THANK YOU AETC Education and Training Center for
























































- Slides: 71

THANK YOU! AETC Education and Training Center for your support of the NJ HIV COORDINATOR’S MEETING PLEASE COMPLETE THE EVALUATION FORM !! IN YOUR FOLDER!!

NJ HIV COORDINATORS MEETING May 16, 2017 New Jersey Hospital Association

Afternoon Agenda: Rutgers - RWJMS • INTRODUCTION: Dr. Martin • • • NJ HIV Map – 2017 Update REGULATORY Update: A Changing Landscape: Dr. Salaru • Clinical Laboratory Licensure – CHANGE IN NJ REQUIREMENTS • The HIV Quality Assurance Program – NO CHANGE • PROGRAM Update – Drs. Martin • REFRESHER: The NJ Rapid Test Algorithm and Linkage To Care • • Determine Combo: Year 2 • • RWJ – Administrative Program Logistics – Joanne Corbo • Cicatelli Associates – Beth Hurley • Sustainable Billing – Billing Survey Starts Today 2016 -7 Initiatives: • Pooled NAAT QA project HIV/Syphilis Screening Pilot HIV/HCV Screening Pilot INSTI Pilot What’s coming?

AFTERNOON INTRODUCTIONS Eugene Martin, Ph. D. Co-Director, NJ HIV

NJ HIV - Rapid Testing Support RUTGERS – RWJMS Eugene Martin, Ph. D. Co-Director NJ HIV , PI - DHSTS grant Gratian Salaru, MD 2016 DISCORDANT Co-Director NJ HIV’ PI - DMHAS MOA Parisa Javidian, MD - BLD Tom Kirn, MD, Ph. D. Joanne Corbo, MT (ASCP), MBA Rapid HIV Program Manager Latasha Adams BS, MT Moeen Ahmed BS, MT Aida Gilanchi, BS, MT Nisha Intwala, BS, MT (ASCP) Franchesca Jackson, BS DAS Mobile HIV Counselor • Marianela Moreno Administration: • Lisa May • Karen Williams Aida Gilanchi, BS, MT 21 SOLO DISCORDANTS

NJ HIV: Rapid DHSTS HIV Testing Sites RWJMS Venue Stationary Mobile Vans 13 Totals CBO 24 37 CLINICS 6 6 CORRECTIONS 5 5 FQHC 21 2 23 HEALTH DEPT 20 3 23 HOSPITALS 2 2 HIGH SCHOOLS 1 1 UNIVERSITIES 3 3 SUBSTANCE ABUSE 3 3 85 18 103 CBO 1 1 2 CORRECTIONS 1 FQHC 15 HEALTH DEPT 3 HOSPITALS 14 HIGH SCHOOLS 0 0 UNIVERSITIES 2 2 SUBSTANCE ABUSE FAMILY PLANNING 0 4 0 0 4 44 125 22 147 NON-RWJMS 1 1 16 3 2 16 4/28/2017 NEW JERSEY (ALL SITES)

REGULATORY Update: A Changing Landscape Gratian Salaru, MD Co-Director, NJ HIV

Regulatory oversight: RUTGERS -RWJMS • CURRENTLY • CLIA Certificate held by Rutgers - RWJMS (Parisa Javidian, MD) for all “RWJMS” sites: 103 • CLIA Certificate held by Others – 44 • On January 9, 2017, Governor Christie signed into law amendments to the New Jersey Clinical Laboratory Improvement Act (Act), N. J. S. A. 45: 9 -42. 26 et seq. See New P. L. 2016, Chapter 86, Senate, No. 976. The amendments to the Act took effect immediately upon enactment. • WHAT’S CHANGED?

NJ CLIS Guidance and its significance • NJ Clinical Lab Improvement Service (CLIS) Laboratory License is no longer required from sites performing POC tests with an existing CLIA Certificate (Waived or Mod. Complexity) • • No significant impact on our ability to support and sustain rapid ID screening • Competency Assessment – Provided by our Med. Tech. liaisons and is the responsibility of CLIA laboratory director • QC requirements are part of test procedures Proficiency Testing Program – Internalized the review and corrective action decisions regarding performance failures.

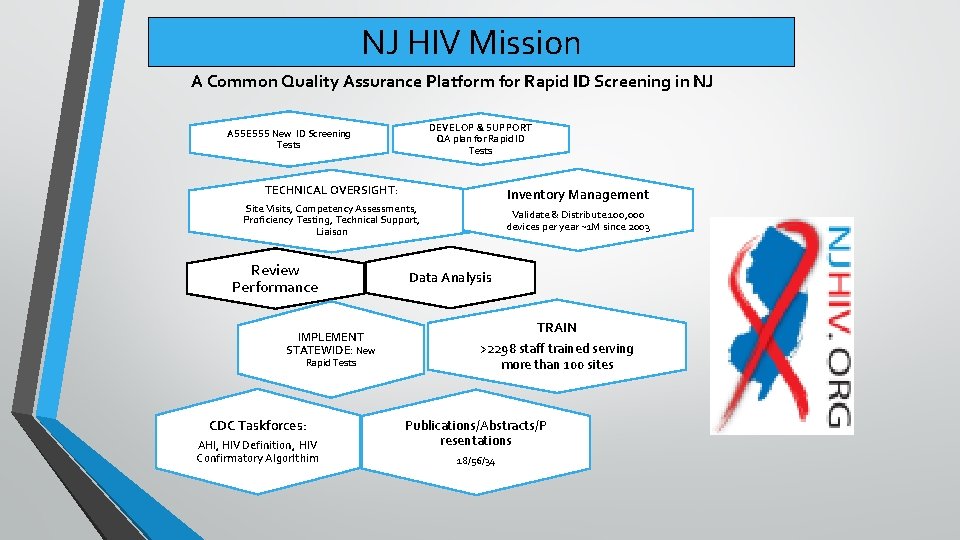
NJ HIV Mission A Common Quality Assurance Platform for Rapid ID Screening in NJ DEVELOP & SUPPORT QA plan for Rapid ID Tests ASSESSS New ID Screening Tests TECHNICAL OVERSIGHT: Inventory Management Site Visits, Competency Assessments, Proficiency Testing, Technical Support, Liaison Review Performance IMPLEMENT STATEWIDE: New Rapid Tests CDC Taskforces: AHI, HIV Definition, HIV Confirmatory Algor. Ithim Validate & Distribute 100, 000 devices per year ~1 M since 2003 Data Analysis TRAIN >2298 staff trained serving more than 100 sites Publications/Abstracts/P resentations 18/56/34

NJ HIV Quality Assurance Program • • • Procedures for infectious disease screening • • Develop and oversee QC procedures for all DHSTS sites Acquisition of inventory for all DHSTS sites Reagent lot-to-lot validation utilizing side-by-side comparison with mixed titer performance panels Competency Assessment (6, 12 month): (elements) Proficiency Testing Program NJ RTA program • • Statewide Discordant Oversight Data acquisition on behalf of DHSTS

WHAT IS IT GOING TO MEAN? Walgreens Healthcare Clinics to Offer: Testing Services for STIs: Including HIV, Hepatitis and Others • Walgreens Healthcare Clinics in: Cincinnati, Cleveland, Columbus (Ohio), Dallas, Denver, Houston, Kansas City, Knoxville, Las Vegas, Louisville, Memphis, Nashville, Orlando, Philadelphia, Phoenix, Tucson, Washington D. C. , and Wichita to offer testing services for (STIs), • Clinics in Houston prescribing Pr. EP for HIV prevention April 25 • “Ongoing collaboration” with health departments and local AIDS service organizations to help provide free HIV testing as part of an ongoing effort to broaden the reach of HIV information and testing in non-traditional settings and to support local organizations’ outreach efforts. • • • How will this impact our efforts • Working together… What does it mean exactly? Testing vs. Screening Will DHSTS be competing with Walgreen’s?

ADMINISTRATIVE ISSUES: Joanne Corbo – NJ HIV Program Manager

Website for NJ HIV Rapid Testing Support: njhiv 1. org

Change In Regulations • • Sites Under the Oversight of RWJMS Day to Day Operations Do Not Change • All Quality Assurance Activities Continue • Training, Monthly visits, Recertification and Proficiency Testing Test Under the RWJ CLIA Certificate (no NJ license required) • • • CLIA Certificate in testing area Procedure Manual in Testing Area Standing order is still needed

Test Logs • RWJ test logs are due the 10 th of the month • They may also be sent as they are completed • Please make sure logs are complete: • Site Number, Contact Information, shipment number • Test information complete: Pos, Neg, Temperature, Start Time End Time, Operator Initials • If doing second test for another site indicate second test and site number of first site • Fax to 732 -235 -9012 or 732 -743 -3632

Supplies • SUPPLY ORDERS For ALL Sites are based on average monthly test • • volume (basically a Standing Order) SUPPLY ORDERS For ALL Sites Not Under the RWJ License • Will be filled based on average testing volume • Will be delivered the last two weeks of the month • If anything is need above the normal or if you need to decrease your order let us know prior to the 15 th of the month. • We pay for each delivery so please help us be cost efficient Call Lisa May at 732 -743 -3624 or Karen Williams at 732 -743 -3630 for any additional items or special situations requiring additional supplies. All orders must be approved by management before delivery

Forms/Protocols • New NJ Positive Tracking form- In Packet • • Use new form included in packet (available on NJ HIV. org) Must be sent in as completed to RWJ Fax to 732 -235 -9012 or 732 -743 -3632 Do NOT Fax Evaluation WEB Forms to RWJ. WE can only accept de-identified information • R 2 R Protocol Updated- Will be emailed

One Time Testing Events • • DHSTS re-examined the value of One Time Event testing After DHSTS and RWJ review of the results of OTEs • Rarely if ever yield the desires outcome of finding a positive test result and linking that patient immediately to care. As of January 1, 2017, unless an OTE can be justified by a recent prior event at the same venue wherein at least one positive test result was obtained, or it is part of a National Testing Awareness Campaign or similar annually recurring event (e. g. , World AIDS Day, National HIV Testing Day, National Black HIV/AIDS Day, Gay Pride events, National Latino HIV Day) it will no longer be supported and such requests automatically denied by RWJ. To increase collaboration and maximize all available resources sites that want to test on a one-time-only basis • Should work collaboratively with other local agencies to utilize one of the many mobile HIV testing vans designed for this type of outreach testing. • Your agency would invite another agency with their van and their van testing staff to the event in question where both agencies would be co-participating in the event.

New Testing Sites/ Outreach • All New Testing sites and Outreach locations need to be approved by DHSTS • Contact Joanne Corbo or your PMO for application or questions

Discordant Work Up Discordant work up/ procedure: • If second rapid or confirmatory does not match first rapid the result is discordant • Draw blood for work up: • Two white top tubes (must be spun down and frozen) • No longer have to freeze upside down • Two gold top serum separators (must be spun down and refrigerated) • You must report all discordant results to RWJ • Call Discordant Hotline (Usually Dr. Martin) 732 -236 -7013. • Leave a message with contact information so RWJ can pick up samples and process ASAP.

Changes to Alere Determine

New Alere Determine Control Pipette • Added fill line indicator on outside of pipette

New Alere Determine Control Pipette • Using a disposable pipette to draw up control solution into pipette. • In a vertical position touch the disposable pipette to the sample pad and depress the bulb on the disposable pipette to release the control reagent into the Sample Pad • Do not use Chase Buffer with control reagents! • Start timer and read results between 20 and 30 minutes • Dispose of disposable pipette in bio hazardous waste.

Alere Determine Label Changes • Alere moved manufacturing of Determine from Israel to Scarborough, Maine in April 2017 • We will begin to see this product in June • NO CHANGE in product • Minor Label Change to reflect the new manufacturing location

CURRENT Alere Determine Label • Shows Manufacturing location: Orgenics Ltd. in Israel • Expiration date and Lot number

NEW Alere Determine Label • Shows New Manufacturing location: Alere in Scarborough, Maine • Expiration date and Lot Number and Date of manufacture LOT EXPIRATION DATE LOT NUMBER MANUFACTURE DATE

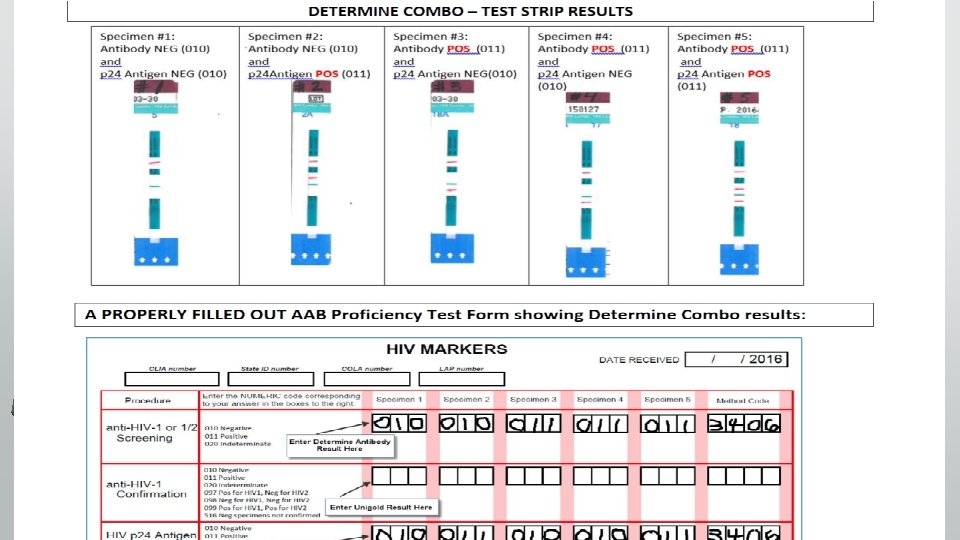
AAB Proficiency Testing Survey • Will be shipped TODAY to RWJ sites for delivery Wednesday or • • • Thursday Results must be sent to RWJ by Wednesday May 24 th Do not enter results online. FAX RESULTS TO YOUR LIAISONat 732 -2359012 • RWJ will enter results and submit them Take care when filling out results form. Look at the test strips when entering results. Avoid cross outs on the results form. We know the form is confusing. Please call us with questions. We have developed a guide to assist you. Proficiency tests are critically important. Failure of Proficiency testing could result in your site not being able to test.

NEW FORM - AAB Proficiency Testing Survey • New Form With bubble entry for positive or negative results

Proficiency Testing Guide

NJ HIV Rapid Testing Support Administrative Issues Proficiency Testing For RWJ Licensed Sites 31

Questions?

Sustainable Billing: The Billing Survey - We Need Your Help! Beth Hurley Cicatelli Associates

Capacity Building Assistance Program for High-Impact HIV Prevention Beth Hurley, MPH Project Director CAI

CBA Program for High Impact HIV Prevention Overview: • CAI, through the CDC’s High Impact HIV Prevention Project, provides capacity building assistance (CBA) related to high-impact HIV prevention for healthcare organizations (HCOs) Goals for HCOs: • Strengthen HCO and clinical setting’s systems to raise HIV detection and provide HIV testing support services • Provide HCO staff with improved methods of HIV test counseling

CBA Services: Examples of Trainings and TA for HIV Testing • HIV Screening in Key Populations - Targeting Strategically • New HIV Testing Technologies • Enhanced HIV Outreach Strategy • HIV Couples Testing • Partner Notification • Integration of Peer Support into Multidisciplinary Teams

Billing for HIV Testing: Capacity Assessment • Stay ahead of transforming healthcare environment • Understand agency’s structure for facilitating reimbursement of HIV testing services • Help us help you! Data informed case for continued HIV testing funding

Due JUNE th 7 !

Program Update Eugene Martin, Ph. D.

Rapid HIV Testing: The epidemic is changing. How are we responding?

It’s not just a question of how sensitive a test is… it’s also a question of how often we test, how well we test, how effectively we link the affected into care, and how well we retain clients in care… BACKGROUND & Perspective 41

Effective screening IS NOT definitive diagnosis DIAGNOSIS • GOAL: Definitively determine who is affected and who is not • • • SCREENING • GOAL: Identify who is likely to be affected and rapidly link them to care for follow-up Sensitivity of the assay Specificity of the assay No limits • • Take however long it takes to arrive at the correct diagnosis. Spare no expense • • • Minimize unnecessary referrals • Personalize the linkage to keep folks connected Maximize the rate of linkage into care Orthogonal confirmation (RTA) • • • Presumptive diagnosis Minimize missed calls Likely diagnosis (<1: 200 are wrong) 42

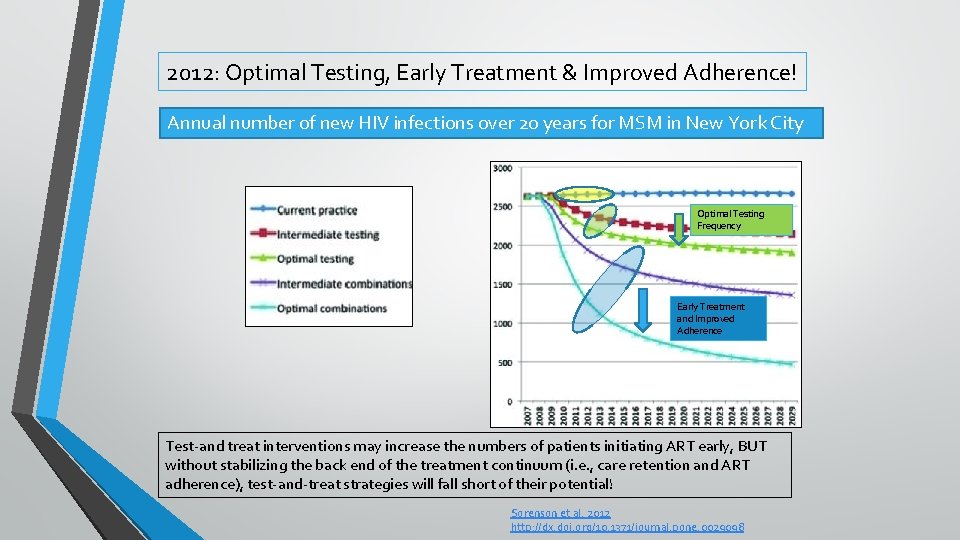
2012: Optimal Testing, Early Treatment & Improved Adherence! Annual number of new HIV infections over 20 years for MSM in New York City Optimal Testing Frequency Early Treatment and Improved Adherence Test-and treat interventions may increase the numbers of patients initiating ART early, BUT without stabilizing the back end of the treatment continuum (i. e. , care retention and ART adherence), test-and-treat strategies will fall short of their potential! Sorenson et al. 2012 http: //dx. doi. org/10. 1371/journal. pone. 0029098

US HIV Incidence and Transmission Goals, 2020 & 2025 Goal for 2020: 90/90/90. Incidence 21, 000 Goal for 2025: 95/95/95. Incidence 12, 000 would achieve tipping point with mortality dropping below 2020

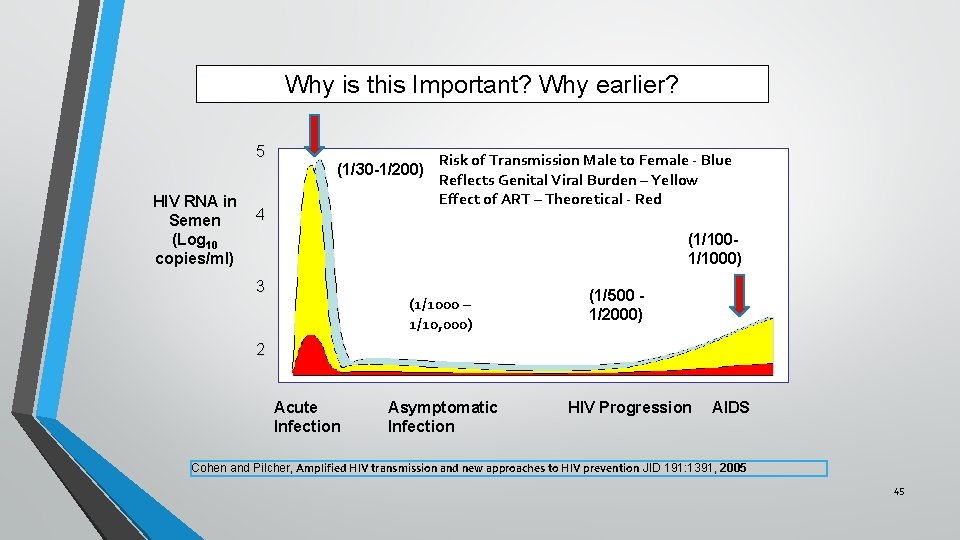
Why is this Important? Why earlier? 5 HIV RNA in Semen (Log 10 copies/ml) (1/30 -1/200) 4 Risk of Transmission Male to Female - Blue Reflects Genital Viral Burden – Yellow Effect of ART – Theoretical - Red (1/1000) 3 (1/1000 – 1/10, 000) (1/500 1/2000) 2 Acute Infection Asymptomatic Infection HIV Progression AIDS Cohen and Pilcher, Amplified HIV transmission and new approaches to HIV prevention JID 191: 1391, 2005 45

Post-testing information for persons seeking HIV testing after a recent exposure Evaluated the intervals between reactivity of the Aptima HIV-1 RNA test (Aptima) and 20 US Food and Drug Administration-approved HIV immunoassays using 222 longitudinally collected plasma specimens from HIV-1 seroconverters from the United States. https: //www. ncbi. nlm. nih. gov/pubmed/27737954

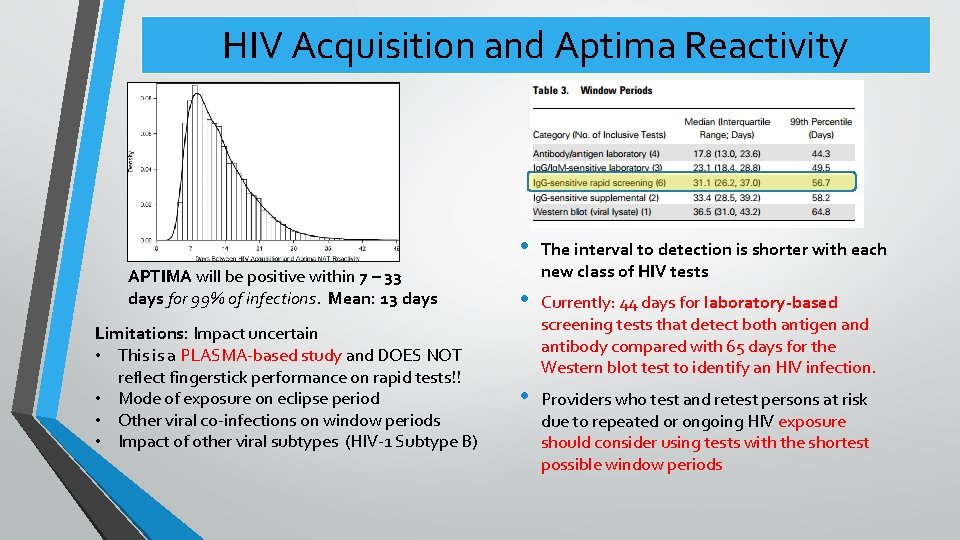
HIV Acquisition and Aptima Reactivity APTIMA will be positive within 7 – 33 days for 99% of infections. Mean: 13 days Limitations: Impact uncertain • This is a PLASMA-based study and DOES NOT reflect fingerstick performance on rapid tests!! • Mode of exposure on eclipse period • Other viral co-infections on window periods • Impact of other viral subtypes (HIV-1 Subtype B) • The interval to detection is shorter with each new class of HIV tests • Currently: 44 days for laboratory-based screening tests that detect both antigen and antibody compared with 65 days for the Western blot test to identify an HIV infection. • Providers who test and retest persons at risk due to repeated or ongoing HIV exposure should consider using tests with the shortest possible window periods

DISCORDANT Work-UP DAYS AFTER THE APTIMA TURNS POSITIVE! BUT REMEMBER: PLASMA NOT FINGERSTICK!! AND IT DOESN’T COUNT THE VARIABLE ECLIPSE PERIOD Current Initial Rapid Screen Current 2 nd Confirmatory RTA Initial Screen 2007 -2015 Initial Screen 2003 -2016 Original Confirmatory Assay

PROGRAM REVIEW & UPDATES 49

The second year with Determine Combo 50

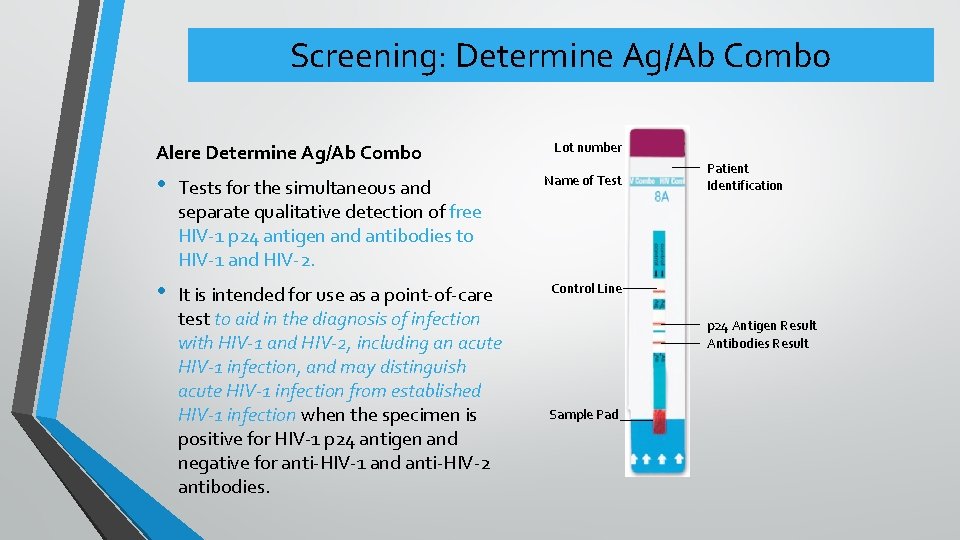
Screening: Determine Ag/Ab Combo Alere Determine Ag/Ab Combo • Tests for the simultaneous and separate qualitative detection of free HIV-1 p 24 antigen and antibodies to HIV-1 and HIV-2. • It is intended for use as a point-of-care test to aid in the diagnosis of infection with HIV-1 and HIV-2, including an acute HIV-1 infection, and may distinguish acute HIV-1 infection from established HIV-1 infection when the specimen is positive for HIV-1 p 24 antigen and negative for anti-HIV-1 and anti-HIV-2 antibodies. Lot number Name of Test Patient Identification Control Line p 24 Antigen Result Antibodies Result Sample Pad

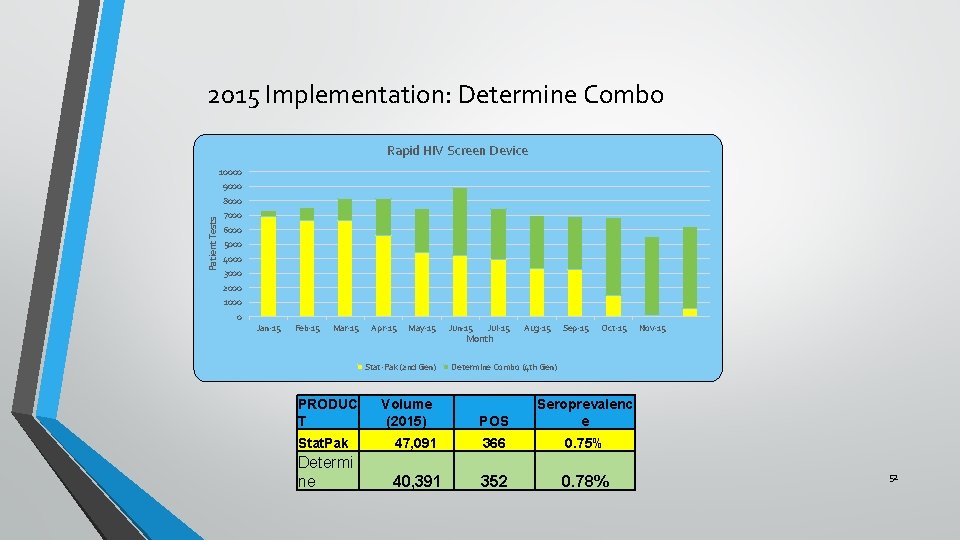
2015 Implementation: Determine Combo Rapid HIV Screen Device 10000 9000 Patient Tests 8000 7000 6000 5000 4000 3000 2000 1000 0 Jan-15 Feb-15 Mar-15 Apr-15 May-15 Stat-Pak (2 nd Gen) PRODUC T Volume (2015) Jun-15 Jul-15 Month Aug-15 Sep-15 Oct-15 Nov-15 Determine Combo (4 th Gen) POS Seroprevalenc e Stat. Pak 47, 091 366 0. 75% Determi ne 40, 391 352 0. 78% 52

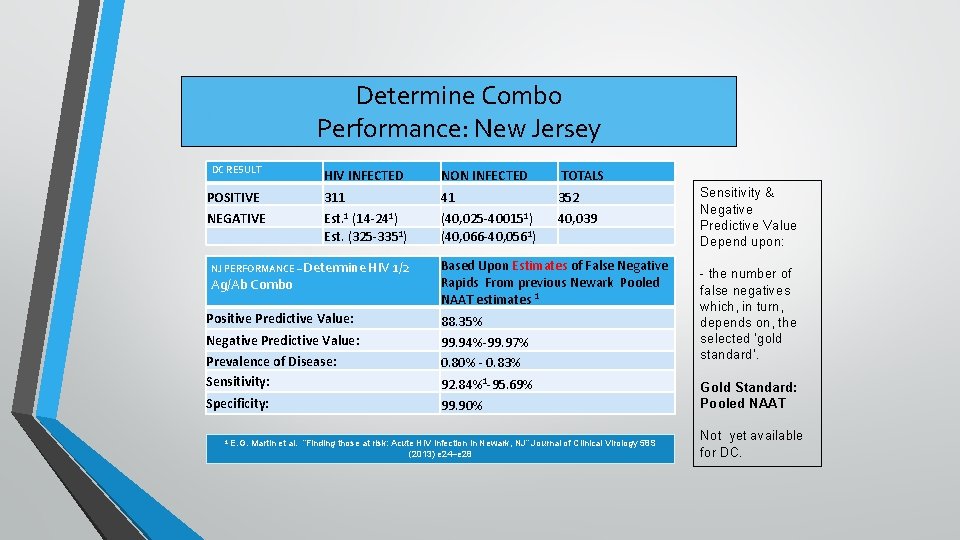
Determine Combo Performance: New Jersey DC RESULT HIV INFECTED NON INFECTED TOTALS POSITIVE 311 41 352 NEGATIVE Est. 1 (14 -241) Est. (325 -3351) (40, 025 -400151) (40, 066 -40, 0561) 40, 039 NJ PERFORMANCE – Determine HIV 1/2 Ag/Ab Combo Based Upon Estimates of False Negative Rapids From previous Newark Pooled NAAT estimates 1 Positive Predictive Value: 88. 35% Negative Predictive Value: Prevalence of Disease: Sensitivity: 99. 94%-99. 97% 0. 80% - 0. 83% Specificity: 99. 90% 1 E. G. 92. 84%1 -95. 69% Martin et al. “Finding those at risk: Acute HIV infection in Newark, NJ” Journal of Clinical Virology 58 S (2013) e 24–e 28 Sensitivity & Negative Predictive Value Depend upon: - the number of false negatives which, in turn, depends on, the selected ‘gold standard’. Gold Standard: Pooled NAAT Not yet available for DC.

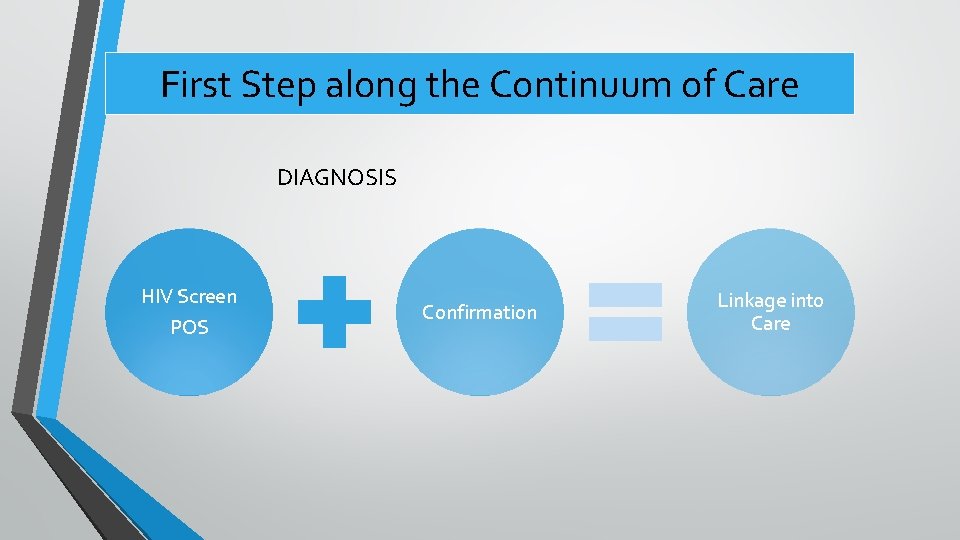
First Step along the Continuum of Care DIAGNOSIS HIV Screen POS Confirmation Linkage into Care

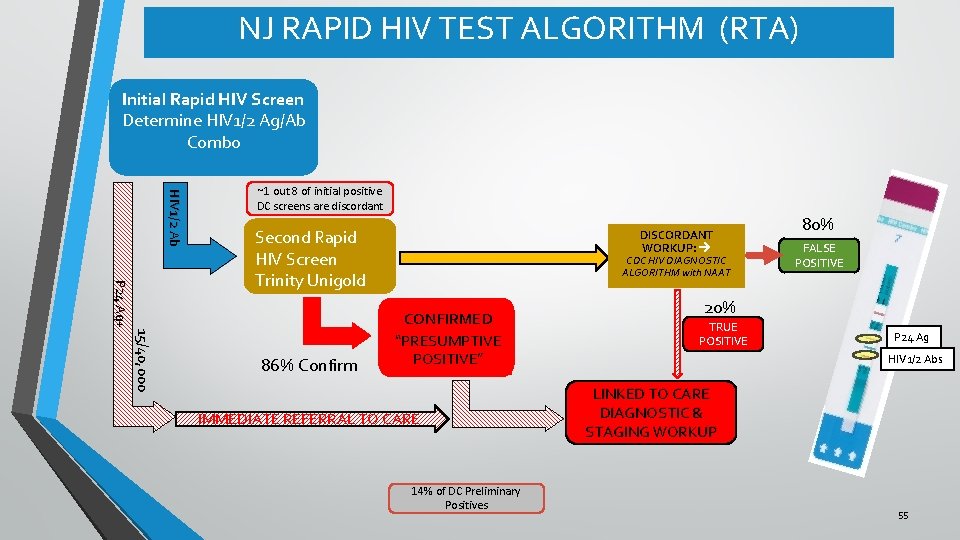
NJ RAPID HIV TEST ALGORITHM (RTA) Initial Rapid HIV Screen Determine HIV 1/2 Ag/Ab Combo HIV 1/2 Ab ~1 out 8 of initial positive DC screens are discordant 15/40, 000 P 24 Ag+ Second Rapid HIV Screen Trinity Unigold 86% Confirm DISCORDANT WORKUP: CDC HIV DIAGNOSTIC ALGORITHM with NAAT CONFIRMED “PRESUMPTIVE POSITIVE” IMMEDIATE REFERRAL TO CARE 14% of DC Preliminary Positives 80% FALSE POSITIVE 20% TRUE POSITIVE P 24 Ag HIV 1/2 Abs LINKED TO CARE DIAGNOSTIC & STAGING WORKUP 55

2016 DETERMINE COMBO PERFORMANCE • • NJ SUMMARY (2016) 80, 405 individuals screened 609 preliminary positive 524 IMMEDIATELY confirmed by RTA (86%) (14%) DISCORDANT with second rapid THE ROLE OF CREDIBLE REFERRALS - 2016 -

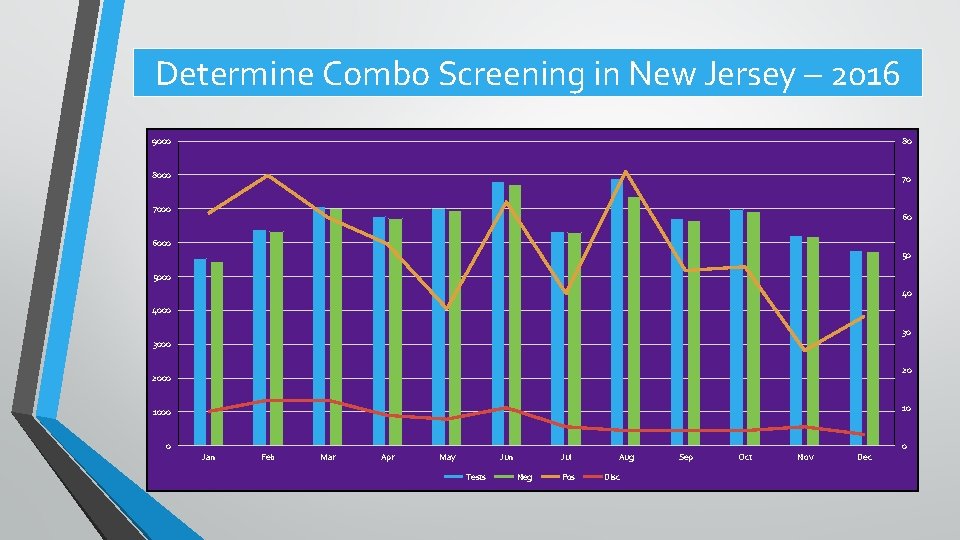
Determine Combo Screening in New Jersey – 2016 9000 80 8000 70 7000 60 6000 50 5000 40 4000 30 3000 20 2000 10 1000 0 Jan Feb Mar Apr May Jun Tests Jul Neg Pos Aug Disc Sep Oct Nov Dec 0

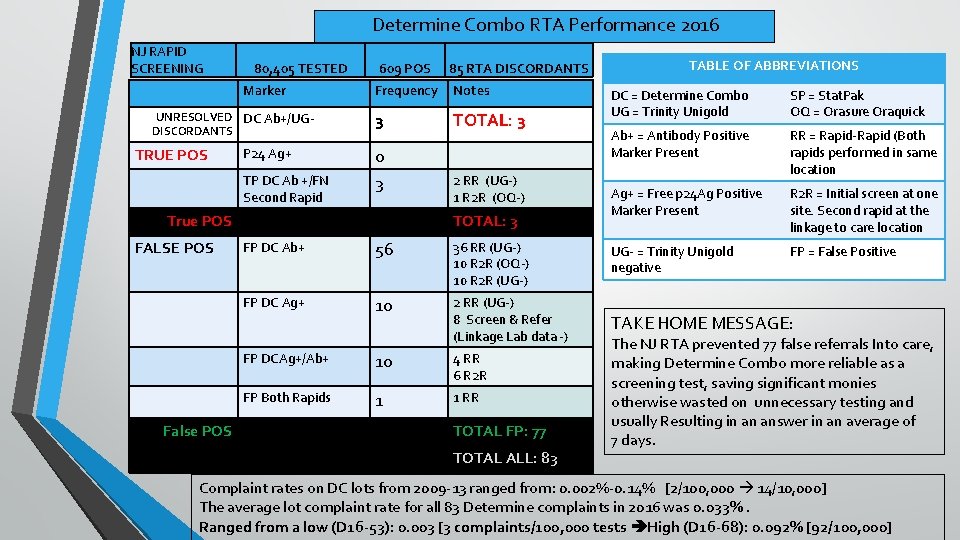
Determine Combo RTA Performance 2016 NJ RAPID SCREENING 80, 405 TESTED Marker UNRESOLVED DC Ab+/UGDISCORDANTS TRUE POS P 24 Ag+ TP DC Ab +/FN Second Rapid 609 POS Frequency Notes 3 TOTAL: 3 0 3 True POS FALSE POS False POS 85 RTA DISCORDANTS 2 RR (UG-) 1 R 2 R (OQ-) TOTAL: 3 TABLE OF ABBREVIATIONS DC = Determine Combo UG = Trinity Unigold SP = Stat. Pak OQ = Orasure Oraquick Ab+ = Antibody Positive Marker Present RR = Rapid-Rapid (Both rapids performed in same location Ag+ = Free p 24 Ag Positive Marker Present R 2 R = Initial screen at one site. Second rapid at the linkage to care location FP = False Positive FP DC Ab+ 56 36 RR (UG-) 10 R 2 R (OQ-) 10 R 2 R (UG-) UG- = Trinity Unigold negative FP DC Ag+ 10 2 RR (UG-) 8 Screen & Refer (Linkage Lab data -) TAKE HOME MESSAGE: FP DCAg+/Ab+ 10 4 RR 6 R 2 R FP Both Rapids 1 1 RR TOTAL FP: 77 TOTAL ALL: 83 The NJ RTA prevented 77 false referrals Into care, making Determine Combo more reliable as a screening test, saving significant monies otherwise wasted on unnecessary testing and usually Resulting in an answer in an average of 7 days. Complaint rates on DC lots from 2009 -13 ranged from: 0. 002%-0. 14% [2/100, 000 14/10, 000] The average lot complaint rate for all 83 Determine complaints in 2016 was 0. 033%. Ranged from a low (D 16 -53): 0. 003 [3 complaints/100, 000 tests High (D 16 -68): 0. 092% [92/100, 000]

- Pilots and Projects -

HCV Pilot: SUMMARY • • • With 2015 carry forward funding to support 300 -400 tests, we initiated rapid HCV screening at selected ARCH nurse sites in the spring of 2015. Five sites included: • • • Oasis Drop-in Center – Atlantic City Camden AHEC in Camden NJCRI in Newark Well of Hope in Paterson Hyacinth in Jersey City December 2015: 226 HCV Screen POS (Seroprevalence: 22. %) Concerns: 1. 2. Not enough testing. Nearly 61% of all testing devices were used to satisfy manufacturer’s required internal quality control (54%) or to perform proficiency testing events (7%). Follow-up results: : “NOT REGULARLY REPORTED” has been a problem at ALL SITES! • NJCRI: : Implemented a standing order to permit immediate phlebotomy and lab testing on initial HCV Positives

NJCRI: With Standing Order for Follow-up Testing Since January 1, 2016: 59 Tested, 8 Positives: 4 Lost to Follow-up, 4 Linked to Lab Follow-up and care. Two or four confirmed. Summary: Out of 8 Positive HCV screens only 2 were confirmed as HCV after linkage. 1. Client LI 016289 Client showed up for his appointment, completed lab work and was seen by the Doctor. Client received 14 days’ supply of Zapatier 50 mg-100 mg tablet scheduled for next 14 days awaiting lab 30 days after treatment start. 2. Client JE 227506 Client also showed up for her appointment. Hep C—client genotype 3 positive, needs NS 5 A lab for drug evaluation. 3. 4. Client ME 095418 Client also showed up for his appointment, spoke with the Doctor client stated he had been treated at the VA 6 months ago. Client was sent to the lab for Hep C viral load. HCV was not detected client lab result from labcorp. Client JO 256592 Client also showed up for her appointment, Hep B Ag came back negative. Client is schedule to see Dr Wilard this month. HCV was not detected

Syphilis pilot SUMMARY • October, 2016: Asbury Park VNA began testing, followed, in November, by NJCRI in Newark. • Dec. 31, 2016: Total of 58 screenings were performed identifying 11 positives (19 % seropositivity). • • Five additional sites are scheduled for training and roll-out in Spring, 2017. We will assess the feasibility, acceptance, and performance of the reverse algorithm after additional sites have entered the program

Pooled NAAT SUMMARY HOW MANYDO WE MISS? • Client willingness to have pooled NAAT marginal: <50%? • Hospital recruitment PROBLEMATIC • So Far: 1. 45 pools 2. 1242 specimens 3. 1 Positive (Not AHI – missed HIV Ab+) 4. Previous Study: 1 AHI/ 675 specimens • • Differences from 2010: Collecting Architect data PLUS Pooled HIV 1 RNA) Why? How many are missed and why matters. QA effort: Evaluate the possibility that as many as 94 cases HIV infection are missed annually in screening 80, 000 individuals by rapid HIV testing. After rapid HIV testing, individuals are offered enhanced screening by a laboratory-based 4 th generation screen (Abbott Architect) and pooled nucleic acid amplification testing (NAAT). • Following sites agreed to participate. Sites not yet operational italicized): 1. 2. 3. 4. Hyacinth Foundation, 100 Hamilton Place, Paterson, NJ 5. 6. 7. NJCRI 393 Central Avenue, 3 rd Floor, Newark, NJ Hyacinth Foundation, 194 Clinton Ave. , Lower Level, Newark, NJ Hyacinth Foundation, 194 Clinton Ave. Newark, NJ – Mobile Van Newark Beth Israel Medical Center, 166 Lyons Ave, Room 109, Newark, NJ Paterson Department of Health, 176 Broadway, Paterson, NJ Rutgers – Newark, 150 Bergen Street, C 130 ADMC Rm 614 A, Newark, NJ 8. Rutgers STOP Program, 65 Bergen Street, Room 177, Newark, NJ 9. St. Michael’s Med Center, 111 Central Avenue, Newark, NJ 10. Trinitas Hospital, 654 E. Elizabeth, NJJersey Street, Plaza 1 st Flr (Rm 161), An NJ HIV QA activity supported by: IRB Protocol Number: 0220080007

Other HIV Screening Assays:

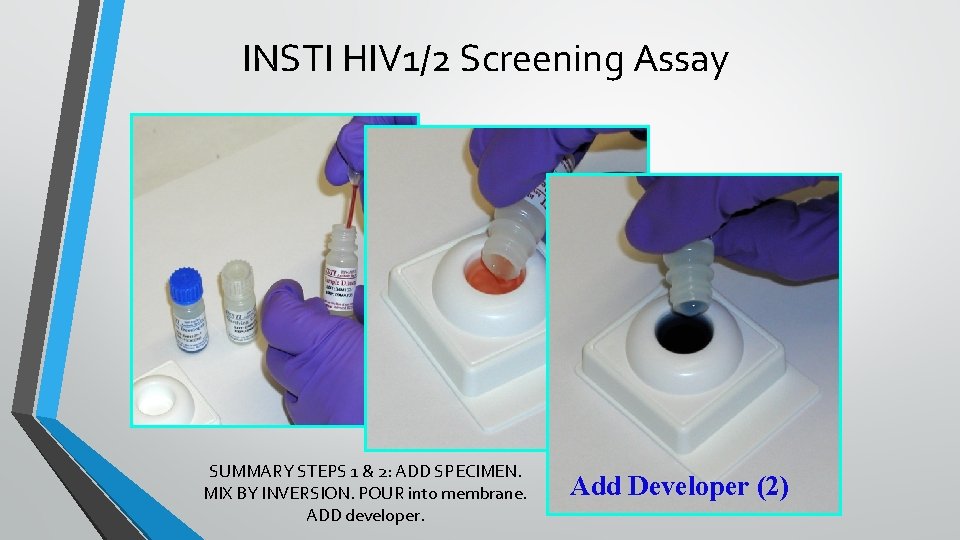
INSTI HIV 1/2 Screening Assay SUMMARY STEPS 1 & 2: ADD SPECIMEN. MIX BY INVERSION. POUR into membrane. ADD developer. Add Developer (2)

INSTI SUMMARY • • • WHY? • • A 1 MIN TEST - MAY HAVE UTILITY IN APPROPRIATE VENUES. REPORTEDLY NEARLY EQUIVALENT IN SENSITIVITY TO THE DC Ab marker CONCERNS: Reservations with regard to multiple steps and reagent order in some venues. WHERE/STATUS: • • • Atlantic. Care Medical Center • • Initial Reaction: Very positive. Second shipment of ~400 devices Outcome: Through March, 2017: 274 NEG, 0 POS Our Lady of Lourdes • Began May 1, 2017 Jersey Shore University Medical Center • • • Counselor trained POC Supervisor validating the assay at JSUMC Start date: May 26, 2016

What’s Next? Silos vs. Collaboration Ebola, Lassa …. HIV "Let us not let the world be defined by the destruction wrought by one virus, but illuminated by billions of hearts and minds working in unity. “ Pardis Sabeti, Ted Talk, 2015 Pardis Sabeti investigates the genomes of microbes, including the Ebola virus, to help understand how to slow them. Why you should listen Pardis Sabeti develops algorithms to detect the genetic signatures of adaption in humans and the microbial organisms that infect humans. Among her lab’s key research areas: examining the genetic factors that drive disease susceptibility to Ebola and Lassa hemorrhagic fever, and investigating the genomes of microbes, including Lassa virus, Ebola virus, Plasmodium falciparum malaria, Vibrio cholera and Mycobacterioum tuberculosis, to help find cures. Pardis Sabeti: TED TALK Ted Women 2015

NJ HIV Mission A Common Quality Assurance Platform for Rapid ID Screening in NJ DEVELOP & SUPPORT QA plan for Rapid ID Tests ASSESSS New ID Screening Tests TECHNICAL OVERSIGHT: Inventory Management Site Visits, Competency Assessments, Proficiency Testing, Technical Support, Liaison Review Performance Validate & Distribute 100, 000 devices per year ~1 M since 2003 Data Analysis IMPLEMENT STATEWIDE: New Rapid Tests CDC Taskforces: AHI, HIV Definition, HIV Confirmatory Algor. Ithim TRAIN >2298 staff trained serving more than 100 sites Publications/Abstracts/P resentations 18/56/34

NJ HIV - Rapid Testing Support RUTGERS – RWJMS Eugene Martin, Ph. D. Co-Director NJ HIV , PI - DHSTS grant Gratian Salaru, MD Co-Director NJ HIV’ PI - DMHAS MOA Parisa Javidian, MD - BLD Tom Kirn, MD, Ph. D. Joanne Corbo, MT (ASCP), MBA Rapid HIV Program Manager Latasha Adams BS, MT Moeen Ahmed BS, MT Aida Gilanchi, BS, MT Nisha Intwala, BS, MT (ASCP) Franchesca Jackson, BS DAS Mobile HIV Counselor • Marianela Moreno Administration: • Lisa May • Karen Williams NJDOH/DHSTS Jihad Slim, MD Medical Director, DHSTS Connie F. Calisti-Meyers Assistant Commissioner, DHSTS Steve Saunders, MS* Director, HIV Prevention Loretta. Dutton Director, HIV Care & Treatment Barbara Bolden, Ph. D. - Surveillance Lisa Jones Cynthia Mimmo Rekha Damaraju Shwetha Kamath Chelsea Betlow Karen Robinson Aye Maung NJ DHS/DMHAS Valerie L. Mielke, MSW Assistant Commissioner, DMHAS Adam Bucon, LSW Office of the Medical Director

NJ ASTHO “VISION” AWARDS – 1 st Place RAPID HIV TESTING PROGRAM PATIENT NAVIGATOR PROGRAM - 2006 - -2013 - 70

- The End -