in the US • Clinical Events Committee (CEC): Duke Clinical Research Institute (DCRI) • Study Endpoints at 30 days and 1 year: ―All-cause mortality ―Stroke ―The echo parameters of MR severity, LVEDV and LVIDd ―NYHA Functional Class ―All-cause and Heart Failure (HF)-related hospitalizations (at 1 year only) • Additional measures of effectiveness at 30 days and 1 year ― 6 -Minute Walk Test (6 MWT) distance ―Kansas City Cardiomyopathy Questionnaire (KCCQ) quality-of-life
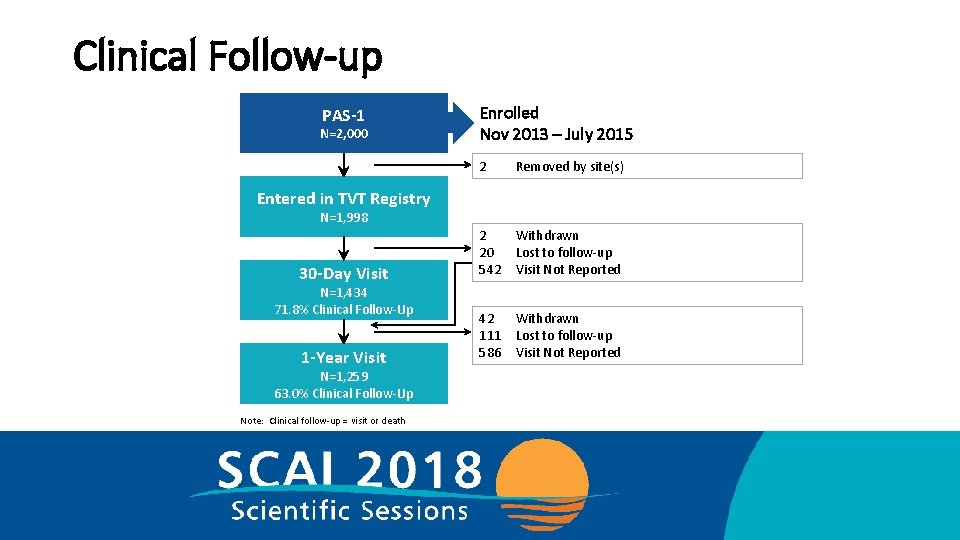
Clinical Follow-up PAS-1 N=2, 000 Enrolled Nov 2013 – July 2015 2 Removed by site(s) 2 20 542 Withdrawn Lost to follow-up Visit Not Reported 42 111 586 Withdrawn Lost to follow-up Visit Not Reported Entered in TVT Registry N=1, 998 30 -Day Visit N=1, 434 71. 8% Clinical Follow-Up 1 -Year Visit N=1, 259 63. 0% Clinical Follow-Up Note: Clinical follow-up = visit or death
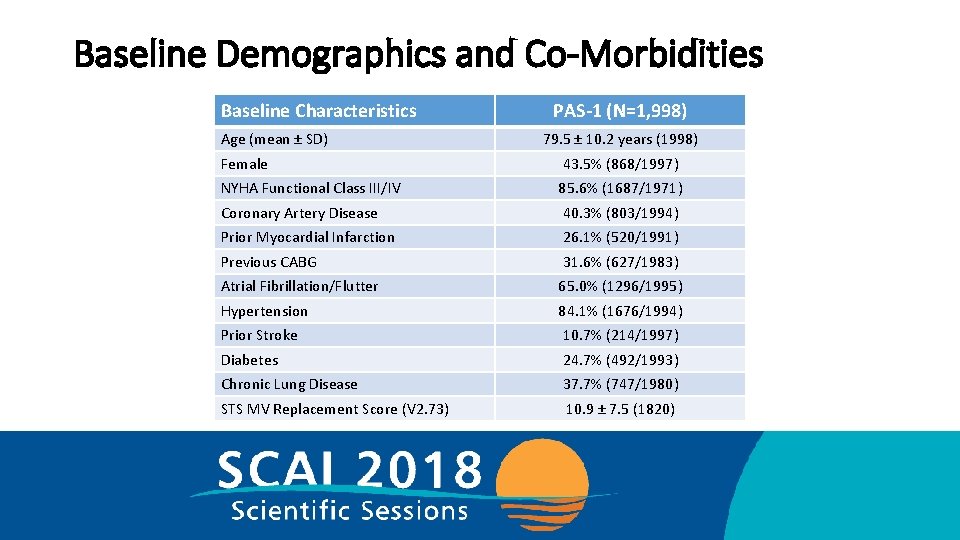
Baseline Demographics and Co-Morbidities Baseline Characteristics Age (mean ± SD) PAS-1 (N=1, 998) 79. 5 ± 10. 2 years (1998) Female 43. 5% (868/1997) NYHA Functional Class III/IV 85. 6% (1687/1971) Coronary Artery Disease 40. 3% (803/1994) Prior Myocardial Infarction 26. 1% (520/1991) Previous CABG 31. 6% (627/1983) Atrial Fibrillation/Flutter 65. 0% (1296/1995) Hypertension 84. 1% (1676/1994) Prior Stroke 10. 7% (214/1997) Diabetes 24. 7% (492/1993) Chronic Lung Disease 37. 7% (747/1980) STS MV Replacement Score (V 2. 73) 10. 9 ± 7. 5 (1820)
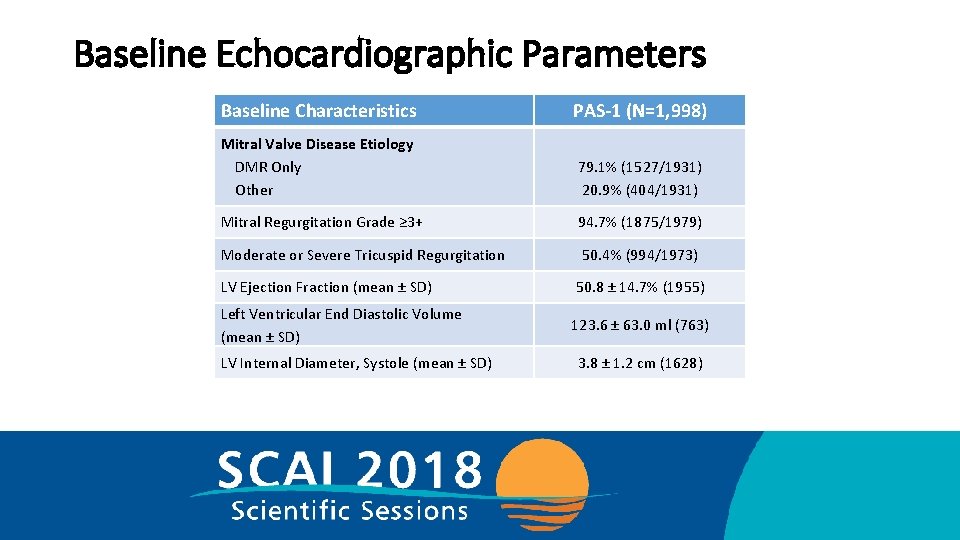
Baseline Echocardiographic Parameters Baseline Characteristics PAS-1 (N=1, 998) Mitral Valve Disease Etiology DMR Only Other 79. 1% (1527/1931) 20. 9% (404/1931) Mitral Regurgitation Grade ≥ 3+ 94. 7% (1875/1979) Moderate or Severe Tricuspid Regurgitation 50. 4% (994/1973) LV Ejection Fraction (mean ± SD) 50. 8 ± 14. 7% (1955) Left Ventricular End Diastolic Volume (mean ± SD) 123. 6 ± 63. 0 ml (763) LV Internal Diameter, Systole (mean ± SD) 3. 8 ± 1. 2 cm (1628)
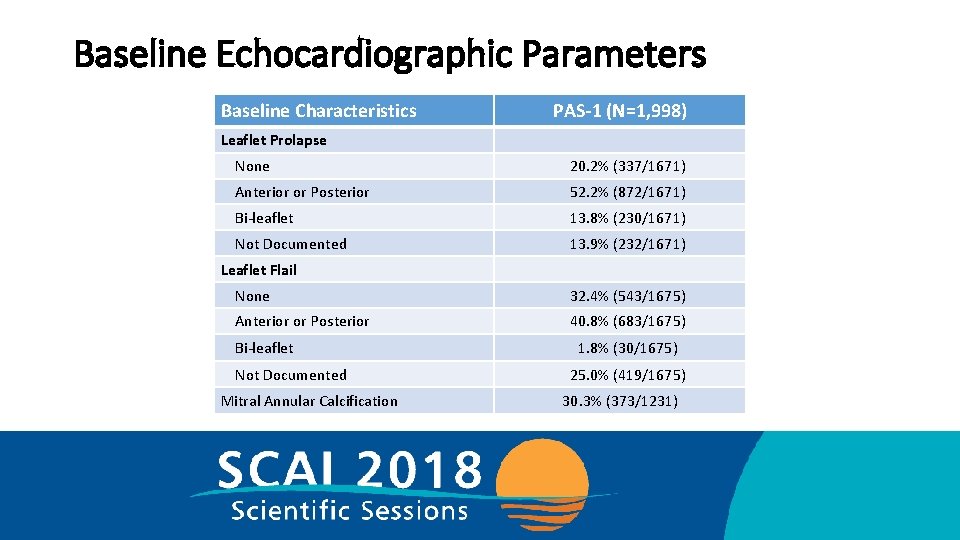
Baseline Echocardiographic Parameters Baseline Characteristics PAS-1 (N=1, 998) Leaflet Prolapse None 20. 2% (337/1671) Anterior or Posterior 52. 2% (872/1671) Bi-leaflet 13. 8% (230/1671) Not Documented 13. 9% (232/1671) Leaflet Flail None 32. 4% (543/1675) Anterior or Posterior 40. 8% (683/1675) Bi-leaflet Not Documented Mitral Annular Calcification 1. 8% (30/1675) 25. 0% (419/1675) 30. 3% (373/1231)
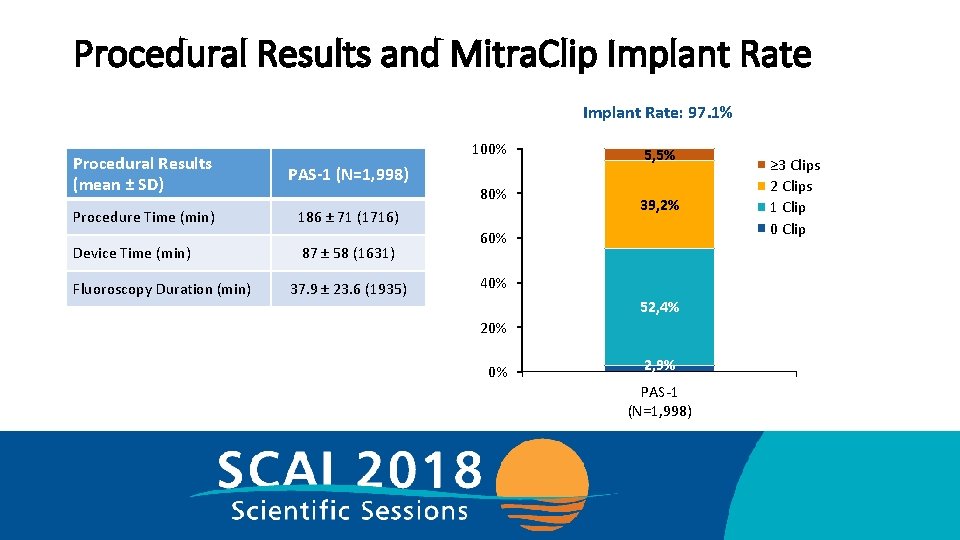
Procedural Results and Mitra. Clip Implant Rate: 97. 1% 100% Procedural Results (mean ± SD) PAS-1 (N=1, 998) Procedure Time (min) 186 ± 71 (1716) Device Time (min) Fluoroscopy Duration (min) 87 ± 58 (1631) 37. 9 ± 23. 6 (1935) 80% 5, 5% 39, 2% 60% 40% 52, 4% 20% 0% 2, 9% PAS-1 (N=1, 998) ≥ 3 Clips 2 Clips 1 Clip 0 Clip
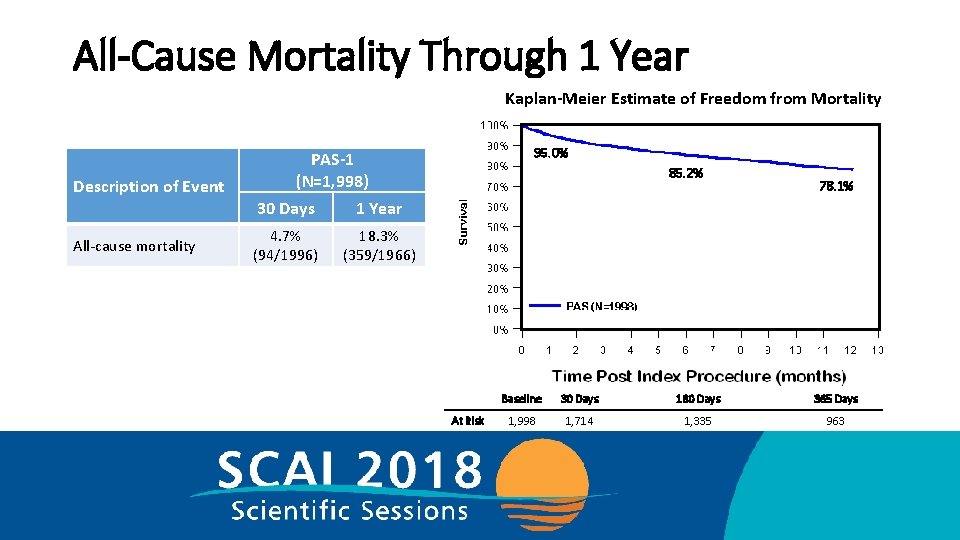
All-Cause Mortality Through 1 Year Kaplan-Meier Estimate of Freedom from Mortality Description of Event PAS-1 (N=1, 998) 30 Days 1 Year All-cause mortality 4. 7% (94/1996) 95. 0% 85. 2% 78. 1% 18. 3% (359/1966) At Risk Baseline 30 Days 180 Days 365 Days 1, 998 1, 714 1, 335 963
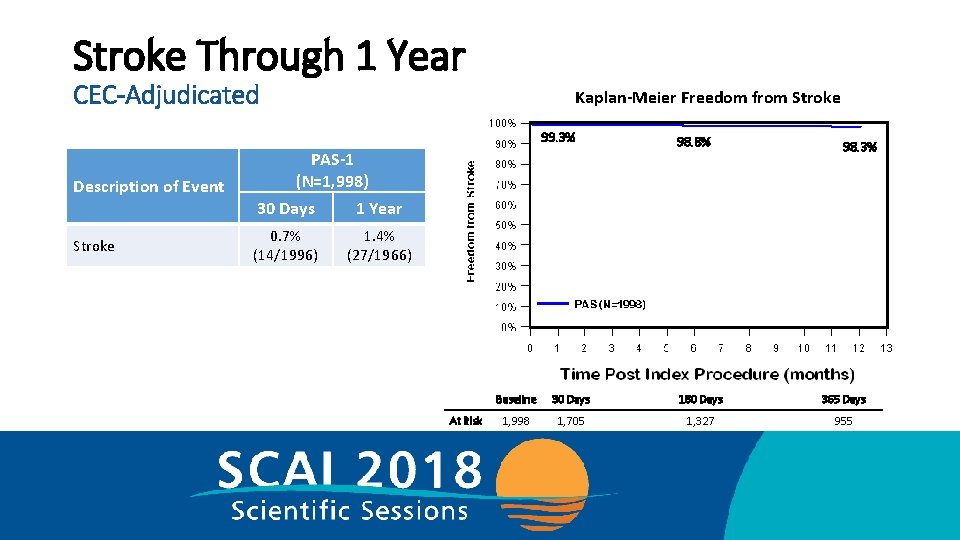
Stroke Through 1 Year CEC-Adjudicated Kaplan-Meier Freedom from Stroke 99. 3% Description of Event PAS-1 (N=1, 998) 30 Days 1 Year Stroke 0. 7% (14/1996) 98. 8% 98. 3% 1. 4% (27/1966) At Risk Baseline 30 Days 180 Days 365 Days 1, 998 1, 705 1, 327 955
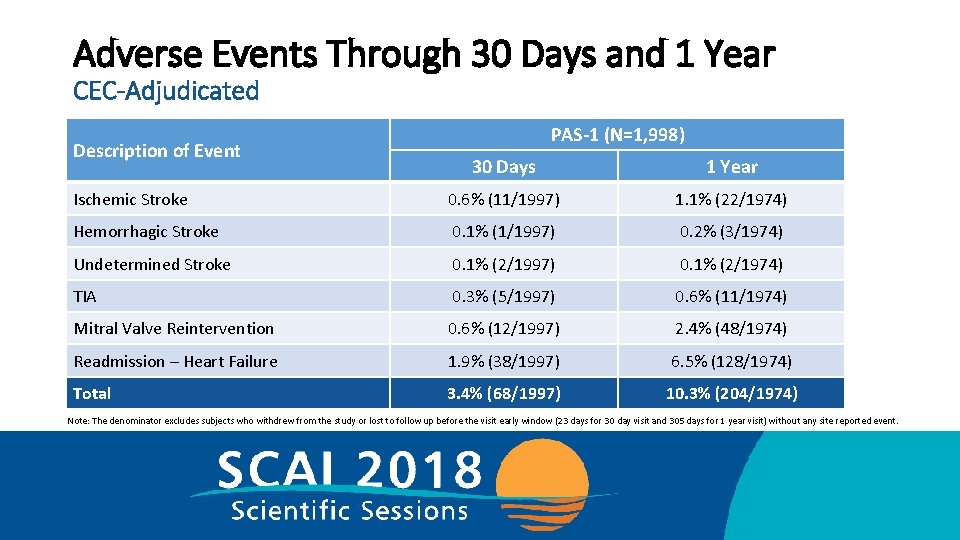
Adverse Events Through 30 Days and 1 Year CEC-Adjudicated Description of Event PAS-1 (N=1, 998) 30 Days 1 Year Ischemic Stroke 0. 6% (11/1997) 1. 1% (22/1974) Hemorrhagic Stroke 0. 1% (1/1997) 0. 2% (3/1974) Undetermined Stroke 0. 1% (2/1997) 0. 1% (2/1974) TIA 0. 3% (5/1997) 0. 6% (11/1974) Mitral Valve Reintervention 0. 6% (12/1997) 2. 4% (48/1974) Readmission – Heart Failure 1. 9% (38/1997) 6. 5% (128/1974) Total 3. 4% (68/1997) 10. 3% (204/1974) Note: The denominator excludes subjects who withdrew from the study or lost to follow up before the visit early window (23 days for 30 day visit and 305 days for 1 year visit) without any site reported event.
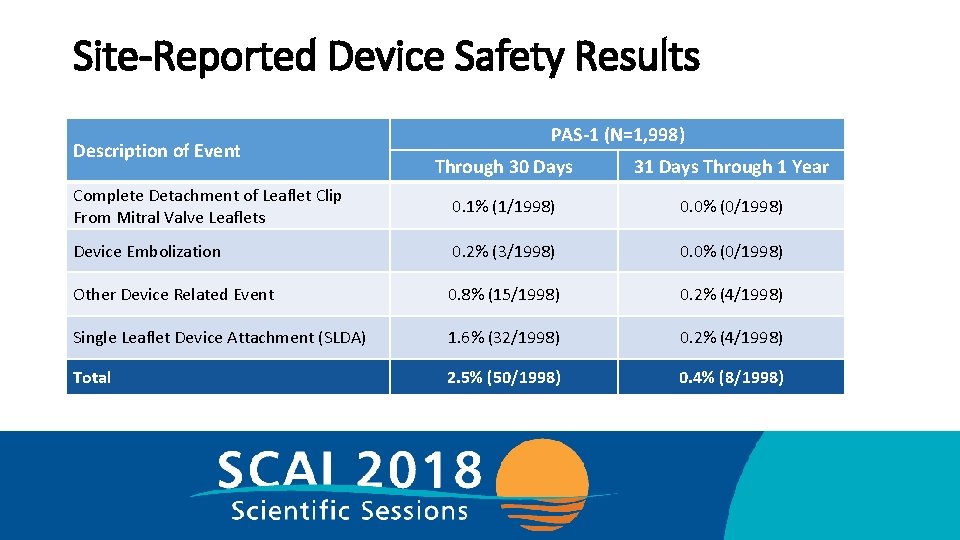
Site-Reported Device Safety Results Description of Event PAS-1 (N=1, 998) Through 30 Days 31 Days Through 1 Year Complete Detachment of Leaflet Clip From Mitral Valve Leaflets 0. 1% (1/1998) 0. 0% (0/1998) Device Embolization 0. 2% (3/1998) 0. 0% (0/1998) Other Device Related Event 0. 8% (15/1998) 0. 2% (4/1998) Single Leaflet Device Attachment (SLDA) 1. 6% (32/1998) 0. 2% (4/1998) Total 2. 5% (50/1998) 0. 4% (8/1998)
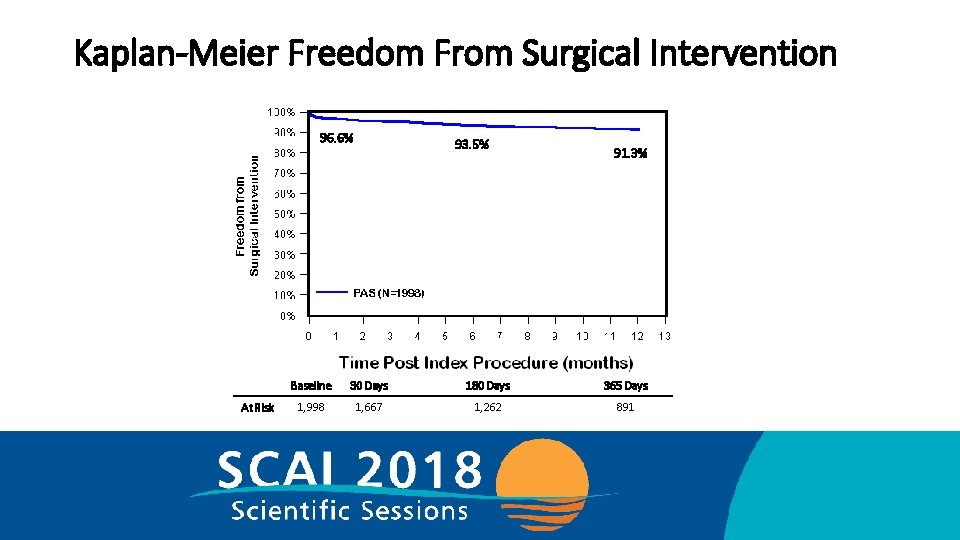
Kaplan-Meier Freedom From Surgical Intervention 96. 6% At Risk 93. 5% 91. 3% Baseline 30 Days 180 Days 365 Days 1, 998 1, 667 1, 262 891
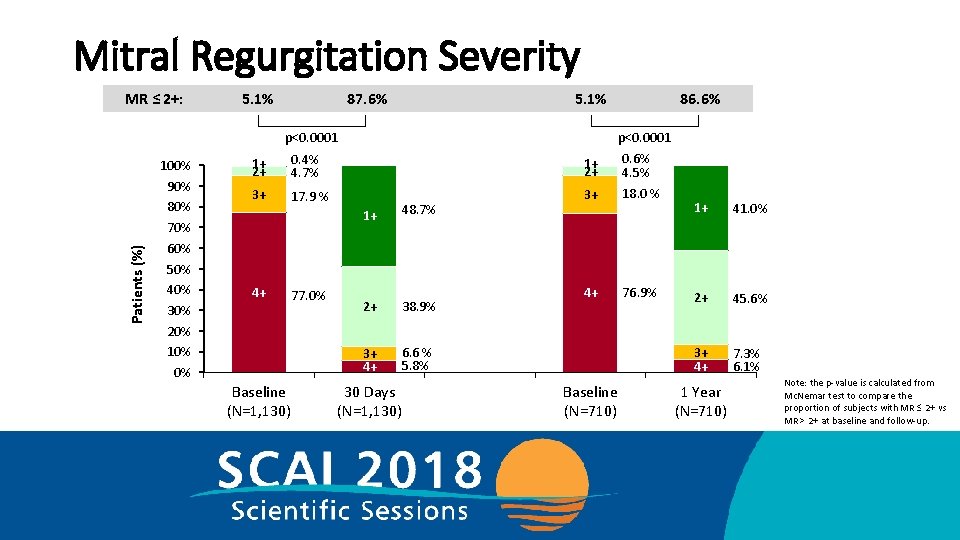
Mitral Regurgitation Severity MR ≤ 2+: 5. 1% 87. 6% 5. 1% Patients (%) p<0. 0001 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% 86. 6% p<0. 0001 1+ 2+ 0. 4% 4. 7% 1+ 2+ 0. 6% 4. 5% 3+ 17. 9 % 3+ 18. 0 % 4+ 76. 9% 4+ 77. 0% Baseline (N=1, 130) 1+ 48. 7% 2+ 38. 9% 3+ 4+ 6. 6 % 5. 8% 30 Days (N=1, 130) Baseline (N=710) 1+ 41. 0% 2+ 45. 6% 3+ 4+ 7. 3% 6. 1% 1 Year (N=710) Note: the p-value is calculated from Mc. Nemar test to compare the proportion of subjects with MR ≤ 2+ vs MR > 2+ at baseline and follow-up.
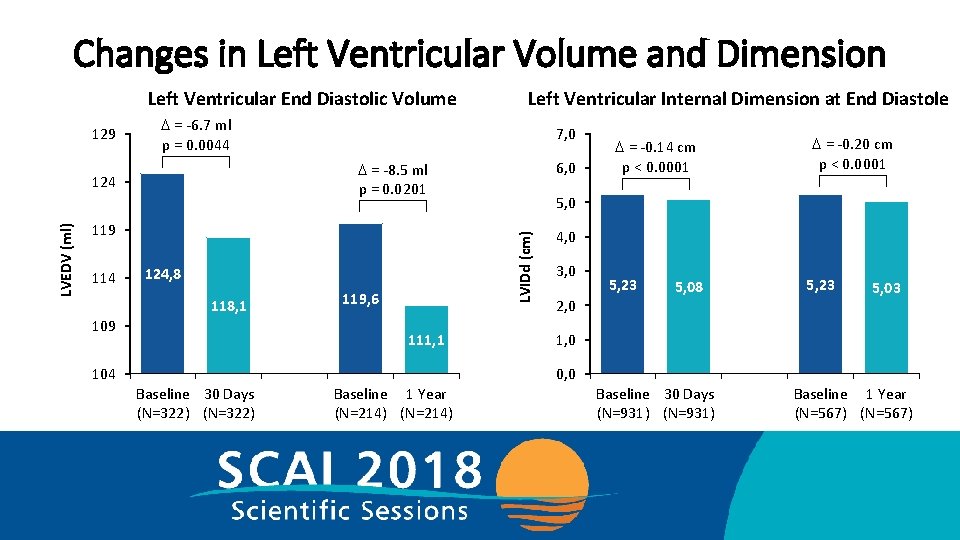
Changes in Left Ventricular Volume and Dimension Left Ventricular End Diastolic Volume 129 D = -6. 7 ml p = 0. 0044 119 124, 8 118, 1 109 6, 0 119, 6 111, 1 104 D = -0. 14 cm p < 0. 0001 D = -0. 20 cm p < 0. 0001 5, 0 LVIDd (cm) LVEDV (ml) 7, 0 D = -8. 5 ml p = 0. 0201 124 114 Left Ventricular Internal Dimension at End Diastole 4, 0 3, 0 2, 0 5, 23 5, 08 5, 23 5, 03 1, 0 0, 0 Baseline 30 Days (N=322) Baseline 1 Year (N=214) Baseline 30 Days (N=931) Baseline 1 Year (N=567)
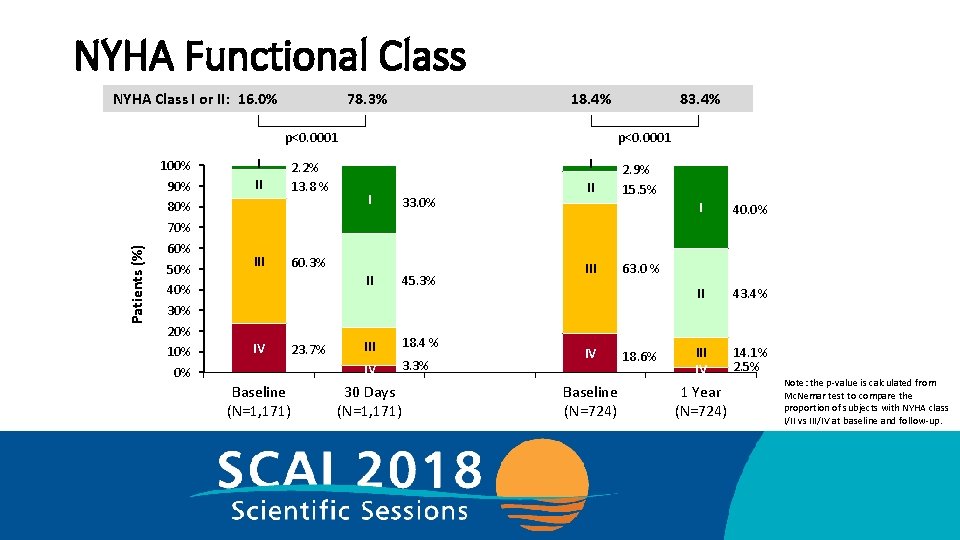
NYHA Functional Class NYHA Class I or II: 16. 0% 78. 3% 18. 4% Patients (%) p<0. 0001 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% I II IV 2. 2% 13. 8 % p<0. 0001 I I 33. 0% II 45. 3% III 18. 4 % IV 3. 3% 60. 3% 23. 7% Baseline (N=1, 171) 83. 4% 30 Days (N=1, 171) II IV Baseline (N=724) 2. 9% 15. 5% I 40. 0% II 43. 4% III IV 14. 1% 2. 5% 63. 0 % 18. 6% 1 Year (N=724) Note: the p-value is calculated from Mc. Nemar test to compare the proportion of subjects with NYHA class I/II vs III/IV at baseline and follow-up.
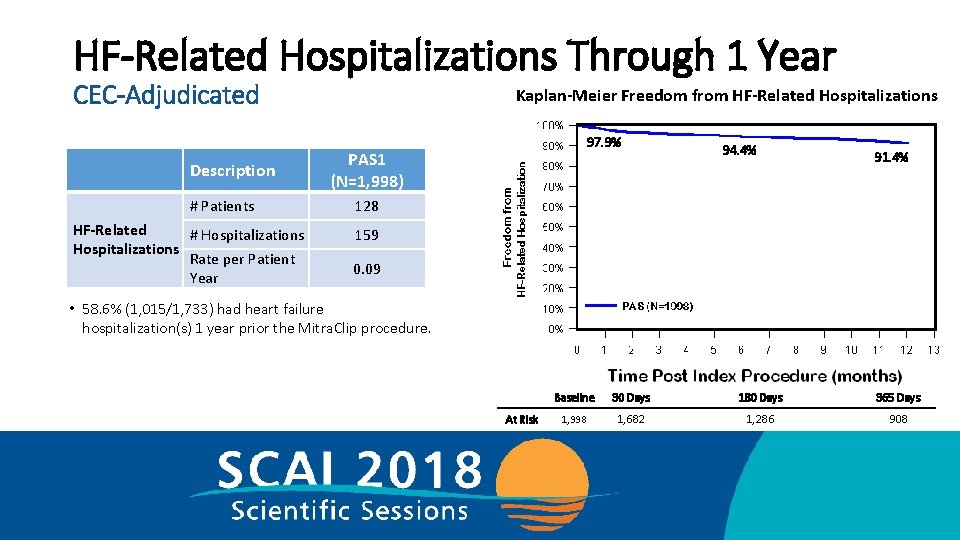
HF-Related Hospitalizations Through 1 Year CEC-Adjudicated Description # Patients HF-Related # Hospitalizations Rate per Patient Year Kaplan-Meier Freedom from HF-Related Hospitalizations 97. 9% PAS 1 (N=1, 998) 94. 4% 91. 4% 128 159 0. 09 • 58. 6% (1, 015/1, 733) had heart failure hospitalization(s) 1 year prior the Mitra. Clip procedure. At Risk Baseline 30 Days 180 Days 365 Days 1, 998 1, 682 1, 286 908
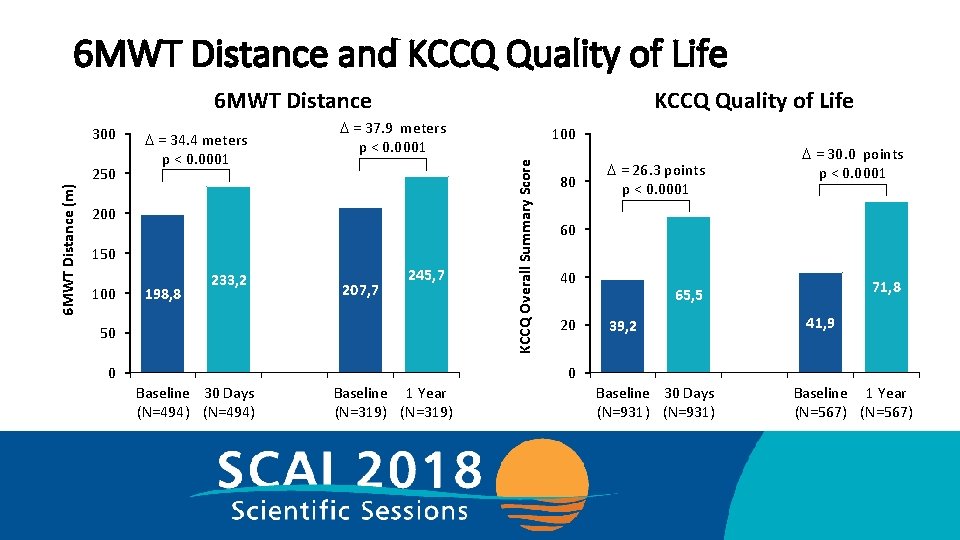
6 MWT Distance and KCCQ Quality of Life 6 MWT Distance (m) 250 D = 34. 4 meters p < 0. 0001 D = 37. 9 meters p < 0. 0001 200 150 100 198, 8 233, 2 207, 7 245, 7 50 0 100 KCCQ Overall Summary Score 300 KCCQ Quality of Life 80 D = 26. 3 points p < 0. 0001 D = 30. 0 points p < 0. 0001 60 40 20 71, 8 65, 5 39, 2 41, 9 0 Baseline 30 Days (N=494) Baseline 1 Year (N=319) Baseline 30 Days (N=931) Baseline 1 Year (N=567)
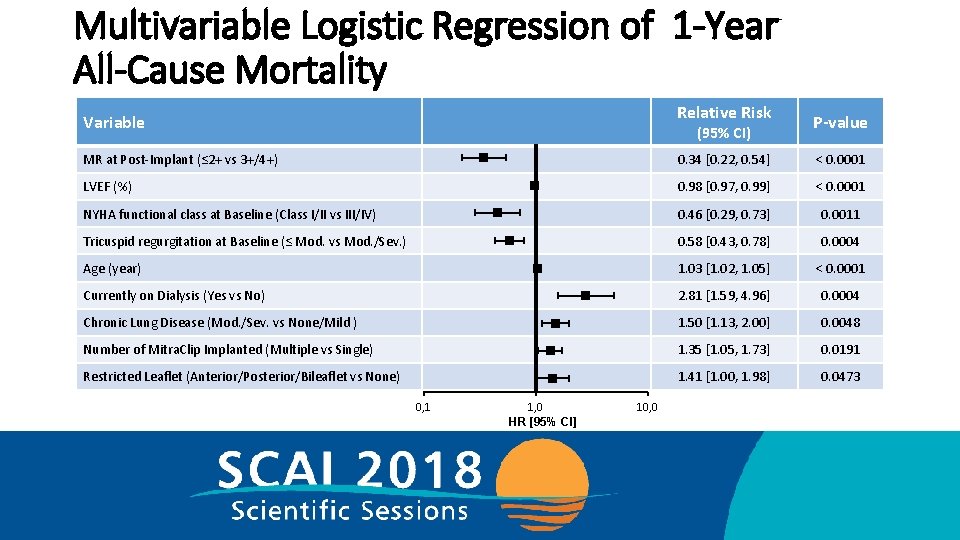
Multivariable Logistic Regression of 1 -Year All-Cause Mortality Relative Risk Variable (95% CI) P-value MR at Post-Implant (≤ 2+ vs 3+/4+) 0. 34 [0. 22, 0. 54] < 0. 0001 LVEF (%) 0. 98 [0. 97, 0. 99] < 0. 0001 NYHA functional class at Baseline (Class I/II vs III/IV) 0. 46 [0. 29, 0. 73] 0. 0011 Tricuspid regurgitation at Baseline (≤ Mod. vs Mod. /Sev. ) 0. 58 [0. 43, 0. 78] 0. 0004 Age (year) 1. 03 [1. 02, 1. 05] < 0. 0001 Currently on Dialysis (Yes vs No) 2. 81 [1. 59, 4. 96] 0. 0004 Chronic Lung Disease (Mod. /Sev. vs None/Mild ) 1. 50 [1. 13, 2. 00] 0. 0048 Number of Mitra. Clip Implanted (Multiple vs Single) 1. 35 [1. 05, 1. 73] 0. 0191 Restricted Leaflet (Anterior/Posterior/Bileaflet vs None) 1. 41 [1. 00, 1. 98] 0. 0473 0, 1 1, 0 HR [95% CI] 10, 0
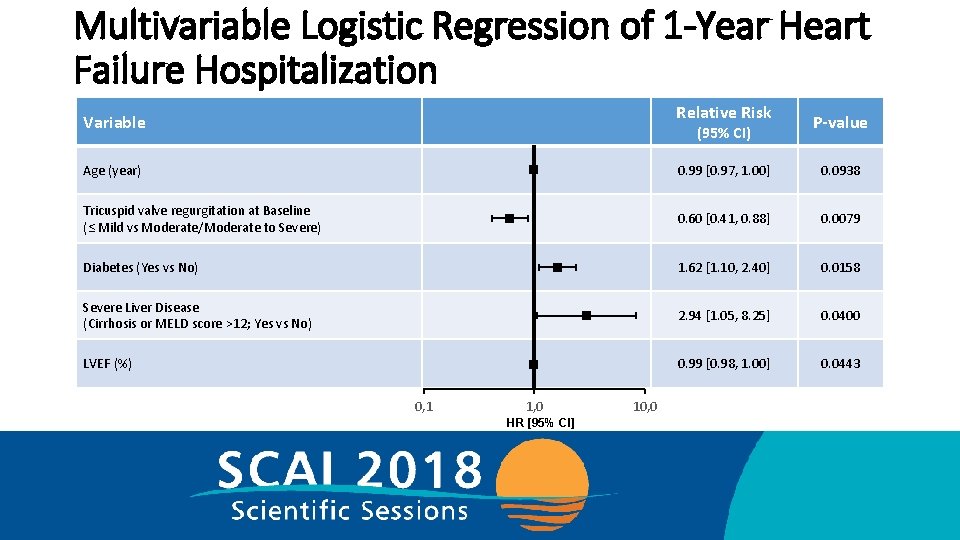
Multivariable Logistic Regression of 1 -Year Heart Failure Hospitalization Relative Risk Variable (95% CI) P-value Age (year) 0. 99 [0. 97, 1. 00] 0. 0938 Tricuspid valve regurgitation at Baseline (≤ Mild vs Moderate/Moderate to Severe) 0. 60 [0. 41, 0. 88] 0. 0079 Diabetes (Yes vs No) 1. 62 [1. 10, 2. 40] 0. 0158 Severe Liver Disease (Cirrhosis or MELD score >12; Yes vs No) 2. 94 [1. 05, 8. 25] 0. 0400 LVEF (%) 0. 99 [0. 98, 1. 00] 0. 0443 0, 1 1, 0 HR [95% CI] 10, 0
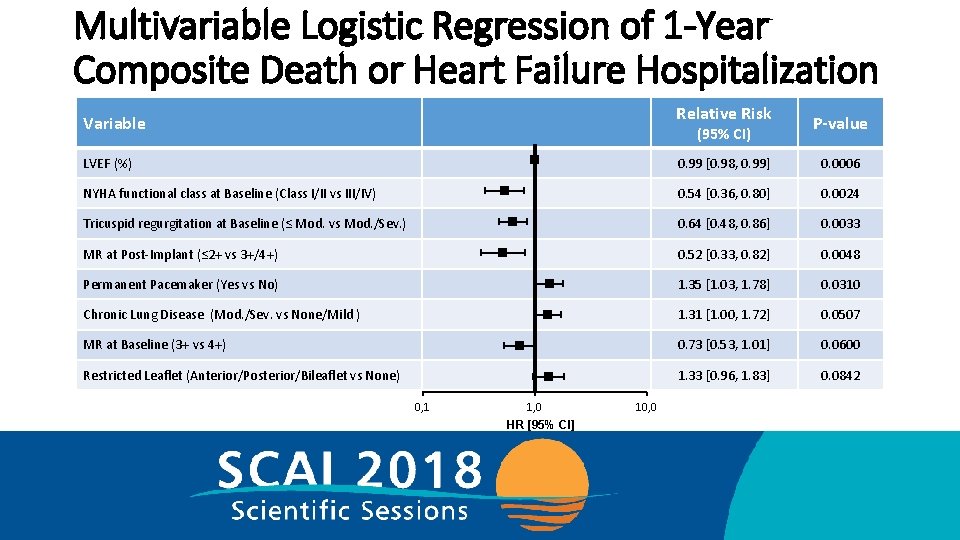
Multivariable Logistic Regression of 1 -Year Composite Death or Heart Failure Hospitalization Relative Risk Variable (95% CI) P-value LVEF (%) 0. 99 [0. 98, 0. 99] 0. 0006 NYHA functional class at Baseline (Class I/II vs III/IV) 0. 54 [0. 36, 0. 80] 0. 0024 Tricuspid regurgitation at Baseline (≤ Mod. vs Mod. /Sev. ) 0. 64 [0. 48, 0. 86] 0. 0033 MR at Post-Implant (≤ 2+ vs 3+/4+) 0. 52 [0. 33, 0. 82] 0. 0048 Permanent Pacemaker (Yes vs No) 1. 35 [1. 03, 1. 78] 0. 0310 Chronic Lung Disease (Mod. /Sev. vs None/Mild ) 1. 31 [1. 00, 1. 72] 0. 0507 MR at Baseline (3+ vs 4+) 0. 73 [0. 53, 1. 01] 0. 0600 Restricted Leaflet (Anterior/Posterior/Bileaflet vs None) 1. 33 [0. 96, 1. 83] 0. 0842 0, 1 1, 0 HR [95% CI] 10, 0

Limitations • Data entry into the TVT Registry remains voluntary and thus subject to reporting bias; • PAS-1 provides valuable information on Mitra. Clip use in the commercial setting, however, data adjudication had limited event categories • Echocardiographic variables are site reported and not assessed by a core laboratory. • Details on the echocardiographic methods, including their timing and loading conditions, were not available.

Conclusion • In US commercial use, the Mitra. Clip procedure provides meaningful clinical benefits in prohibitive risk DMR patients. • Improvement in MR severity and LV measures • Improvement in NYHA, KCCQ and 6 MWT • Acute and 1 -year outcomes in the Mitra. Clip PAS-1 are consistent with other clinical and real-world use in high risk patients. • Commercial use of the Mitra. Clip device remains a safe and effective therapeutic option for select patients and has not presented any new concerns during the follow-up of the PAS-1 study.
- Slides: 27