Test bank Chapter 5 7 8 9 Dr

Test bank Chapter 5, 7, 8, 9 ﺍﻟﺤﺮﺑﻲ ﻟﻴﻠﻰ. ﺩ ﺇﻋﺪﺍﺩ Dr. Laila Al-Harbi

Which is correct? 1. All gases are fluids. 2. All gases are diatomic. a. b. c. d. Both statements are true. Both statements are false. Statement 1 is true, but statement 2 is false. Statement 1 is false, but statement 2 is true. Dr. Laila Al-Harbi

As a helium-filled balloon rises, its volume increases. This is an example of: a. b. c. d. Avogadro’s Law. Boyle’s Law. Charles’ Law. Gay-Lussac’s Law. Dr. Laila Al-Harbi

Hot air balloons rise. This is an example of: a. b. c. d. Avogadro’s Law. Boyle’s Law. Charles’ Law. Gay-Lussac’s Law. Dr. Laila Al-Harbi

Avogadro’s Law states that the volume of a gas is directly proportional to: a. b. c. d. the number of gas particles. the mass of the gas. the pressure of the gas. the Kelvin temperature of the gas. Dr. Laila Al-Harbi

Standard temperature and pressure (STP) for a gas is: a. b. c. d. 0 atmospheres and 25 degrees C. 1 atmosphere and 0 degrees C. (273 K) 1 atmosphere and 25 degrees C. 0 atmospheres and 0 degrees C. Dr. Laila Al-Harbi

The partial pressure of each gas in a mixture is proportional to: a. the mass of the gas. b. the molecular weight of the gas. c. the square root of the molecular weight of the gas. d. the mole fraction of the gas. Dr. Laila Al-Harbi

4. Under conditions of fixed temperature and amount of gas, Boyle's law requires that I. P 1 V 1 = P 2 V 2 II. PV = constant III. P 1/P 2 = V 2/V 1 (a) I only (b) II only (c) III only (d) I, II, and III (e) another combination Dr. Laila Al-Harbi

A sample of two moles of nitrogen gas (N 2) will occupy what volume at STP? PV=n. RT = 2 x 0. 082 x 273/1= a. b. c. d. 11. 2 liters 22. 4 liters 44. 8 liters 89. 6 liters Dr. Laila Al-Harbi

• A sample of hydrogen gas was collected over water at 21 C and 0. 9 atm. The volume of the container was 7. 80 L. Calculate the mass of H 2(g) collected. (Vapor pressure of water = 0. 025 atm at 21 C. ) • (A) 0. 283 g • (B) 435 g • (C) 0. 571 g • (D) 7. 14 g Dr. Laila Al-Harbi

If 250 m. L of NO is placed in a flask with O 2, what volume of O 2 is needed for complete reaction? 2 NO (g) + O 2 (g) 2 NO 2 (g) From the equation because all reactant are gases 250 x 1 /2 = 125 • • • 100 m. L 125 m. L 200 m. L 250 m. L Cannot be determined from the given information. Dr. Laila Al-Harbi

If an equal mass of each gas is put into a separate balloon, which will have the greatest volume? Assume that they are all the same temperature and pressure. • • • He (ℳ =2) H 2 (ℳ =1) N 2 (ℳ =28) Ne (ℳ =20) O 2 (ℳ =32) Assume that they are all the same temperature and pressure. PV =(m/ℳ ) RT → V ⋉ 1/ ℳ Sine all other variables are constants Dr. Laila Al-Harbi

If equal masses of CH 4, C 2 H 6, and C 3 H 8 are placed in a flask, which of the following is true? • • • PCH 4 = PC 2 H 6 = PC 3 H 8 PCH 4 ~ PC 2 H 6 ~ PC 3 H 8 PCH 4 > PC 2 H 6 > PC 3 H 8 PCH 4 < PC 2 H 6< PC 3 H 8 None of the above PV =(m/ℳ ) RT Dr. Laila Al-Harbi

Chapter 7 Dr. Laila Al-Harbi

The wavelength of electromagnetic energy multiplied by its frequency equals: ν = c/λ → ν λ=c a. c, the speed of light b. h, Planck’s constant c. Avogadro’s number d. 4. 184 Dr. Laila Al-Harbi

The energy of a photon of electromagnetic energy divided by its frequency equals: E =hν → E/ν = h a. c, the speed of light b. h, Planck’s constant c. Avogadro’s number d. 4. 184 Dr. Laila Al-Harbi

Light that contains colors of all wavelengths is called: a. b. c. d. a continuous spectrum. monochromatic. a line spectrum. a Balmer series. Dr. Laila Al-Harbi

The lowest energy state of an atom is referred to as its: a. b. c. d. bottom state. ground state. fundamental state. original state. Dr. Laila Al-Harbi

All s orbitals are: a. b. c. d. shaped like four-leaf clovers. dumbbell-shaped. spherical. triangular. Dr. Laila Al-Harbi

The way electrons are distributed among the various orbitals of an atom is referred to as the atom’s: a. b. c. d. orbital diagram. electron configuration. electron distribution. electron spread. Dr. Laila Al-Harbi
![[He]2 s 22 p 2 is the electron configuration of which element? a. b. [He]2 s 22 p 2 is the electron configuration of which element? a. b.](http://slidetodoc.com/presentation_image/4bf296ed789e757c36a0d24d6fffed0a/image-21.jpg)
[He]2 s 22 p 2 is the electron configuration of which element? a. b. c. d. beryllium boron carbon nitrogen Dr. Laila Al-Harbi

Considering only the n = 1 to n = 5 states in the hydrogen atom, which transition will emit the longest wavelength? • n • • = 5 to n = 4 n = 5 to n = 2 n = 3 to n = 1 n = 3 to n = 2 n = 4 to n = 2 E =hν → E & h are directly proportional E =hc/λ → E & λ are inversely proportional Dr. Laila Al-Harbi

What are the valence electrons of vanadium (V)? • • • 4 s 2 3 d 3 4 s 23 d 3 3 d 5 4 d 5 Dr. Laila Al-Harbi

What are the valence electrons of gallium Ga? • • • 4 s 2 4 p 1 4 s 23 d 10 4 s 24 p 1 Dr. Laila Al-Harbi

What is the maximum number of orbitals described by the quantum numbers: • • • 1 3 5 8 9 n=3 l=2 the maximum number of orbitals = 2ℓ+1=2 x 2+1=5 Dr. Laila Al-Harbi

What is the maximum number of electrons described by the quantum numbers: • • • 7 14 16 32 48 n =4 the maximum number of electrons = 2 n 2 =2(4) 2 =32 Dr. Laila Al-Harbi

How many unpaired electrons does selenium Se have? • • • 4 s 24 p 4 0 2 4 6 8 Dr. Laila Al-Harbi

How many unpaired electrons does chromium have? • • • 0 2 4 6 8 4 s 13 d 5 Dr. Laila Al-Harbi
![The electron configuration of a neutral atom is [Ne] 3 s 2 3 p The electron configuration of a neutral atom is [Ne] 3 s 2 3 p](http://slidetodoc.com/presentation_image/4bf296ed789e757c36a0d24d6fffed0a/image-29.jpg)
The electron configuration of a neutral atom is [Ne] 3 s 2 3 p 1. The four quantum numbers of the last electron are: (A) (2, 1, -1, +1/2) (B) (3, 3, -1 , +1/2) (C) (3, 0, -1, +1/2) (D) (3, 1, -1, +1/2 )# Dr. Laila Al-Harbi

Chapter 8 Dr. Laila Al-Harbi
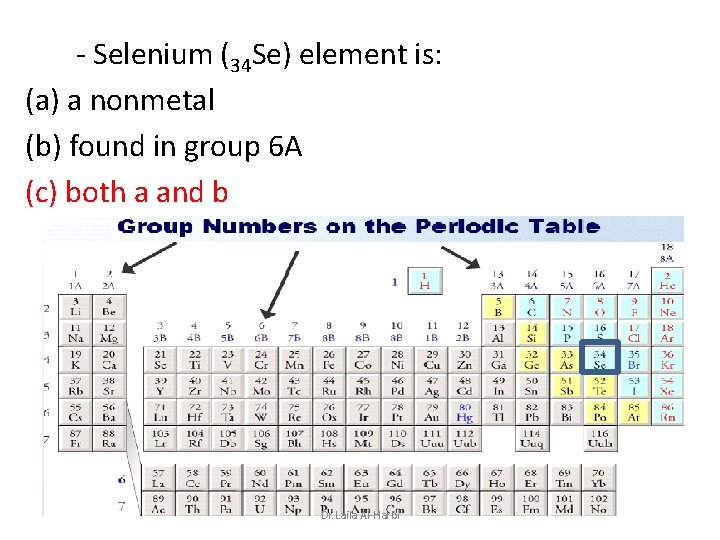
- Selenium (34 Se) element is: (a) a nonmetal (b) found in group 6 A (c) both a and b Dr. Laila Al-Harbi

• The outer electron configuration of the noble gases is (a) ns 2 np 6 (b) ns 2 nd 10 (c) ns 2 np 4 • Which of the following species is isoelectronic with Cl(a) F- (b) O 2 - (c) K+ (d) Na+ Dr. Laila Al-Harbi
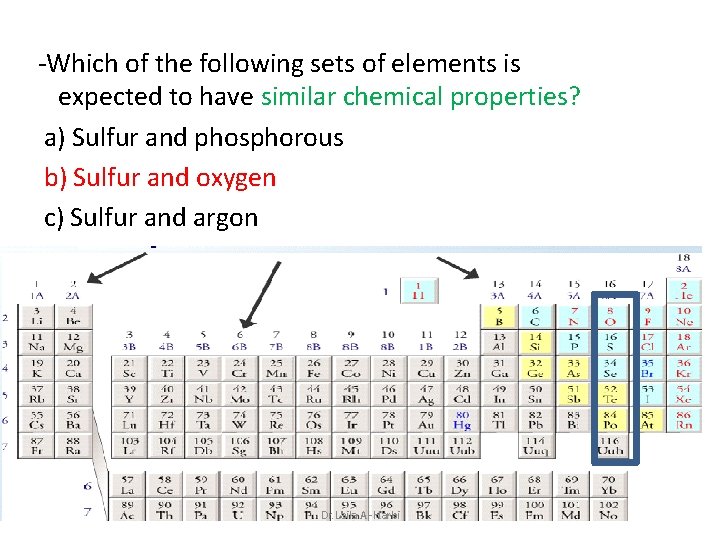
-Which of the following sets of elements is expected to have similar chemical properties? a) Sulfur and phosphorous b) Sulfur and oxygen c) Sulfur and argon Dr. Laila Al-Harbi
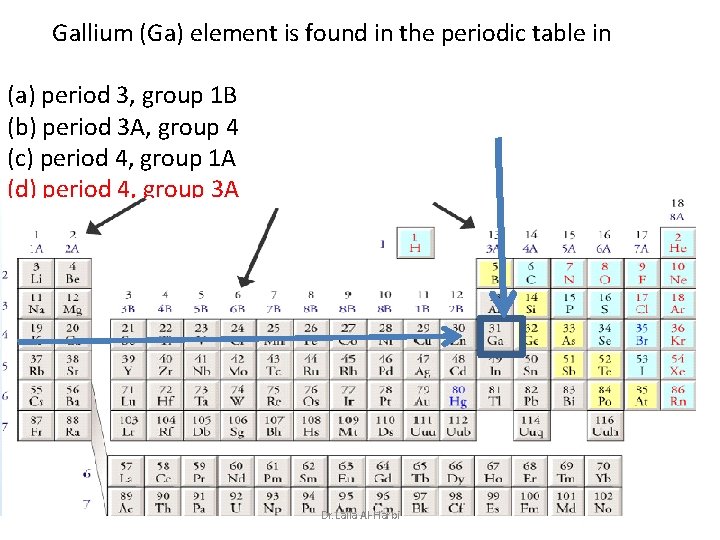
Gallium (Ga) element is found in the periodic table in (a) period 3, group 1 B (b) period 3 A, group 4 (c) period 4, group 1 A (d) period 4, group 3 A Dr. Laila Al-Harbi
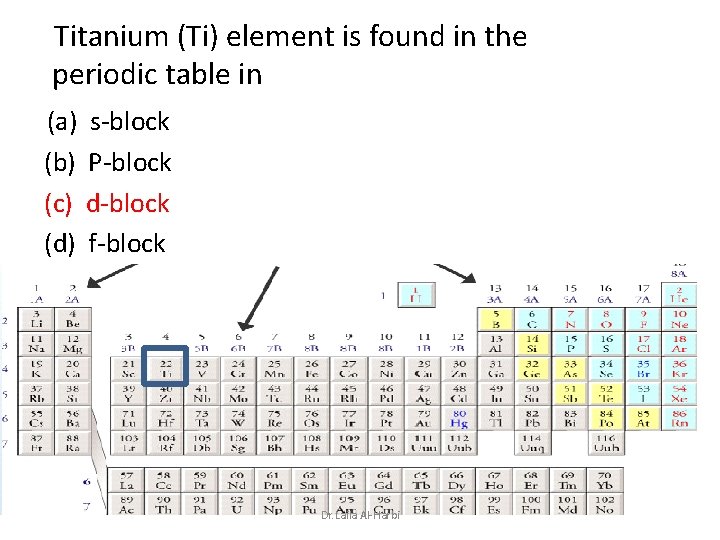
Titanium (Ti) element is found in the periodic table in (a) s-block (b) P-block (c) d-block (d) f-block Dr. Laila Al-Harbi

Choose the incorrect quantum numbers for electron in an atom. A) (3, 0, +1/2). (B) (4, 3, – 2, +1/2). (C) (1, 0, -1/2). Correct (1, 0, 0, -1/2) (D) (3, 2, 0, +1/2) ( Dr. Laila Al-Harbi
![• Write the electronic configuration for Co 2+ (a) [Ar] 4 s 2 • Write the electronic configuration for Co 2+ (a) [Ar] 4 s 2](http://slidetodoc.com/presentation_image/4bf296ed789e757c36a0d24d6fffed0a/image-37.jpg)
• Write the electronic configuration for Co 2+ (a) [Ar] 4 s 2 3 d 5 (b) [Ar] 3 d 7 (c) [Ar] 4 s 1 3 d 6 § Select the correct order of radius of the two ions (A) A+ > A(B) A- < A (C) A 2+ > A+ (D) A 2+ < A+ Dr. Laila Al-Harbi

Which choice below correctly lists the elements in order of increasing atomic radii? a. b. c. d. Na < Mg < K < Rb Mg < Na < K < Rb Rb < K < Na < Mg Rb < K < Mg < Na Dr. Laila Al-Harbi

Two ions are referred to as isoelectronic if they have the same number of: a. b. c. d. electrons. protons. atoms. neutrons. Dr. Laila Al-Harbi

Which statement is false? a. Fluoride ions are larger than rubidium ions. b. Bromide ions are larger than sodium ions. c. Iodide ions are larger than rubidium ions. d. Chloride ions are larger than potassium ions. Dr. Laila Al-Harbi

The energy required to remove an electron from an atom in its ground state is known as the: a. b. c. d. potential energy. activation energy. electron affinity. ionization energy. Dr. Laila Al-Harbi

Which choice correctly lists the elements in order of increasing electron affinity (most negative to least negative)? a. b. c. d. O, Cl, B, C O, Cl, C, B Cl, O, B, C Dr. Laila Al-Harbi

Which choice correctly lists the elements in order of increasing first ionization energy? a. b. c. d. Na < Ca < Al < Sn Na < Al < Ca < Sn Na < Al < Sn < Ca Ca < Na < Sn < Al Dr. Laila Al-Harbi

An unknown solid gives a green flame test. When it was dissolved in water and reacted with Na. OCl, a purple color appeared. The unknown is: a. b. c. d. barium bromide. calcium iodide. barium iodide. calcium bromide. Dr. Laila Al-Harbi

Characteristics of noble gases include: a. b. c. d. filled s and p subshells. monatomic gases. generally unreactive chemically. all of the above. Dr. Laila Al-Harbi

In an germanium atom, which electron will experience the greatest effective nuclear charge? An electron in a 1. 2. 3. 4. 5. 2 s orbital. 2 p orbital. 3 s orbital. 3 p orbital. 3 d orbital. Dr. Laila Al-Harbi
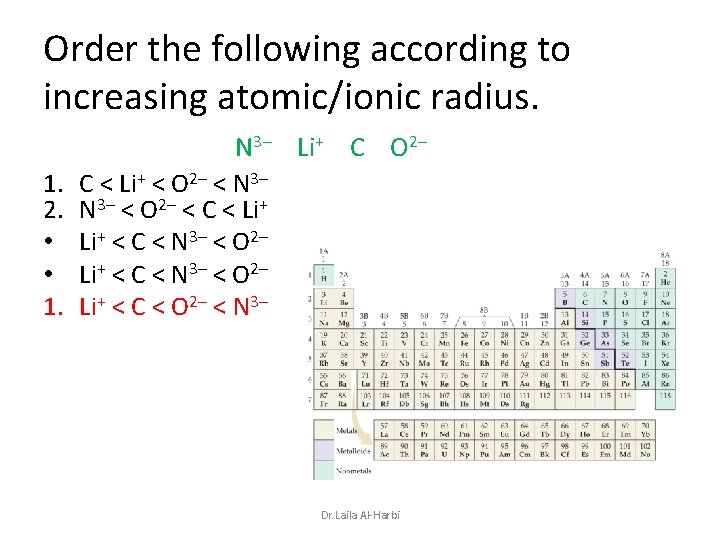
Order the following according to increasing atomic/ionic radius. 1. 2. • • 1. N 3– Li+ C O 2– C < Li+ < O 2– < N 3– < O 2– < C < Li+ < C < N 3– < O 2– Li+ < C < O 2– < N 3– Dr. Laila Al-Harbi

Which will have the highest ionization energy? • • • C N O Al Si Dr. Laila Al-Harbi
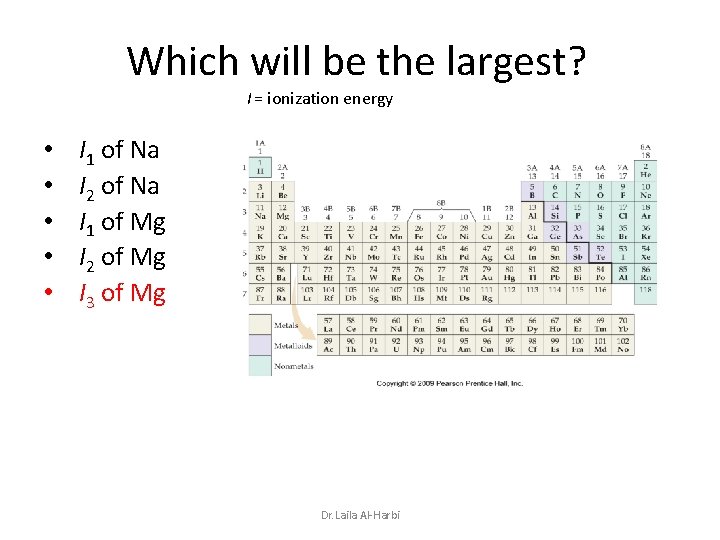
Which will be the largest? I = ionization energy • • • I 1 of Na I 2 of Na I 1 of Mg I 2 of Mg I 3 of Mg Dr. Laila Al-Harbi

Chapter 9 Dr. Laila Al-Harbi

The two types of chemical bonds commonly found in compounds are: a. b. c. d. doric and covalent. ionic and electrolytic. ionic and covalent. electrolytic and compound. Dr. Laila Al-Harbi

The electrons used by atoms to form chemical bonds are the: a. b. c. d. core electrons. valence electrons. lone pair electrons. unpaired electrons. Dr. Laila Al-Harbi

“Atoms tend to gain, lose, or share electrons until they are surrounded by eight valence electrons” is a statement of: a. b. c. d. the rule of octaves. the double quartet rule. the eight electron rule. the octet rule. Dr. Laila Al-Harbi

When a transition metal atom becomes a +1 ion, the electron lost usually comes from what type of orbital? a. p b. f c. d d. s Dr. Laila Al-Harbi

A molecule of CS 2 contains: a. b. c. d. two single bonds. two double bonds. one single bond and one double bond. one single bond and one triple bond. Dr. Laila Al-Harbi

• An atom in the ground state has atomic number Z=5. Choose the correct electron-dot structure which represents this atom? Dr. Laila Al-Harbi

What is the formal charge on the nitrogen atom in the nitrate ion (NO 3– 1) ? a. b. c. d. +2 +1 0 – 1 Dr. Laila Al-Harbi

Which compound below contains an atom that is surrounded by more than an octet of electrons? a. b. c. d. PF 5 CH 4 NBr 3 OF 2 Dr. Laila Al-Harbi

Which choice below correctly lists the elements in order of increasing electronegativity? a. b. c. d. C < N < O < F N < C < F < O C < N < F < O Dr. Laila Al-Harbi

As the number of bonds between two atoms increases, the distance between the atoms: a. b. c. d. increases. decreases. remains unchanged. varies with the atoms involved. Dr. Laila Al-Harbi

Which atom sometimes violates the octet rule? 1. C 2. N 3. O 4. F 5. S Dr. Laila Al-Harbi
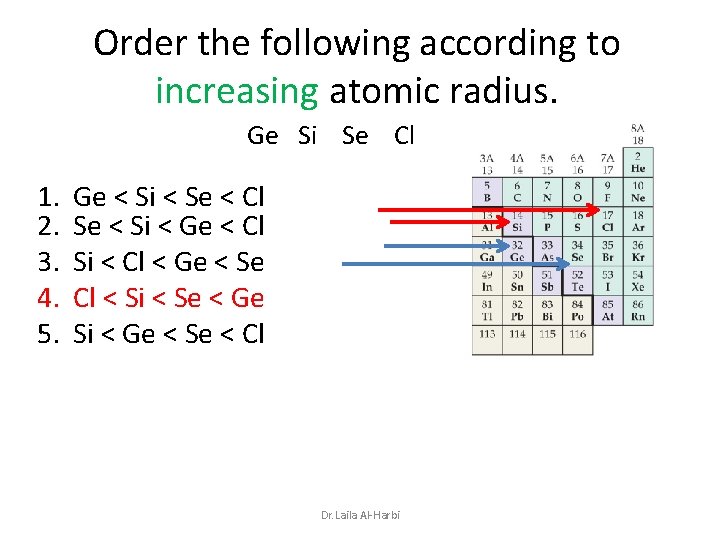
Order the following according to increasing atomic radius. Ge Si Se Cl 1. 2. 3. 4. 5. Ge < Si < Se < Cl Se < Si < Ge < Cl Si < Cl < Ge < Se Cl < Si < Se < Ge Si < Ge < Se < Cl Dr. Laila Al-Harbi
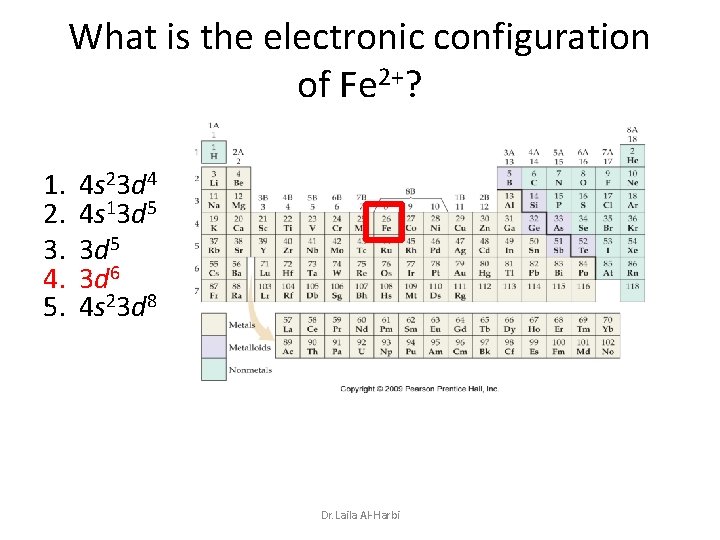
What is the electronic configuration of Fe 2+? 1. 2. 3. 4. 5. 4 s 23 d 4 4 s 13 d 5 3 d 6 4 s 23 d 8 Dr. Laila Al-Harbi

How many unpaired electrons are there in Cu+? 1. 2. 3. 4. 5. 0 1 2 3 4 Dr. Laila Al-Harbi
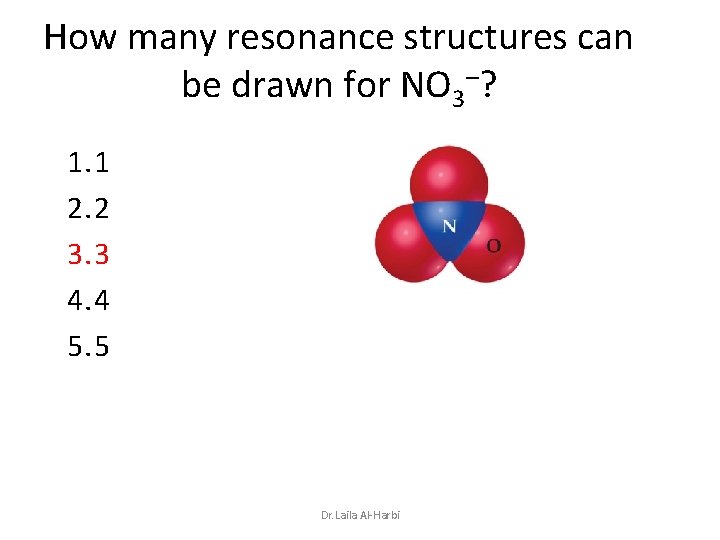
How many resonance structures can be drawn for NO 3–? 1. 1 2. 2 3. 3 4. 4 5. 5 Dr. Laila Al-Harbi
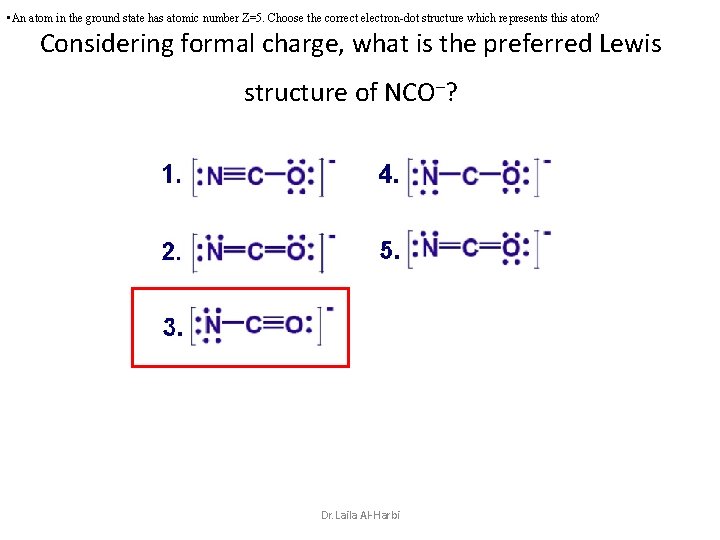
• An atom in the ground state has atomic number Z=5. Choose the correct electron-dot structure which represents this atom? Considering formal charge, what is the preferred Lewis structure of NCO–? Dr. Laila Al-Harbi

- : : : _ _ : : • In Lewis structure of (SO 4)-2 structure the correct formal charge on sulfur (S) is: a) +2 b) -2 : O: +2 c) +1 6 4 : O-S-O : _ d) 0 : : O: Dr. Laila Al-Harbi +2
- Slides: 67