The 4 Laws of Thermodynamics pg 54 EQ














- Slides: 19

The 4 Laws of Thermodynamics pg. 54 EQ: What are the laws of thermodynamics? § We will evaluate the laws of thermodynamics § I will evaluate a situation and explain how the 2 nd law applies to it

The 4 Laws of Thermodynamics

The 0 th Law: Equilibrium When two objects are separately in thermodynamic equilibrium with a third object, they are in equilibrium with each other.

The st 1 Law: Energy PERPETUAL MOTION MACHINE, A DEVICE THAT WILL RUN FOREVER WITHOUT ANY OUTSIDE SOURCE OF ENERGY HTTP: //SHOWSTUFFWORKS. COM/STUFF-THEY-DONT-WANT-YOU-TOKNOW/STDWYTK-PERPETUAL-MOTION-MACHINE-VIDEO. HTM https: //www. youtube. com/watch? v=jx. KX 9_h. Uo 8 E Conservation of energy – energy is not created or destroyed, just transformed to other forms

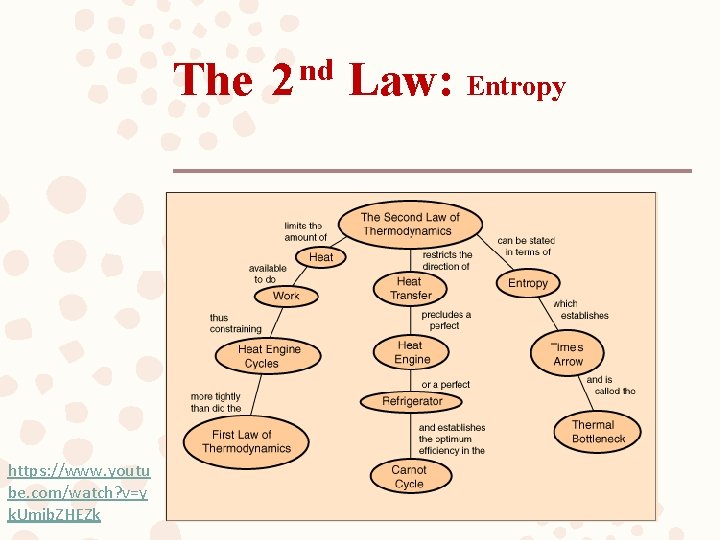
The 2 nd Law: Entropy https: //www. youtu be. com/watch? v=y k. Umib. ZHEZk

The 2 nd Law: Entropy Energy of all kinds in our material world disperses or spreads out if it is not hindered from doing so.

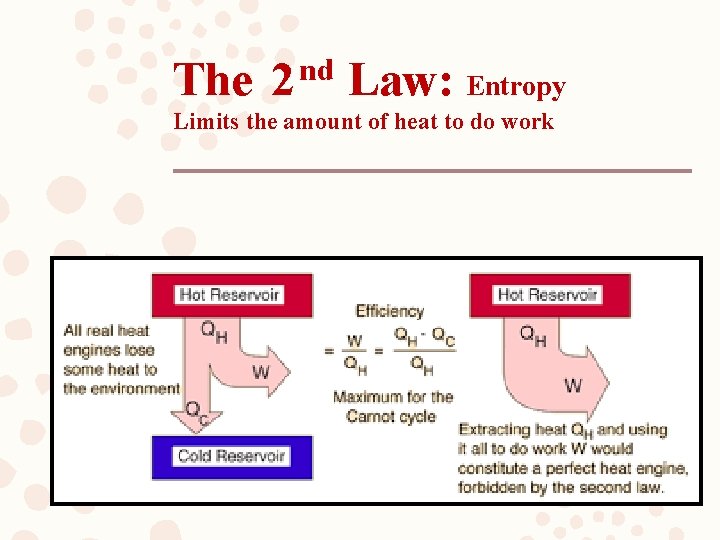
The 2 nd Law: Entropy Limits the amount of heat to do work

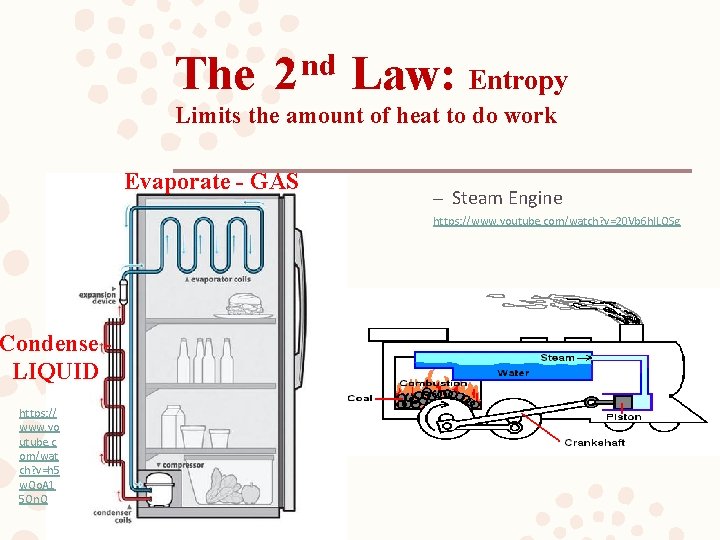
The nd 2 Law: Entropy Limits the amount of heat to do work Evaporate - GAS – Steam Engine https: //www. youtube. com/watch? v=20 Vb 6 hl. LQSg Condense LIQUID https: // www. yo utube. c om/wat ch? v=h 5 w. Qo. A 1 5 On. Q

The 2 nd Law: Entropy Restricts the direction of heat transfer – If two objects are not the same temperature then: – Heat will always flow from high to low temperatures. o Hot object will decrease in temperature and cold object will increase in temperature until they are both the same temperature.

The 2 nd Law : Entropy which establishes time’s arrow

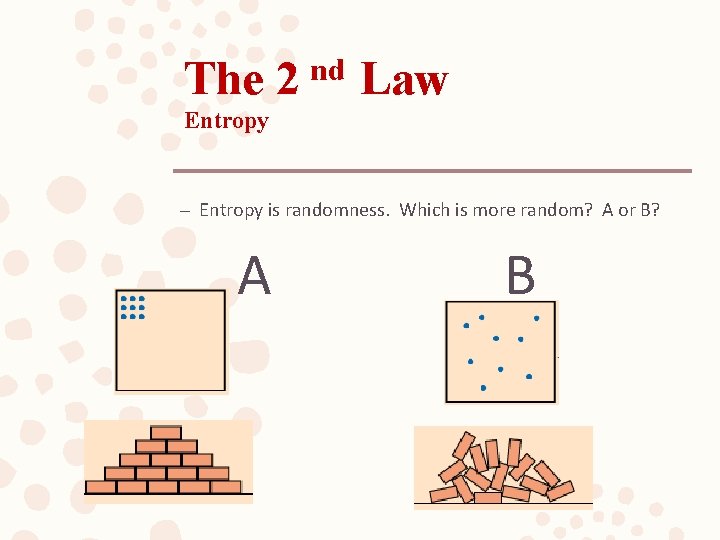
The 2 nd Law Entropy – Entropy is randomness. Which is more random? A or B? A B

The 2 nd Law Entropy – 2 nd Law of Therm. says that nature always goes from order to disorder. A B

Can’t you just restack the bricks? – Entropy can be lowered by doing work on the system. – You can stack the bricks back up by hand. – By providing energy, you can drive chemical reactions backwards. High entropy Low entropy

The 2 nd Law Entropy – Stored energy = Less entropy (unnatural) – Work has to be done to have stored energy

The 2 nd Law Entropy

The 2 nd Law Entropy – On the large scale, the ice “looks” more disordered. – On the small scale, the solid phase severely limits where the molecules could be. – The ice crystal molecules (less entropy) are much more ordered than the free moving liquid water molecules (more entroopy).

In Conclusion: Heat Death – Eventually, all energy will be entropic. – Chemical reactions will burn themselves out. – Temperatures will equalize. – Matter and energy will be spread out evenly across the universe, and no more chemical or physical processes will occur. https: //www. youtube. com/watch? v=GOr Wy_y. NBv. Y

The rd 3 Law: Temperature

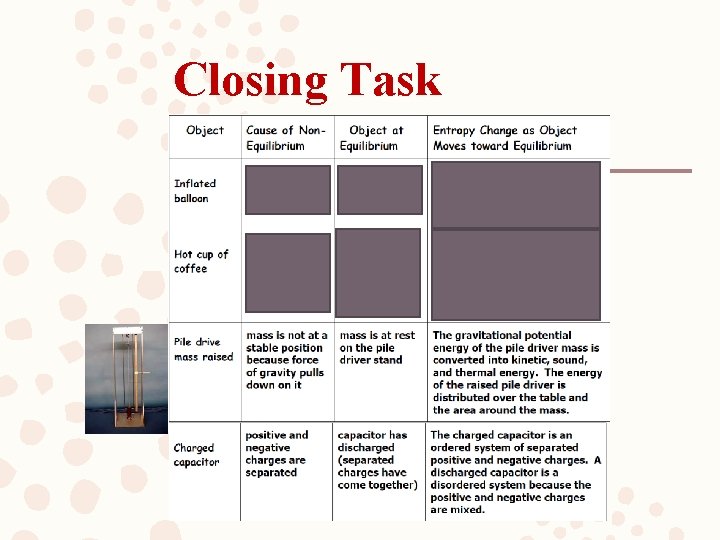
Closing Task