Textbook Claude CohenTannoudji born 1933 Bernard Diu born

Textbook Claude Cohen-Tannoudji (born 1933) Bernard Diu (born 1935) Franck Laloë (born 1940)

Chapter 1 Introduction

Quantum physics • Problems at the end of XIX century that classical physics couldn’t explain: • Blackbody radiation – electromagnetic radiation emitted by a heated object • Photoelectric effect – emission of electrons by an illuminated metal • Spectral lines – emission of sharp spectral lines by gas atoms in an electric discharge tube 1

Quantum physics • Phenomena occurring on atomic and subatomic scales cannot be explained outside the framework of quantum physics • There are many phenomena revealing quantum behavior on a macroscopic scale, e. g. enables one to understand the very existence of a solid body and parameters associated with it (density, elasticity, etc. ) • However, as of today, there is no satisfactory theory unifying quantum physics and relativistic mechanics • In this course we will discuss non-relativistic quantum mechanics 1

Kindergarten stuff • Is light a wave or a flux of particles? • Newton vs. Young Isaac Newton (1642 – 1727) Thomas Young (1773 – 1829) 1. A. 1
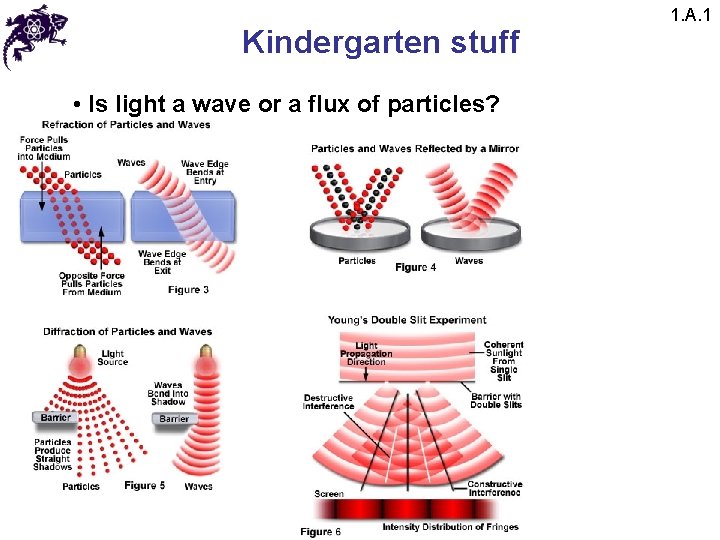
Kindergarten stuff • Is light a wave or a flux of particles? 1. A. 1
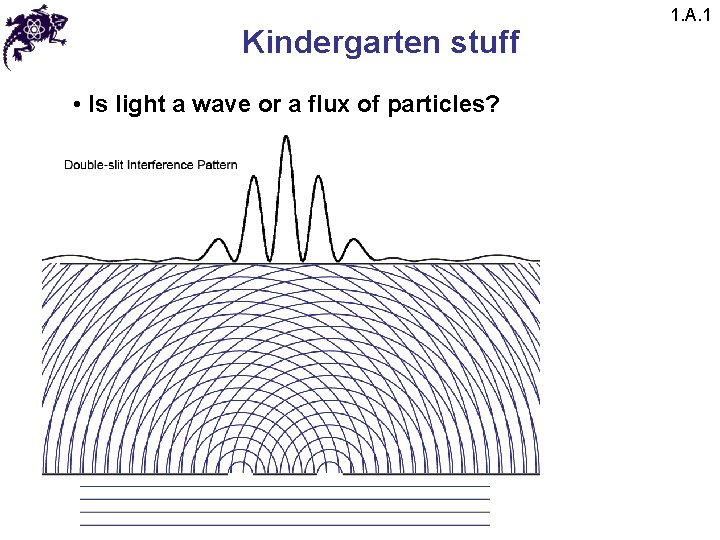
Kindergarten stuff • Is light a wave or a flux of particles? 1. A. 1

Kindergarten stuff • Is light a wave or a flux of particles? 1. A. 1

Kindergarten stuff • Is light a wave or a flux of particles? • However: • 1) Blackbody radiation • 2) Photoelectric effect • 3) Spectral lines • 4) Etc. 1. A. 1

1. A. 1 Wave-particle duality • EM waves appear to consist of particles – photons • Particle and wave parameters are linked by fundamental relationships: • h – Planck’s constant, 6. 626 × 10 -34 J∙s Max Karl Ernst Ludwig Planck 1858 – 1947 Albert Einstein 1879 – 1955
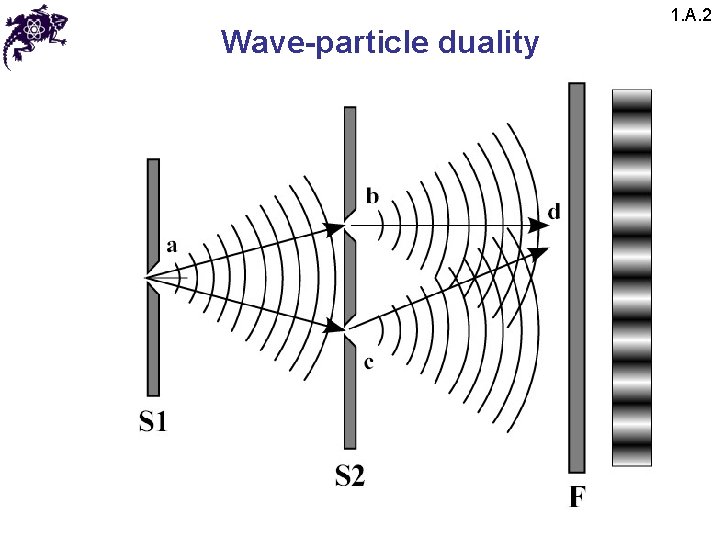
Wave-particle duality 1. A. 2
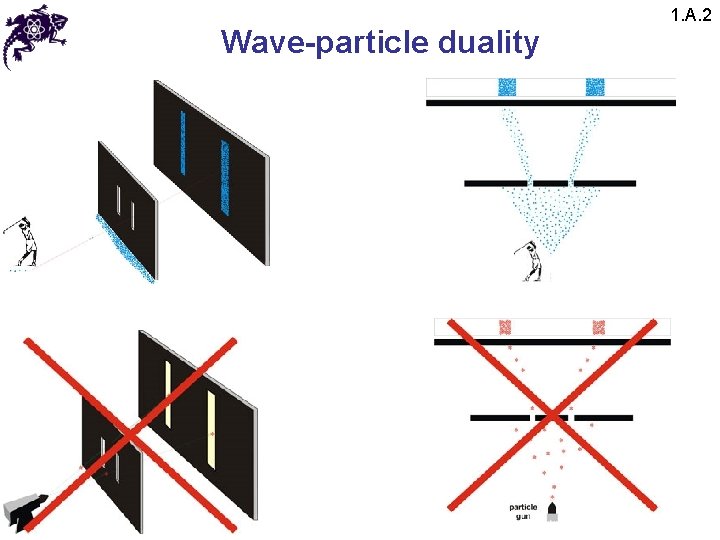
Wave-particle duality 1. A. 2
Wave-particle duality 1. A. 2
Wave-particle duality 1. A. 2
Wave-particle duality 1. A. 2
Wave-particle duality 1. A. 2
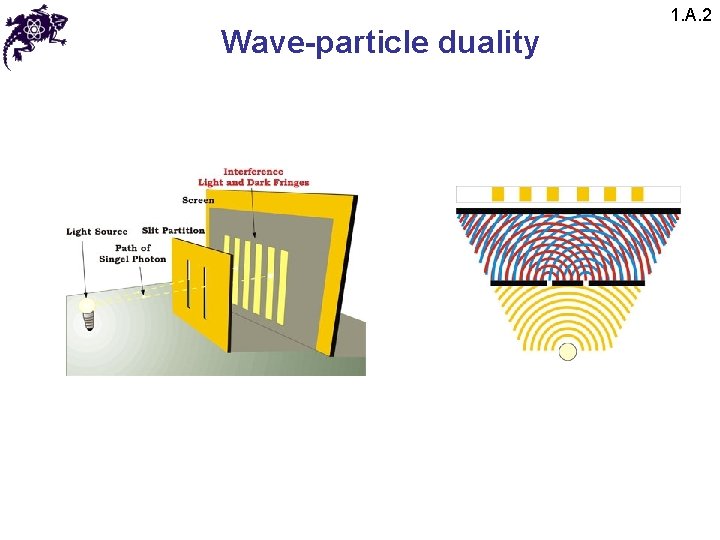
Wave-particle duality 1. A. 2

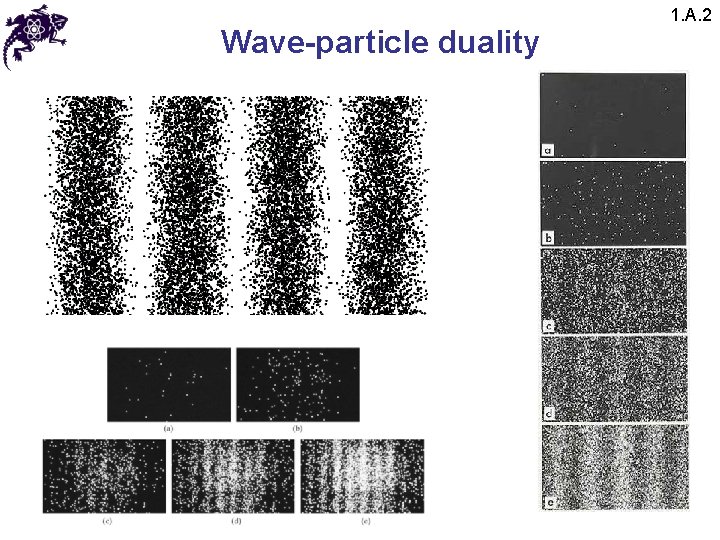
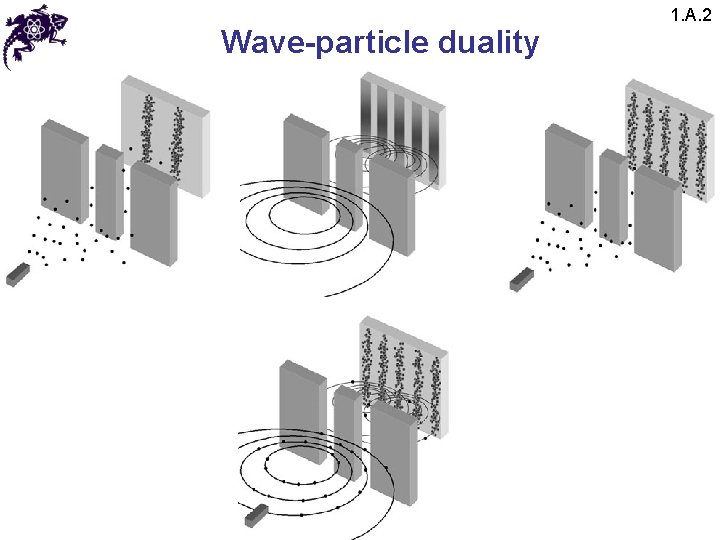
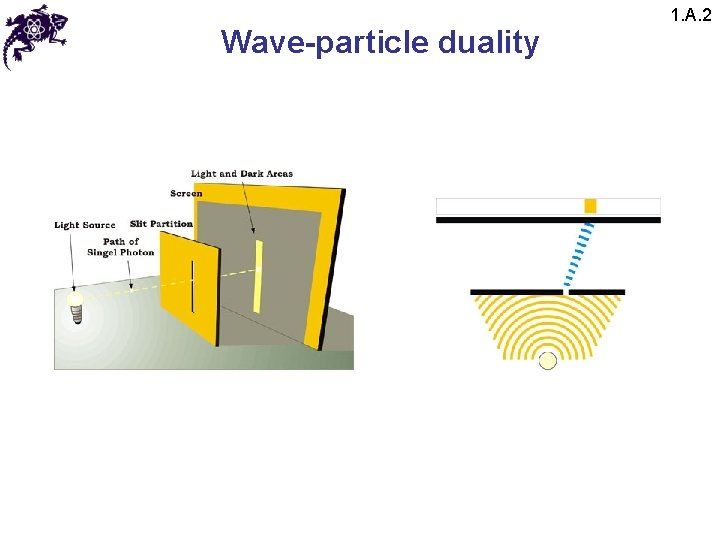
Wave-particle duality 1. A. 2 • The results of this experiment lead to a paradox: • Since the interference pattern disappears when one of the slits is covered, why then this phenomena changes so drastically? • Crucial: the process of measurement • When one performs a measurement on a microscopic system, one disturbs it in a fundamental fashion • It is impossible to observe the interference pattern and to know at the same time through which slit each photon has passed

Wave-particle duality 1. A. 2 • Light behaves simultaneously as a wave and a flux of particles • The wave enables calculation of particle-related probabilities; e. g. , when the photon is emitted, the probability of its striking the screen is proportional to light intensity, which in turn is proportional to the square of the field amplitude

Wave-particle duality 1. A. 2 • Predictions of the behavior of a photon can be only probabilistic: information about the photon at time t is given by the electric field, which is a solution of the Maxwell’s equations – the field is interpreted as a probability amplitude of a photon appearing at time t at a certain location: James Clerk Maxwell 1831 -1879

Principle of spectral decomposition 1. A. 3 • Malus’ Law: the intensity of the polarized beam transmitted through the second polarizing sheet (the analyzer) varies as I = Io cos 2 θ, where Io is the intensity of the polarized wave incident on the analyzer Étienne-Louis Malus 1775 – 1812

Principle of spectral decomposition • What will happen, when intensity is low enough for the photons to reach the analyzer one by one? • NB: the detector does not register “a fraction of a photon”) • We cannot predict which photon can pass the analyzer 1. A. 3

Principle of spectral decomposition 1. A. 3 • The analyzer and detector can give only certain specific results – eigen (proper) results: either a photon passes the analyzer or not • To each of the eigen results there is an eigenstate • When the state before measurement is arbitrary, only the probabilities of obtaining the different eigen results can be predicted • To find these probabilities, the state has to be decomposed into a linear combination of eigenstates

Principle of spectral decomposition 1. A. 3 • The probability of an eigen result is proportional to the square of the absolute value of the coefficient of the corresponding eigenstate • The sum of all the probabilities should be equal to 1 • Measurement disturbs the photons in a fundamental fashion

Wave properties of particles 1. B. 1 • In 1924, Louis de Broglie postulated that because photons have wave and particle characteristics, perhaps all forms of matter have both properties • Furthermore, the frequency and wavelength of matter waves can be determined • The de Broglie wavelength of a particle is • The frequency of matter waves is Louis de Broglie 1892 – 1987

Wave properties of particles 1. B. 1 • The de Broglie equations show the dual nature of matter • Each contains matter concepts (energy and momentum) and wave concepts (wavelength and frequency) • The de Broglie wavelength of a particle is • The frequency of matter waves is Louis de Broglie 1892 – 1987

Wave properties of particles 1. B. 1 • Davisson and Germer scattered low-energy electrons from a nickel target and followed this with extensive diffraction measurements from various materials • The wavelength of the electrons calculated from the diffraction data agreed with the expected de Broglie wavelength Clinton Joseph Davisson (1881 – 1958) and Lester Halbert Germer (1896 – 1971)

The wave function 1. B. 2 • In quantum mechanics the object is described by a state (not trajectory) • The state is characterized by a wave function, ψ, which depends on the particle’s position and the time • The wave function is interpreted as a probability amplitude of quantum object’s presence (recall electric field as a probability amplitude of photon’s presence) • The probability density (probability of finding the object at time t inside an elementary volume dxdydz; C – normalization constant):

The wave function 1. B. 2 • The principle of spectral decomposition applies: • The outcome of a measurement at t 0 must belong to a set of eigen results {a} • An eigenstate (eigenfunction) ψa(r) is associated with each eigenvalue a, if the measurement yields a: • The probability of measuring an eigenvalue a at t 0 can be found by performing spectral decomposition:

The wave function • Important relationships: 1. B. 2

Schrödinger equation 1. B. 2 • In 1926 Schrödinger proposed an equation for the wave function describing the manner in which matter waves change in space and time • Schrödinger equation is a key element in quantum mechanics • V – potential energy (“potential”) • Superposition principle applies Erwin Rudolf Josef Alexander Schrödinger 1892 – 1987

Schrödinger equation • For a free particle: • Solution: • Using the superposition principle: • For 1 D 1. C. 1
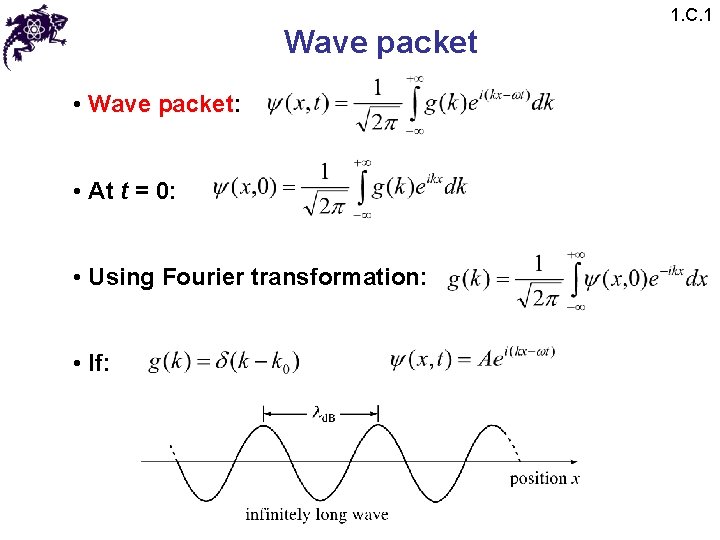
Wave packet • Wave packet: • At t = 0: • Using Fourier transformation: • If: 1. C. 1
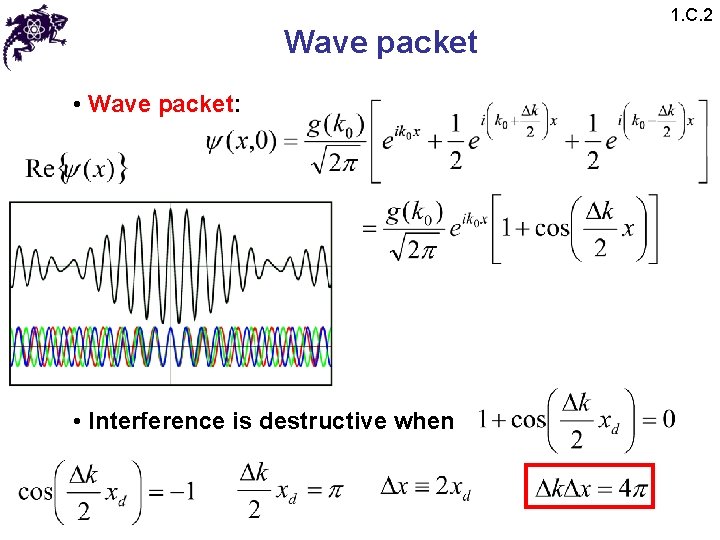
Wave packet • Wave packet: • Interference is destructive when 1. C. 2
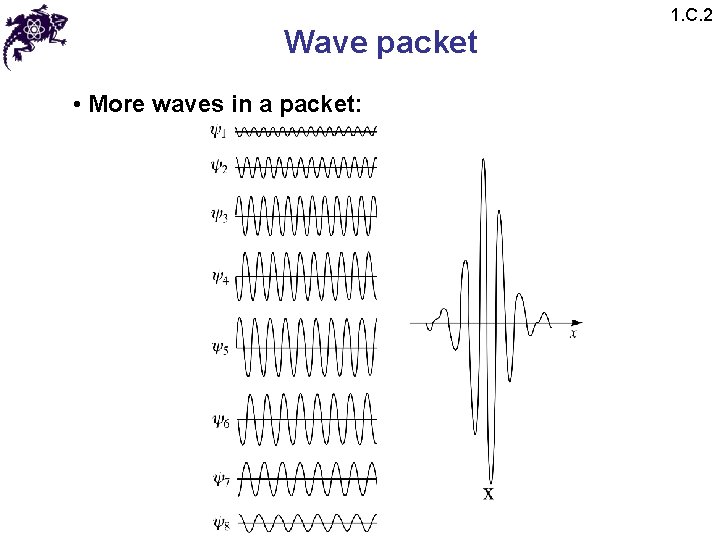
Wave packet • More waves in a packet: 1. C. 2
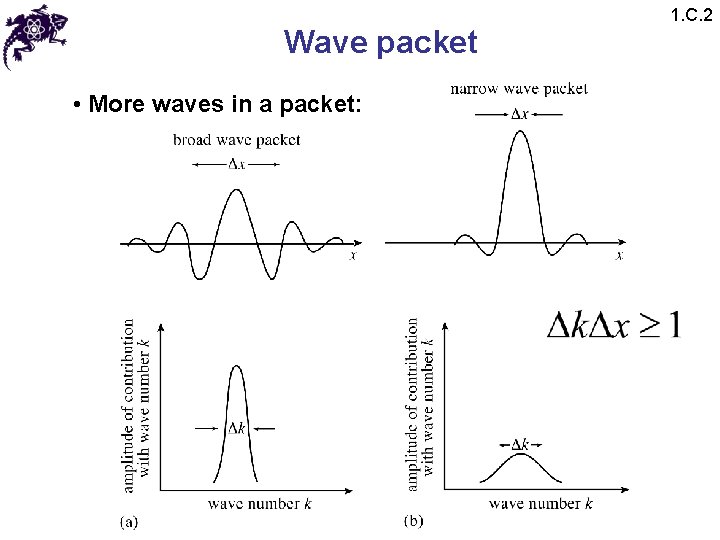
Wave packet • More waves in a packet: 1. C. 2

Wave packet 1. C. 3 • More waves in a packet: • From Fourier calculus: • Probability of finding a particle between x and x+dx : • Probability of measuring a momentum between p and p+dp:

Heisenberg uncertainty principle 1. C. 3 • In 1927 Heisenberg introduced the uncertainty principle: If a measurement of position of a particle is made with precision Δx and a simultaneous measurement of linear momentum is made with precision Δpx, then the product of the two uncertainties can never be smaller than h/2 Werner Karl Heisenberg 1901 – 1976

Evolution of a free packet • For a single wave: • Phase velocity: • Three-wave packet: • Maximum occurs when: 1. C. 4

Evolution of a free packet • For multiple waves in a packet : • Group velocity: 1. C. 4

Particle in a time-independent scalar potential • Schrödinger equation: • Separating variables: 1. D. 1

Particle in a time-independent scalar potential • This is called a stationary solution for a stationary eigenstate 1. D. 1

Particle in a time-independent scalar potential • Introducing linear differential operator • This is an eigenvalue equation for H • Linear superposition of solutions is a solution 1. D. 1

1 D square potentials 1. D. 2
- Slides: 44