The 2 year Clinical Outcomes of the ABSORB

The 2 -year Clinical Outcomes of the ABSORB II Trial: First Randomized Comparison between the Absorb Everolimus Eluting Bioresorbable Vascular Scaffold and the XIENCE Everolimus Eluting Stent Bernard Chevalier Institut Jacques Cartier, Massy, France Patrick W. Serruys Imperial College, London, UK Erasmus University MC, Netherlands on behalf of the ABSORB II Investigators 12 October 2015 - San Francisco, CA - U. S. A Plenary Session VII. First Report Investigations 1. 12: 00 pm

Presentor Disclosures Bernard Chevalier is a consultant for Abbott Vascular Patrick Serruys is a member of the international advisory board of Abbott Vascular
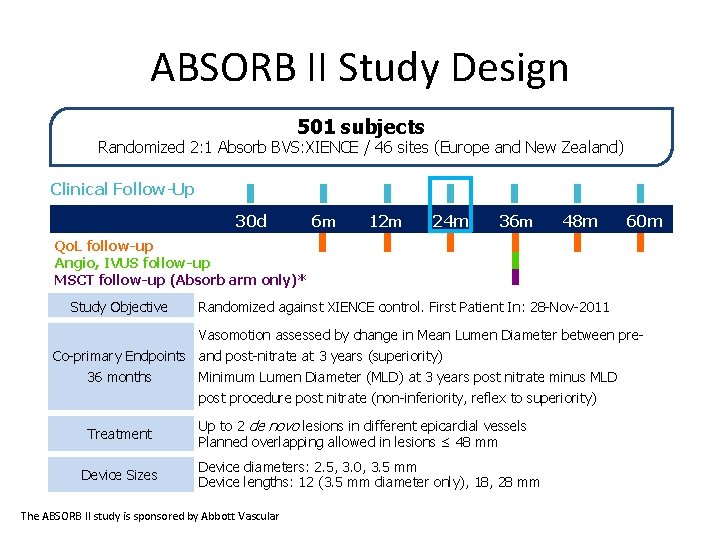
ABSORB II Study Design 501 subjects Randomized 2: 1 Absorb BVS: XIENCE / 46 sites (Europe and New Zealand) Clinical Follow-Up 30 d 6 m 12 m 24 m 36 m 48 m 60 m Qo. L follow-up Angio, IVUS follow-up MSCT follow-up (Absorb arm only)* Study Objective Randomized against XIENCE control. First Patient In: 28 -Nov-2011 Vasomotion assessed by change in Mean Lumen Diameter between pre- Co-primary Endpoints and post-nitrate at 3 years (superiority) 36 months Minimum Lumen Diameter (MLD) at 3 years post nitrate minus MLD post procedure post nitrate (non-inferiority, reflex to superiority) Treatment Device Sizes Up to 2 de novo lesions in different epicardial vessels Planned overlapping allowed in lesions ≤ 48 mm Device diameters: 2. 5, 3. 0, 3. 5 mm Device lengths: 12 (3. 5 mm diameter only), 18, 28 mm The ABSORB II study is sponsored by Abbott Vascular
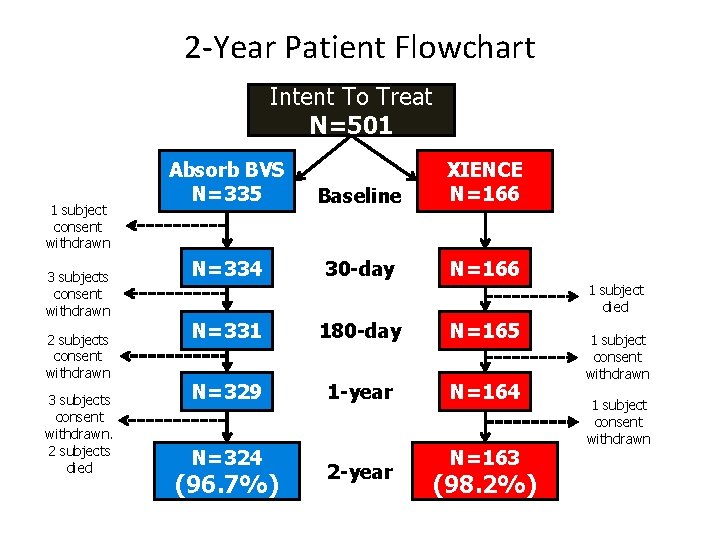
2 -Year Patient Flowchart Intent To Treat N=501 1 subject consent withdrawn 3 subjects consent withdrawn 2 subjects consent withdrawn 3 subjects consent withdrawn. 2 subjects died Absorb BVS N=335 Baseline XIENCE N=166 N=334 30 -day N=166 1 subject died N=331 180 -day N=165 N=329 1 -year N=164 N=324 (96. 7%) 2 -year N=163 (98. 2%) 1 subject consent withdrawn
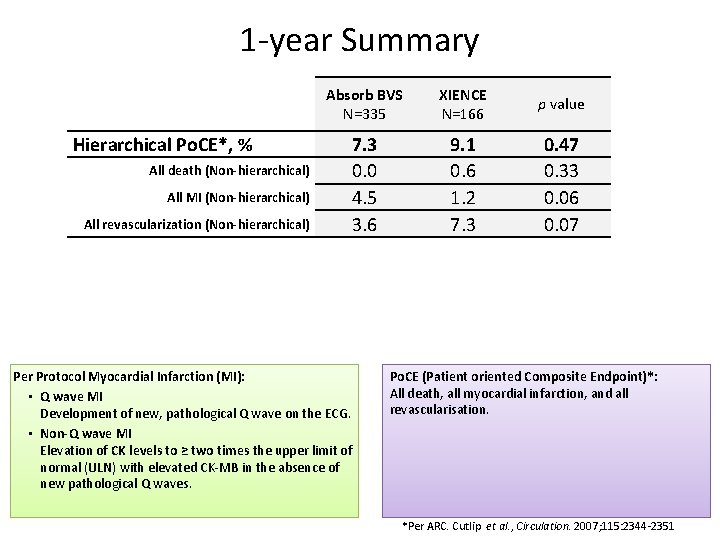
1 -year Summary Hierarchical Po. CE*, % All death (Non-hierarchical) All MI (Non-hierarchical) All revascularization (Non-hierarchical) Absorb BVS N=335 XIENCE N=166 p value 7. 3 0. 0 4. 5 3. 6 9. 1 0. 6 1. 2 7. 3 0. 47 0. 33 0. 06 0. 07 Per Protocol Myocardial Infarction (MI): • Q wave MI Development of new, pathological Q wave on the ECG. • Non-Q wave MI Elevation of CK levels to ≥ two times the upper limit of normal (ULN) with elevated CK-MB in the absence of new pathological Q waves. Po. CE (Patient oriented Composite Endpoint)*: All death, all myocardial infarction, and all revascularisation. *Per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
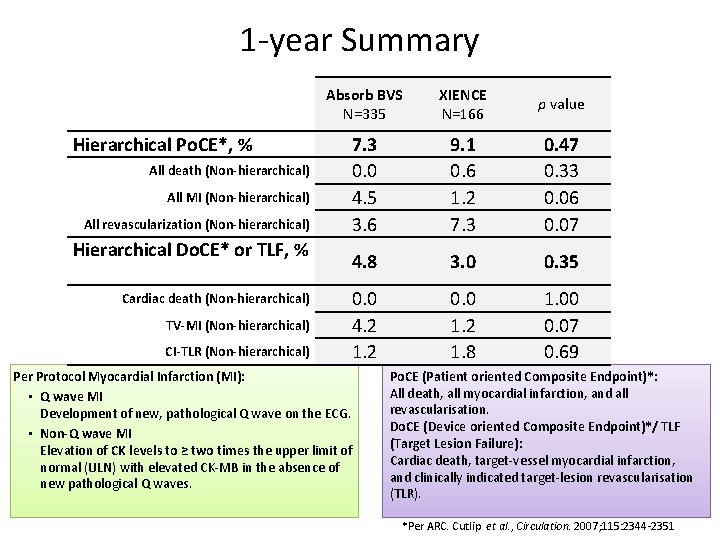
1 -year Summary Hierarchical Po. CE*, % All death (Non-hierarchical) All MI (Non-hierarchical) All revascularization (Non-hierarchical) Hierarchical Do. CE* or TLF, % Cardiac death (Non-hierarchical) TV-MI (Non-hierarchical) CI-TLR (Non-hierarchical) Absorb BVS N=335 XIENCE N=166 p value 7. 3 0. 0 4. 5 3. 6 9. 1 0. 6 1. 2 7. 3 0. 47 0. 33 0. 06 0. 07 4. 8 3. 0 0. 35 0. 0 4. 2 1. 2 0. 0 1. 2 1. 8 1. 00 0. 07 0. 69 Per Protocol Myocardial Infarction (MI): • Q wave MI Development of new, pathological Q wave on the ECG. • Non-Q wave MI Elevation of CK levels to ≥ two times the upper limit of normal (ULN) with elevated CK-MB in the absence of new pathological Q waves. Po. CE (Patient oriented Composite Endpoint)*: All death, all myocardial infarction, and all revascularisation. Do. CE (Device oriented Composite Endpoint)*/ TLF (Target Lesion Failure): Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR). *Per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
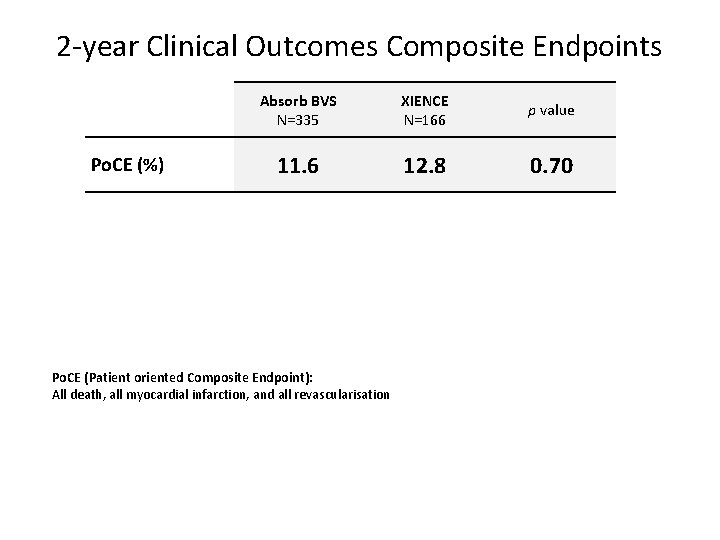
2 -year Clinical Outcomes Composite Endpoints Po. CE (%) Absorb BVS N=335 XIENCE N=166 p value 11. 6 12. 8 0. 70 Po. CE (Patient oriented Composite Endpoint): All death, all myocardial infarction, and all revascularisation
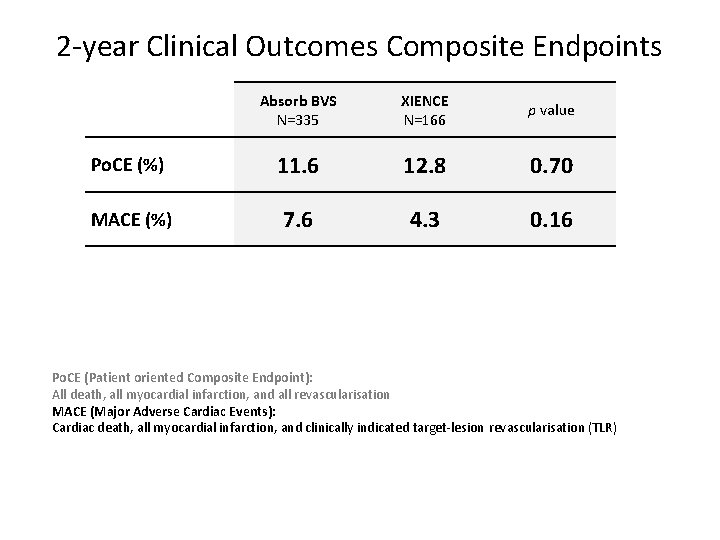
2 -year Clinical Outcomes Composite Endpoints Absorb BVS N=335 XIENCE N=166 p value Po. CE (%) 11. 6 12. 8 0. 70 MACE (%) 7. 6 4. 3 0. 16 Po. CE (Patient oriented Composite Endpoint): All death, all myocardial infarction, and all revascularisation MACE (Major Adverse Cardiac Events): Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation (TLR)
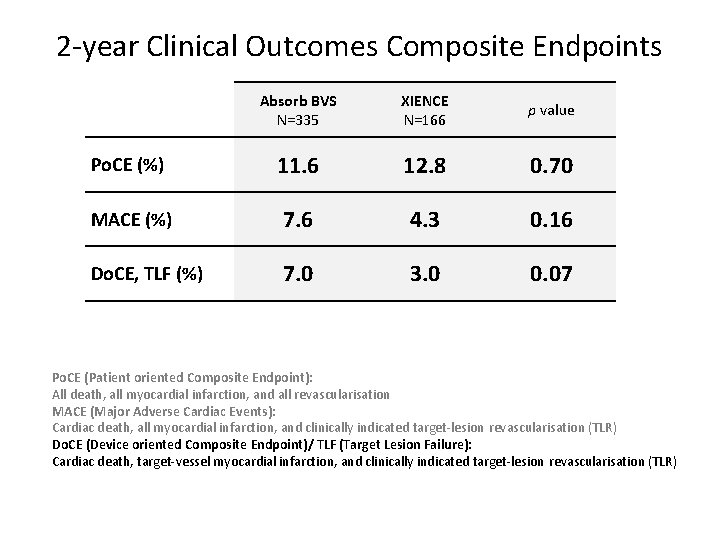
2 -year Clinical Outcomes Composite Endpoints Absorb BVS N=335 XIENCE N=166 p value Po. CE (%) 11. 6 12. 8 0. 70 MACE (%) 7. 6 4. 3 0. 16 Do. CE, TLF (%) 7. 0 3. 0 0. 07 Po. CE (Patient oriented Composite Endpoint): All death, all myocardial infarction, and all revascularisation MACE (Major Adverse Cardiac Events): Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation (TLR) Do. CE (Device oriented Composite Endpoint)/ TLF (Target Lesion Failure): Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR)
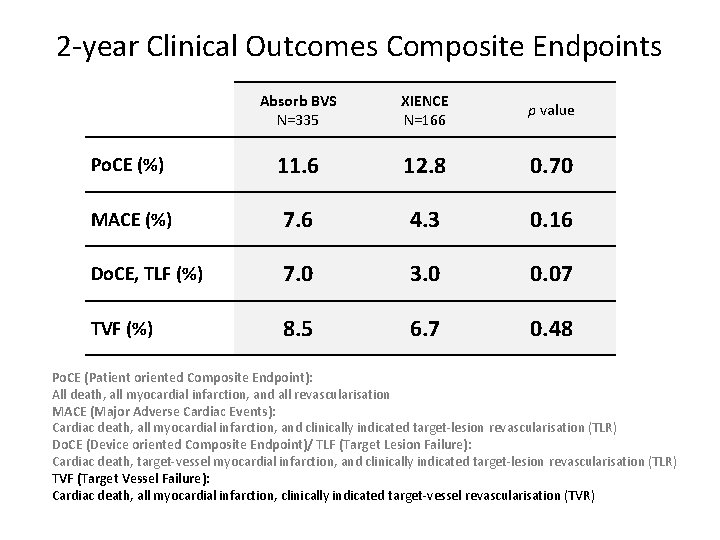
2 -year Clinical Outcomes Composite Endpoints Absorb BVS N=335 XIENCE N=166 p value Po. CE (%) 11. 6 12. 8 0. 70 MACE (%) 7. 6 4. 3 0. 16 Do. CE, TLF (%) 7. 0 3. 0 0. 07 TVF (%) 8. 5 6. 7 0. 48 Po. CE (Patient oriented Composite Endpoint): All death, all myocardial infarction, and all revascularisation MACE (Major Adverse Cardiac Events): Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation (TLR) Do. CE (Device oriented Composite Endpoint)/ TLF (Target Lesion Failure): Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR) TVF (Target Vessel Failure): Cardiac death, all myocardial infarction, clinically indicated target-vessel revascularisation (TVR)
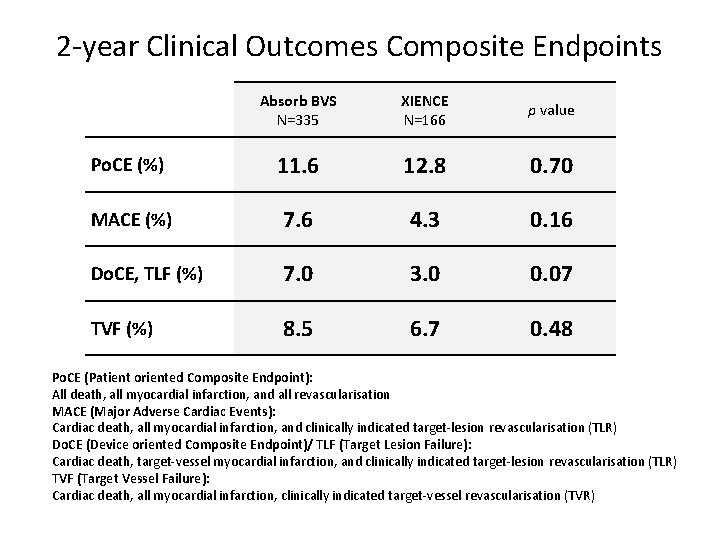
2 -year Clinical Outcomes Composite Endpoints Absorb BVS N=335 XIENCE N=166 p value Po. CE (%) 11. 6 12. 8 0. 70 MACE (%) 7. 6 4. 3 0. 16 Do. CE, TLF (%) 7. 0 3. 0 0. 07 TVF (%) 8. 5 6. 7 0. 48 Po. CE (Patient oriented Composite Endpoint): All death, all myocardial infarction, and all revascularisation MACE (Major Adverse Cardiac Events): Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation (TLR) Do. CE (Device oriented Composite Endpoint)/ TLF (Target Lesion Failure): Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR) TVF (Target Vessel Failure): Cardiac death, all myocardial infarction, clinically indicated target-vessel revascularisation (TVR)
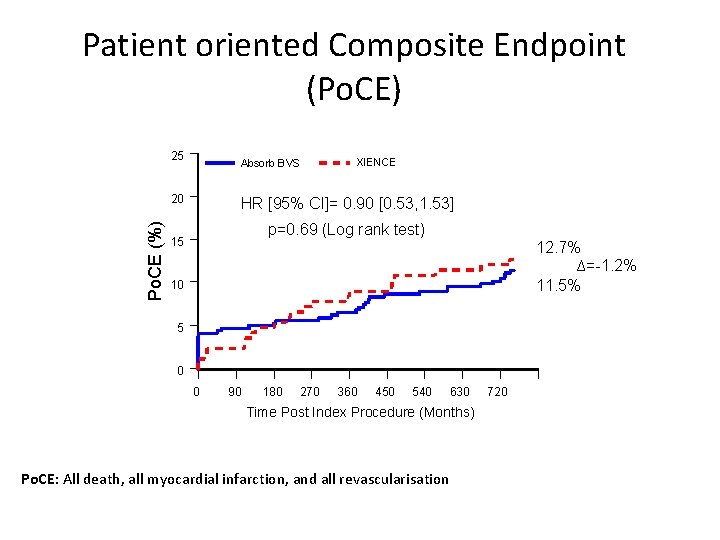
Patient oriented Composite Endpoint (Po. CE) 25 Absorb BVS Po. CE (%) 20 XIENCE HR [95% CI]= 0. 90 [0. 53, 1. 53] p=0. 69 (Log rank test) 15 12. 7% ∆=-1. 2% 11. 5% 10 5 0 0 90 180 270 360 450 540 630 Time Post Index Procedure (Months) Po. CE: All death, all myocardial infarction, and all revascularisation 720
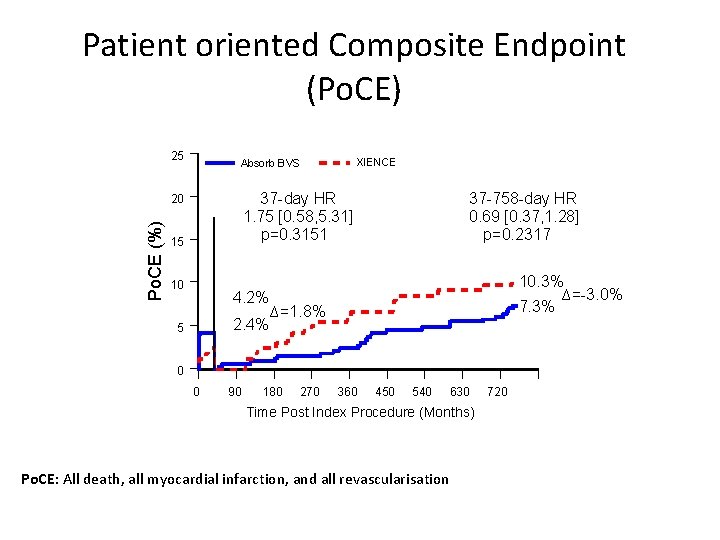
Patient oriented Composite Endpoint (Po. CE) 25 37 -day HR 1. 75 [0. 58, 5. 31] p=0. 3151 20 Po. CE (%) XIENCE Absorb BVS 15 10 4. 2% 2. 4% 5 37 -758 -day HR 0. 69 [0. 37, 1. 28] p=0. 2317 10. 3% ∆=-3. 0% 7. 3% ∆=1. 8% 0 0 90 180 270 360 450 540 630 Time Post Index Procedure (Months) Po. CE: All death, all myocardial infarction, and all revascularisation 720
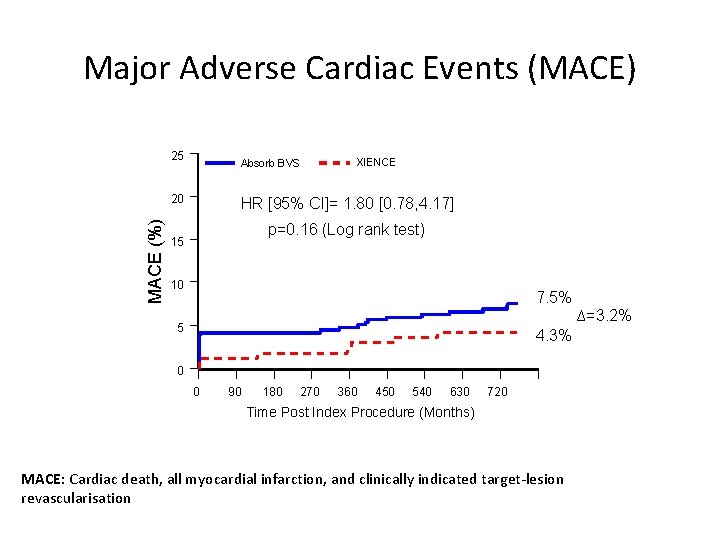
Major Adverse Cardiac Events (MACE) 25 Absorb BVS MACE (%) 20 XIENCE HR [95% CI]= 1. 80 [0. 78, 4. 17] p=0. 16 (Log rank test) 15 10 7. 5% ∆=3. 2% 5 4. 3% 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) MACE: Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation
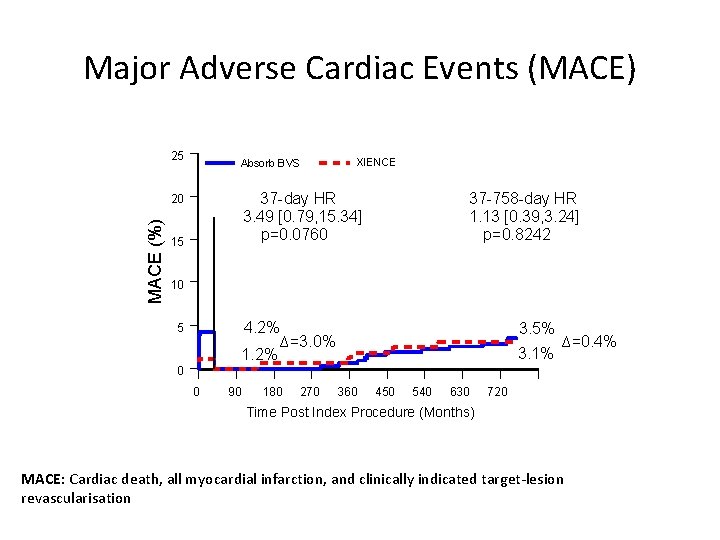
Major Adverse Cardiac Events (MACE) 25 Absorb BVS 37 -day HR 3. 49 [0. 79, 15. 34] p=0. 0760 20 MACE (%) XIENCE 15 37 -758 -day HR 1. 13 [0. 39, 3. 24] p=0. 8242 10 4. 2% ∆=3. 0% 1. 2% 5 3. 5% 3. 1% ∆=0. 4% 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) MACE: Cardiac death, all myocardial infarction, and clinically indicated target-lesion revascularisation
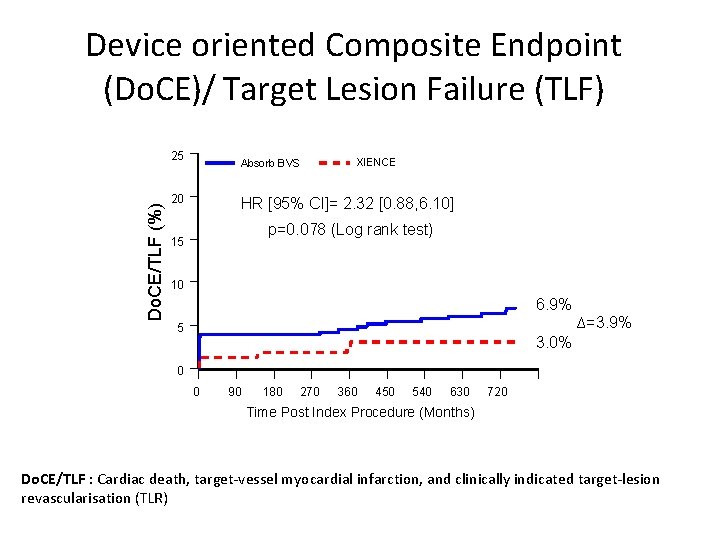
Device oriented Composite Endpoint (Do. CE)/ Target Lesion Failure (TLF) Do. CE/TLF (%) 25 Absorb BVS 20 XIENCE HR [95% CI]= 2. 32 [0. 88, 6. 10] p=0. 078 (Log rank test) 15 10 6. 9% ∆=3. 9% 5 3. 0% 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) Do. CE/TLF : Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR)
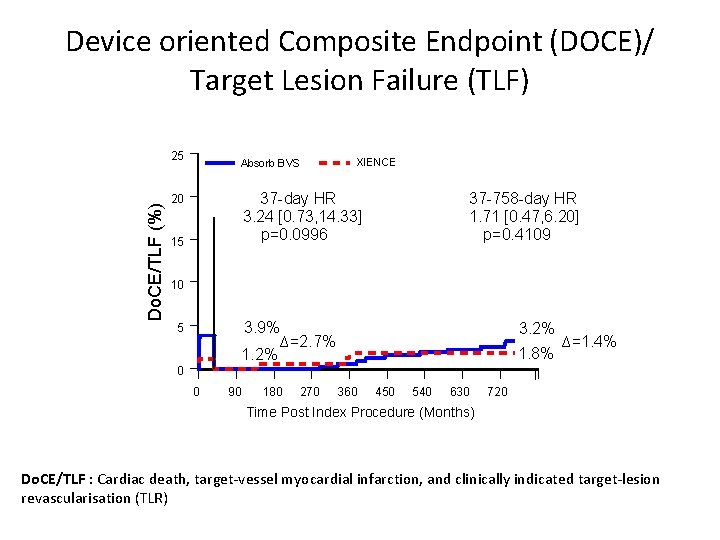
Device oriented Composite Endpoint (DOCE)/ Target Lesion Failure (TLF) Do. CE/TLF (%) 25 Absorb BVS XIENCE 37 -day HR 3. 24 [0. 73, 14. 33] p=0. 0996 20 15 37 -758 -day HR 1. 71 [0. 47, 6. 20] p=0. 4109 10 3. 9% ∆=2. 7% 1. 2% 5 3. 2% 1. 8% ∆=1. 4% 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) Do. CE/TLF : Cardiac death, target-vessel myocardial infarction, and clinically indicated target-lesion revascularisation (TLR)
![Target Vessel Failure (TVF) 25 Absorb BVS TVF (%) 20 XIENCE HR [95% CI]= Target Vessel Failure (TVF) 25 Absorb BVS TVF (%) 20 XIENCE HR [95% CI]=](http://slidetodoc.com/presentation_image/31288ed616b5363056fb29b791abb0ae/image-18.jpg)
Target Vessel Failure (TVF) 25 Absorb BVS TVF (%) 20 XIENCE HR [95% CI]= 1. 28 [0. 64, 2. 57] p=0. 49 (Log rank test) 15 10 8. 5% ∆=1. 8% 6. 7% 5 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) TVF : Cardiac death, all myocardial infarction, clinically indicated target-vessel revascularisation
![Target Vessel Failure (TVF) 25 37 -day HR 2. 33 [0. 67, 8. 10] Target Vessel Failure (TVF) 25 37 -day HR 2. 33 [0. 67, 8. 10]](http://slidetodoc.com/presentation_image/31288ed616b5363056fb29b791abb0ae/image-19.jpg)
Target Vessel Failure (TVF) 25 37 -day HR 2. 33 [0. 67, 8. 10] p=0. 1683 20 TVF (%) XIENCE Absorb BVS 15 37 -758 -day HR 0. 89 [0. 37, 2. 12] p=0. 7914 10 4. 2% ∆=2. 4% 1. 8% 5 4. 9% 4. 5% ∆=-0. 4% 0 0 90 180 270 360 450 540 630 720 Time Post Index Procedure (Months) TVF : Cardiac death, all myocardial infarction, clinically indicated target-vessel revascularisation
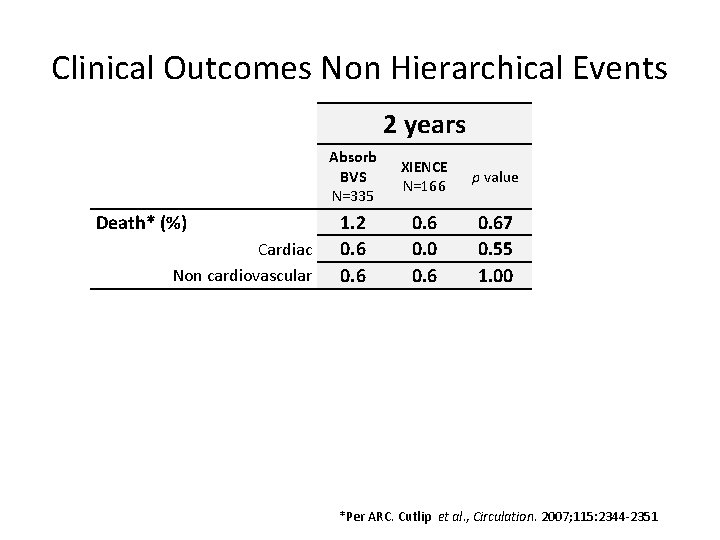
Clinical Outcomes Non Hierarchical Events 2 years Death* (%) Cardiac Non cardiovascular Absorb BVS N=335 XIENCE N=166 p value 1. 2 0. 6 0. 0 0. 67 0. 55 1. 00 *Per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
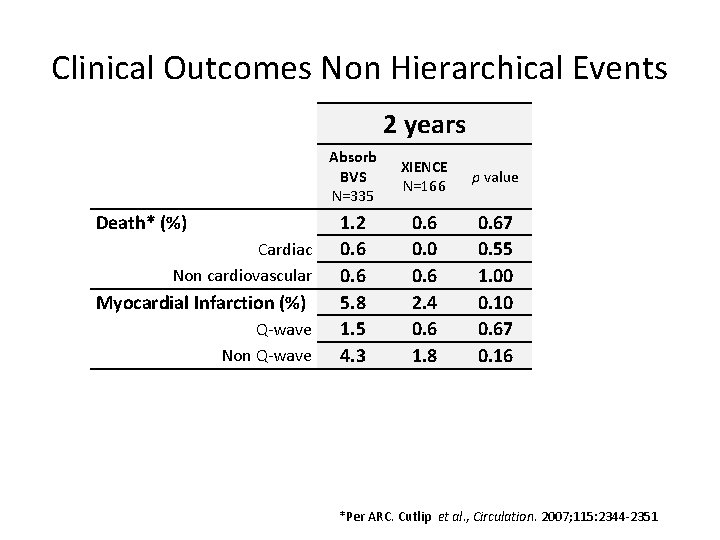
Clinical Outcomes Non Hierarchical Events 2 years Death* (%) Cardiac Non cardiovascular Myocardial Infarction (%) Q-wave Non Q-wave Absorb BVS N=335 XIENCE N=166 p value 1. 2 0. 6 5. 8 1. 5 4. 3 0. 6 0. 0 0. 6 2. 4 0. 6 1. 8 0. 67 0. 55 1. 00 0. 10 0. 67 0. 16 *Per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
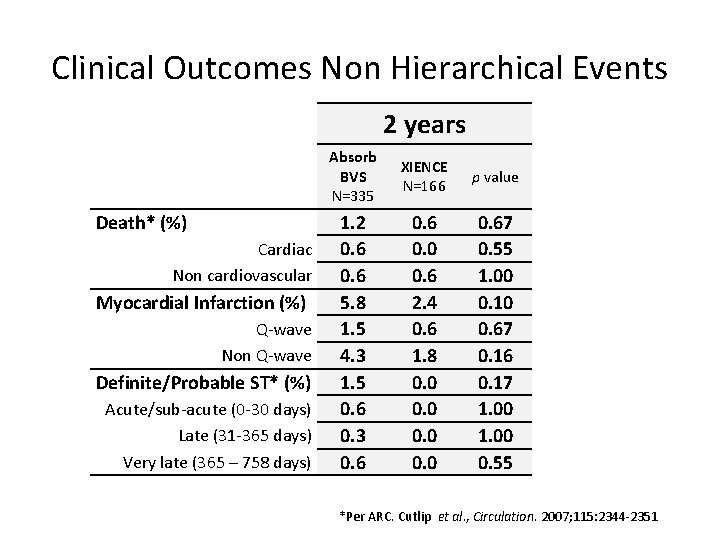
Clinical Outcomes Non Hierarchical Events 2 years Death* (%) Cardiac Non cardiovascular Myocardial Infarction (%) Q-wave Non Q-wave Definite/Probable ST* (%) Acute/sub-acute (0 -30 days) Late (31 -365 days) Very late (365 – 758 days) Absorb BVS N=335 XIENCE N=166 p value 1. 2 0. 6 5. 8 1. 5 4. 3 1. 5 0. 6 0. 3 0. 6 0. 0 0. 6 2. 4 0. 6 1. 8 0. 0 0. 67 0. 55 1. 00 0. 10 0. 67 0. 16 0. 17 1. 00 0. 55 *Per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
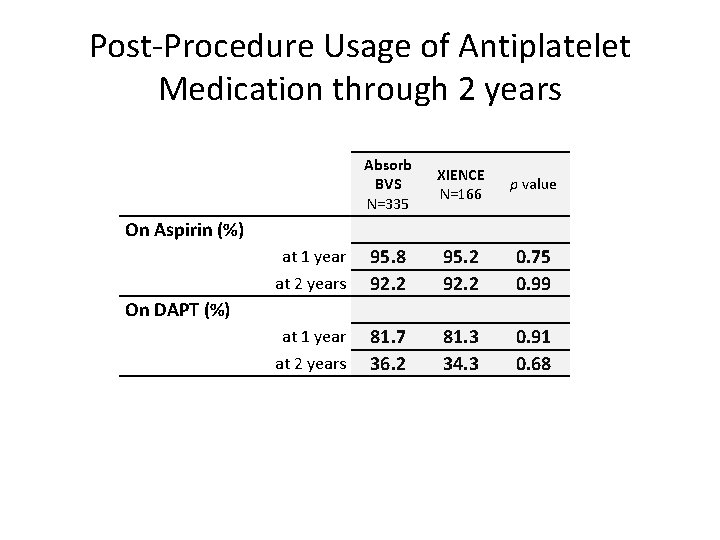
Post-Procedure Usage of Antiplatelet Medication through 2 years Absorb BVS N=335 XIENCE N=166 p value at 1 year at 2 years 95. 8 92. 2 95. 2 92. 2 0. 75 0. 99 at 1 year at 2 years 81. 7 36. 2 81. 3 34. 3 0. 91 0. 68 On Aspirin (%) On DAPT (%)

Very Late Scaffold Thrombosis Cases
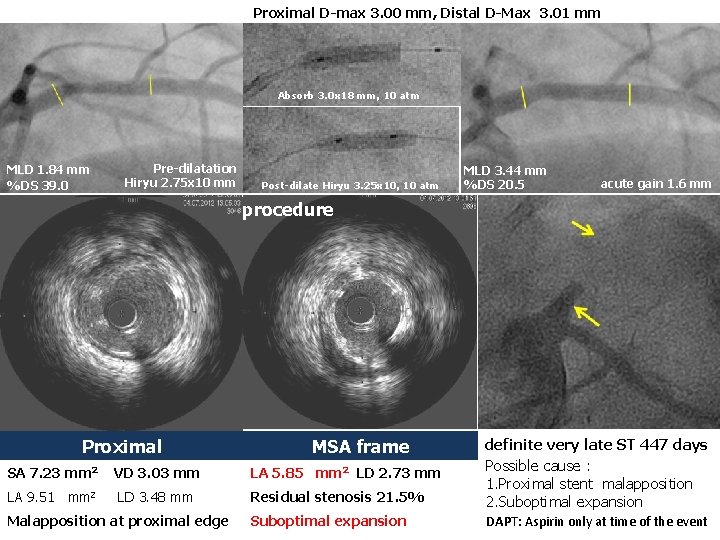
Proximal D-max 3. 00 mm, Distal D-Max 3. 01 mm Absorb 3. 0 x 18 mm, 10 atm MLD 1. 84 mm %DS 39. 0 Pre-dilatation Hiryu 2. 75 x 10 mm Post-dilate Hiryu 3. 25 x 10, 10 atm MLD 3. 44 mm %DS 20. 5 acute gain 1. 6 mm IVUS Post procedure Malapposed struts Proximal MSA frame LA 9. 51 mm 2 LD 3. 48 mm Residual stenosis 21. 5% definite very late ST 447 days Possible cause : 1. Proximal stent malapposition 2. Suboptimal expansion Malapposition at proximal edge Suboptimal expansion DAPT: Aspirin only at time of the event SA 7. 23 mm 2 VD 3. 03 mm LA 5. 85 mm 2 LD 2. 73 mm
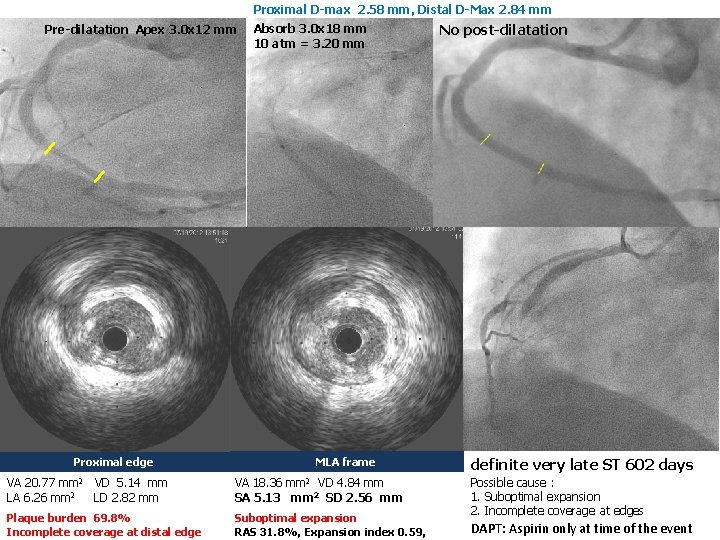
Proximal D-max 2. 58 mm, Distal D-Max 2. 84 mm Pre-dilatation Apex 3. 0 x 12 mm Absorb 3. 0 x 18 mm 10 atm = 3. 20 mm Proximal edge MLA frame VA 20. 77 mm 2 VD 5. 14 mm VA 18. 36 mm 2 VD 4. 84 mm Plaque burden 69. 8% Incomplete coverage at distal edge Suboptimal expansion RAS 31. 8%, Expansion index 0. 59, LA 6. 26 mm 2 LD 2. 82 mm SA 5. 13 mm 2 SD 2. 56 mm No post-dilatation definite very late ST 602 days Possible cause : 1. Suboptimal expansion 2. Incomplete coverage at edges DAPT: Aspirin only at time of the event
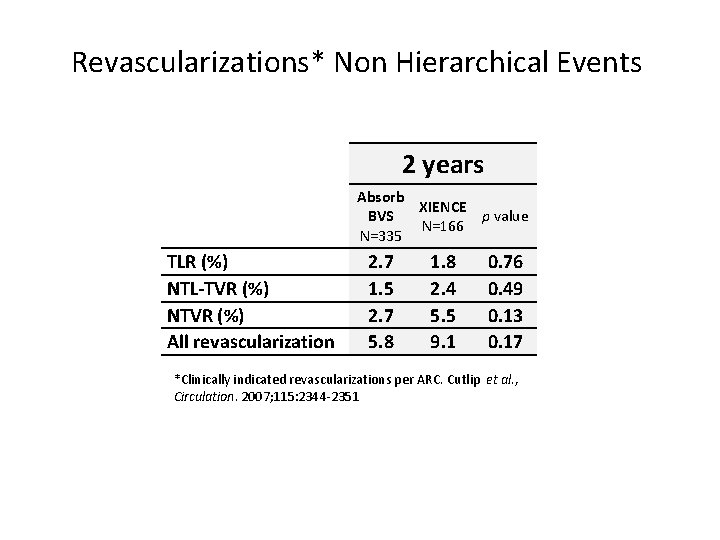
Revascularizations* Non Hierarchical Events 2 years Absorb XIENCE BVS p value N=166 N=335 TLR (%) NTL-TVR (%) NTVR (%) All revascularization 2. 7 1. 5 2. 7 5. 8 1. 8 2. 4 5. 5 9. 1 0. 76 0. 49 0. 13 0. 17 *Clinically indicated revascularizations per ARC. Cutlip et al. , Circulation. 2007; 115: 2344 -2351
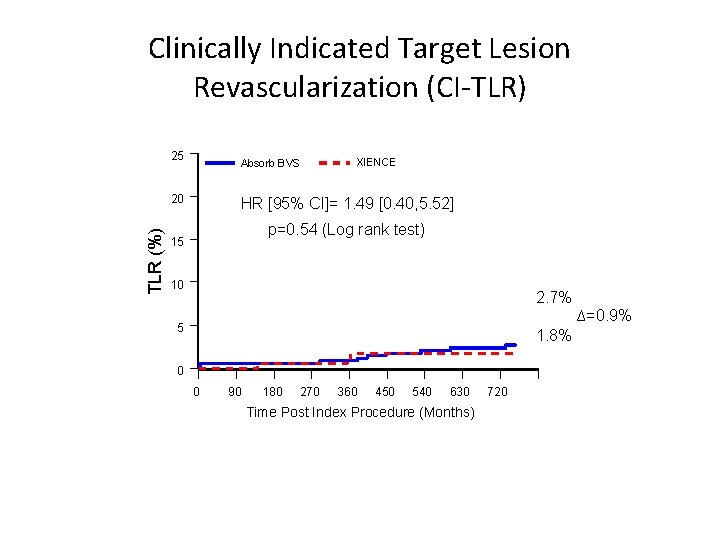
Clinically Indicated Target Lesion Revascularization (CI-TLR) 25 Absorb BVS TLR (%) 20 XIENCE HR [95% CI]= 1. 49 [0. 40, 5. 52] p=0. 54 (Log rank test) 15 10 2. 7% ∆=0. 9% 5 1. 8% 0 0 90 180 270 360 450 540 630 Time Post Index Procedure (Months) 720

Limitations • The ABSORB II study was not powered for clinical endpoints • The 2 -year endpoint represents a non pre-specified interim analysis • Investigators long experience with XIENCE as compared to Absorb BVS might have impacted the results

Conclusions • At 2 years there were no significant differences in the clinical outcomes between the two arms: – Po. CE (all death, all MI and all revascularization) Absorb BVS: 11. 6% vs XIENCE: 12. 8%, p=0. 70 – Do. CE/TLF (cardiac death, TV-MI and TLR) Absorb BVS: 7. 0% vs XIENCE: 3. 0%, p=0. 07 • The exploratory observations presented in this report are hypothesis generating and need to be confirmed in larger randomized trials such as ABSORB III
- Slides: 30